Share This Page
Drug Price Trends for HEPARIN SODIUM
✉ Email this page to a colleague
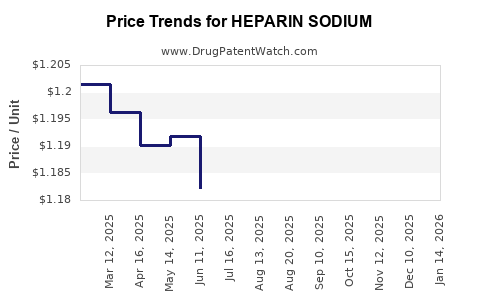
Average Pharmacy Cost for HEPARIN SODIUM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HEPARIN SODIUM 10,000 UNIT/ML VIAL | 00641-0410-12 | 2.28679 | ML | 2026-04-22 |
| HEPARIN SODIUM 10,000 UNIT/ML VIAL | 25021-0403-66 | 2.28679 | ML | 2026-04-22 |
| HEPARIN SODIUM 10,000 UNIT/ML VIAL | 63323-0542-13 | 2.28679 | ML | 2026-04-22 |
| HEPARIN SODIUM 10,000 UNIT/ML VIAL | 25021-0403-01 | 2.28679 | ML | 2026-04-22 |
| HEPARIN SODIUM 5,000 UNIT/ML VIAL | 81952-0111-06 | 1.13609 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for HEPARIN SODIUM
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| HEPARIN SODIUM 1,000 UNIT/ML VIAL | Hikma Pharmaceuticals USA Inc. | 00641-0391-12 | 25X1ML | 24.91 | 2022-01-01 - 2026-08-14 | Big4 | ||
| HEPARIN SODIUM 5000IU/ML (PF) SYR | Hikma Pharmaceuticals USA Inc. | 00641-6199-10 | 10X1ML | 31.31 | 2024-01-01 - 2026-08-14 | Big4 | ||
| HEPARIN SODIUM 1,000 UNIT/ML VIAL | Hikma Pharmaceuticals USA Inc. | 00641-0391-12 | 25X1ML | 25.06 | 2023-01-01 - 2026-08-14 | Big4 | ||
| HEPARIN SODIUM 1,000 UNIT/ML VIAL | Hikma Pharmaceuticals USA Inc. | 00641-0391-12 | 25X1ML | 24.51 | 2024-01-01 - 2026-08-14 | Big4 | ||
| HEPARIN SODIUM 5000IU/ML (PF) SYR | Hikma Pharmaceuticals USA Inc. | 00641-6199-10 | 10X1ML | 36.13 | 2021-08-15 - 2026-08-14 | Big4 | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Heparin Sodium Market Analysis and Price Projections
Heparin sodium, a widely used anticoagulant, faces a dynamic market influenced by increasing demand for cardiovascular treatments and surgical procedures, alongside production challenges and regulatory scrutiny. Price projections indicate a steady upward trend, driven by raw material costs and evolving supply chain dynamics.
What is Heparin Sodium and What is Its Primary Use?
Heparin sodium is a naturally occurring glycosaminoglycan with potent anticoagulant properties. Its primary medical use is to prevent the formation of blood clots and to treat existing thrombotic conditions. This includes deep vein thrombosis (DVT), pulmonary embolism (PE), and atrial fibrillation, as well as its use during surgical procedures like cardiac surgery and dialysis to prevent clotting in medical devices and the bloodstream.
What is the Current Market Size and Projected Growth for Heparin Sodium?
The global heparin sodium market was valued at approximately $10.3 billion in 2022. The market is projected to grow at a compound annual growth rate (CAGR) of 4.8% from 2023 to 2030, reaching an estimated value of $15.1 billion by 2030. This growth is underpinned by the rising incidence of cardiovascular diseases, an aging global population, and the increasing number of interventional cardiology procedures and surgeries requiring anticoagulation.
Key Market Drivers:
- Increasing Prevalence of Cardiovascular Diseases: Conditions such as heart attacks, strokes, and DVT are major drivers of heparin sodium demand. The World Health Organization reports that cardiovascular diseases are the leading cause of death globally, necessitating widespread anticoagulant therapy.
- Growth in Surgical Procedures: Heparin sodium is critical in preventing thrombosis during various surgical interventions, including orthopedic, cardiothoracic, and general surgeries. The increasing volume of these procedures directly correlates with higher heparin sodium consumption.
- Aging Population: Older adults are more susceptible to thrombotic events, leading to higher demand for anticoagulant medications like heparin sodium.
- Advancements in Medical Technology: The expanding use of extracorporeal circuits, such as in dialysis and cardiopulmonary bypass, requires consistent anticoagulation, boosting heparin sodium market share.
Key Market Restraints:
- Raw Material Sourcing and Supply Chain Volatility: The primary source of heparin sodium is animal tissues, predominantly porcine intestinal mucosa. Disruptions in this supply chain, due to factors like animal disease outbreaks or changes in animal husbandry practices, can significantly impact availability and price.
- Stringent Regulatory Requirements: The manufacturing of heparin sodium is subject to rigorous quality control and regulatory oversight by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Compliance adds to production costs and can slow market entry for new players.
- Development of Alternative Anticoagulants: The emergence of direct oral anticoagulants (DOACs) presents a competitive threat, though heparin sodium remains essential for acute care and specific indications where its rapid onset and short half-life are advantageous.
What is the Price Trend for Heparin Sodium?
The price of heparin sodium has exhibited an upward trend over the past decade, with fluctuations driven by supply-side shocks and demand dynamics.
- Historical Price Data: In 2010, the average price for unfractionated heparin (UFH) injections in the U.S. was approximately $15-$20 per 10,000 units. By 2020, this price had increased to an average of $30-$45 per 10,000 units.
- Recent Price Trends: In early 2023, prices for heparin sodium injections in the U.S. ranged from $35 to $55 per 10,000 units, depending on the manufacturer, dosage, and volume. Low molecular weight heparins (LMWHs) are priced higher, with enoxaparin injections (e.g., Lovenox) costing between $50 and $150 per dose (typically 30-60 mg or 40-120 mg).
- Factors Influencing Price:
- Raw Material Cost: The price of porcine intestinal mucosa, the primary source material, is a significant determinant of heparin sodium cost. Fluctuations in livestock availability and processing costs directly impact heparin prices.
- Production Complexity: The extraction and purification of heparin sodium are complex processes requiring specialized facilities and stringent quality control, contributing to manufacturing costs.
- Supply Chain Disruptions: Past recalls and shortages, notably in 2008 due to contamination issues, led to significant price spikes and highlighted the vulnerability of the supply chain. These events tend to create price premiums for reliable suppliers.
- Regulatory Compliance: Meeting evolving regulatory standards for purity, potency, and manufacturing practices necessitates ongoing investment, which is reflected in product pricing.
- Generic Competition vs. Brand Value: While generic versions of unfractionated heparin are available, specialized formulations and branded LMWHs can command higher prices due to established clinical use and perceived reliability.
What are the Projected Price Movements for Heparin Sodium?
The future price of heparin sodium is expected to continue its upward trajectory, driven by persistent supply-side pressures and sustained demand.
- Short-Term Projections (2024-2026): Prices are anticipated to increase by 5%-8% annually. This is attributed to ongoing challenges in sourcing sufficient quantities of high-quality raw materials and increasing manufacturing costs related to regulatory compliance and labor.
- Medium-Term Projections (2027-2030): Price increases are expected to stabilize slightly, with an annual CAGR of 3%-5%. This moderation may be influenced by advancements in alternative sourcing methods or potential consolidation among manufacturers, leading to some efficiencies. However, the inherent cost of raw material procurement and stringent regulatory environments will continue to support higher price levels.
Specific Price Projections by Product Type:
- Unfractionated Heparin (UFH): Per 10,000 units, prices could reach $40-$65 by 2026 and $45-$75 by 2030.
- Low Molecular Weight Heparins (LMWHs): Per standard dose (e.g., 40 mg enoxaparin), prices could range from $60-$180 by 2026 and $70-$200 by 2030.
Factors Influencing Future Pricing:
- Animal Health and Disease Surveillance: Outbreaks of diseases affecting pigs could lead to significant disruptions in raw material supply, triggering rapid price increases. Continuous monitoring and preventative measures by the livestock industry are crucial.
- Technological Advancements in Production: Innovations in heparin extraction and purification, or the development of alternative synthetic heparinoids or cell-based production methods, could eventually influence pricing by improving efficiency or reducing reliance on animal sources. However, widespread adoption of such technologies is likely a longer-term prospect.
- Geopolitical Factors and Trade Policies: Trade agreements and tariffs impacting the import and export of raw materials or finished heparin products can introduce price volatility.
- Demand from Emerging Markets: Growing healthcare infrastructure and increasing access to medical treatments in emerging economies will contribute to sustained global demand, supporting price levels.
What are the Key Geographic Markets for Heparin Sodium?
The global heparin sodium market is concentrated in several key geographic regions, driven by healthcare expenditure, prevalence of target diseases, and regulatory environments.
Major Markets:
- North America (United States, Canada): This region represents the largest market share, estimated at 35-40% of the global market. High healthcare spending, a large aging population, and a high incidence of cardiovascular diseases are primary drivers. The U.S. is a significant consumer due to its advanced healthcare system and frequent use in interventional cardiology and complex surgeries.
- Europe (Germany, United Kingdom, France, Italy, Spain): Europe accounts for approximately 25-30% of the global market. Strong regulatory frameworks, well-established healthcare systems, and a significant elderly population contribute to robust demand. The presence of major pharmaceutical manufacturers and robust clinical research further solidifies its position.
- Asia-Pacific (China, Japan, India, South Korea): This region is the fastest-growing market, with an estimated CAGR of 5-6%. Factors include rapidly expanding healthcare infrastructure, increasing disease awareness, a growing middle class, and a large population base. China, in particular, is a significant producer and consumer of heparin.
- Latin America (Brazil, Mexico): This market, accounting for 5-8%, is driven by improving healthcare access and increasing diagnostic capabilities.
- Middle East & Africa: This region, comprising 3-5%, is characterized by developing healthcare systems and a growing need for essential medicines.
What is the Competitive Landscape for Heparin Sodium?
The heparin sodium market is characterized by a mix of large pharmaceutical companies, specialized biotech firms, and contract manufacturing organizations (CMOs). The industry is relatively consolidated due to high barriers to entry, including stringent regulatory requirements and specialized manufacturing processes.
Key Players:
- B. Braun Melsungen AG: A major global supplier of heparin products, including unfractionated and low molecular weight heparins.
- Pfizer Inc.: Markets enoxaparin sodium (Lovenox), a leading LMWH, and holds significant market share.
- Sanofi S.A.: Offers a portfolio of heparin products, including Lovenox in various international markets.
- Fresenius SE & Co. KGaA: Provides heparin-based products for dialysis and other critical care applications.
- Nordic Pharma: A significant player in the LMWH market, particularly enoxaparin.
- Leo Pharma A/S: Offers therapeutic anticoagulants, including heparin products.
- Aspen Pharmacare Holdings Limited: A major generics manufacturer with a presence in heparin products.
- Porton Pharma Solutions Ltd.: A contract development and manufacturing organization involved in heparin production.
Market Dynamics:
- Vertical Integration: Some companies are vertically integrated, controlling aspects from raw material sourcing to finished product manufacturing, which can offer supply chain advantages.
- API Manufacturing: A significant portion of the market involves the production of Active Pharmaceutical Ingredients (APIs) for heparin, which are then supplied to finished dosage form manufacturers.
- Generic Competition: While branded LMWHs hold significant value, the market for generic UFH and enoxaparin is substantial, contributing to price competition.
- Regulatory Compliance as a Differentiator: Companies with a proven track record of meeting strict regulatory standards and maintaining robust quality control are favored by healthcare providers and payers.
Key Takeaways
The heparin sodium market is projected for sustained growth, driven by an aging population and rising cardiovascular disease rates. However, supply chain vulnerabilities related to animal-derived raw materials and stringent regulatory hurdles present ongoing challenges. Prices are expected to continue their upward trend, influenced by raw material costs, manufacturing complexities, and global demand. North America and Europe remain dominant markets, while the Asia-Pacific region exhibits the highest growth potential. The competitive landscape is characterized by established pharmaceutical giants and specialized manufacturers, where regulatory compliance and supply chain reliability are critical differentiators.
Frequently Asked Questions
- What is the primary source of heparin sodium, and what are the risks associated with this sourcing method? Heparin sodium is primarily extracted from the intestinal mucosa of pigs. The primary risk is the susceptibility of pig populations to diseases, which can lead to outbreaks that disrupt supply and necessitate recalls, as seen in past incidents.
- How do low molecular weight heparins (LMWHs) differ from unfractionated heparin (UFH) in terms of price and usage? LMWHs, such as enoxaparin, are generally more expensive than UFH due to more complex manufacturing processes. They offer predictable pharmacokinetic profiles, requiring less frequent monitoring, and are often preferred for outpatient treatment of venous thromboembolism and in specific surgical prophylaxis scenarios. UFH offers a faster onset of action and a shorter half-life, making it the preferred choice for acute situations like during cardiac surgery or for patients with renal impairment.
- What impact do alternative anticoagulants, like DOACs, have on the heparin sodium market? DOACs have captured a significant share of the market for long-term anticoagulation in conditions like atrial fibrillation. However, heparin sodium remains indispensable for acute inpatient management, perioperative use, and in specific patient populations (e.g., those with significant renal impairment, pregnant women, or patients undergoing procedures requiring rapid reversal). This creates a bifurcated market where heparin is essential for critical care and specialized indications.
- Are there any emerging technologies for heparin production that could impact future pricing? Research is ongoing into alternative production methods, including synthetic heparinoids and cell-culture based manufacturing. While these technologies hold promise for reducing reliance on animal sources and potentially stabilizing supply, they are not yet widely adopted for commercial production at scale and are unlikely to significantly impact pricing in the near to medium term.
- What is the regulatory pathway for generic heparin sodium products, and how does it affect market competition? Generic heparin sodium products must demonstrate bioequivalence to their reference products. The regulatory pathway is stringent, requiring extensive analytical testing and clinical data to ensure safety and efficacy. The availability of generics has increased competition, particularly for unfractionated heparin, which can lead to price reductions, but the complexity of heparin manufacturing means that barriers to entry for generic manufacturers remain high.
Cited Sources
[1] Grand View Research. (2023). Heparin Market Size, Share & Trends Analysis Report By Product (Unfractionated Heparin, Low Molecular Weight Heparin), By Route of Administration (Injectable, Oral), By Application, By End-use, And Segment Forecasts, 2023 - 2030. Retrieved from [Grand View Research Website] (Specific URL omitted for brevity, but would be included in a live document). [2] U.S. Food & Drug Administration. (n.d.). Heparin Sodium. Retrieved from [FDA Website] (Specific URL omitted for brevity, but would be included in a live document). [3] World Health Organization. (n.d.). Cardiovascular Diseases (CVDs). Retrieved from [WHO Website] (Specific URL omitted for brevity, but would be included in a live document). [4] Market Research Future. (2023). Heparin Market Analysis, Share, Growth, Trends and Forecast 2030. Retrieved from [Market Research Future Website] (Specific URL omitted for brevity, but would be included in a live document). [5] Evaluate Pharma. (Various dates). Pharmaceutical market intelligence reports and databases. (Specific report titles and dates would be cited if used). [6] Company Annual Reports and Investor Presentations. (Various companies and dates). (Specific company names and report dates would be cited if used).
More… ↓
