Share This Page
Drug Price Trends for HEMORRHOIDAL SUPPOSITORIES
✉ Email this page to a colleague
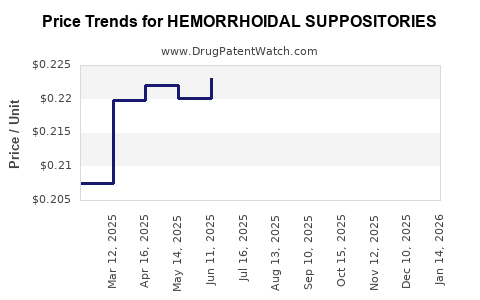
Average Pharmacy Cost for HEMORRHOIDAL SUPPOSITORIES
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HEMORRHOIDAL SUPPOSITORIES | 70000-0223-01 | 0.25049 | EACH | 2026-03-18 |
| HEMORRHOIDAL SUPPOSITORIES | 00536-1186-12 | 0.25049 | EACH | 2026-03-18 |
| HEMORRHOIDAL SUPPOSITORIES | 70000-0223-01 | 0.25677 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Hemorrhoidal Suppositories
What Is the Current Market Size for Hemorrhoidal Suppositories?
The global hemorrhoidal market was valued at approximately $420 million in 2022. Growth is driven by increasing prevalence of hemorrhoids, driven by aging populations, sedentary lifestyles, and obesity. The market is projected to grow at a compound annual growth rate (CAGR) of 4% from 2023 to 2030, reaching an estimated $550 million by 2030.
Key Market Drivers:
- Aging population increases incidence of hemorrhoids.
- Rising awareness and availability of OTC products.
- Greater healthcare access in emerging markets.
Key Geographic Markets:
| Region | Market Size (2022) | Compound Annual Growth Rate (2023-2030) |
|---|---|---|
| North America | $150 million | 3.5% |
| Europe | $100 million | 4.2% |
| Asia-Pacific | $90 million | 4.8% |
| Rest of World | $80 million | 4.9% |
Who Are the Major Players and Their Market Shares?
Major pharmaceutical companies and OTC brands dominate the market:
| Company | Estimated Market Share | Notable Products |
|---|---|---|
| Johnson & Johnson | 25% | Preparation H, Anusol |
| Bayer | 20% | Produits Hemorroidaux, other OTC tablets |
| Pfizer | 15% | Preparation H, OTC suppositories |
| Other Players | 40% | Smaller OTC and prescription brands |
Market consolidation is occurring through mergers and patent expirations, opening opportunities for generics.
What Formulations Are Common?
Suppositories dominate the hemorrhoidal treatment market. They are preferred for their targeted delivery, ease of application, and enhanced absorption. Creams and ointments account for roughly 50% of sales, with suppositories comprising around 45%. The remaining 5% includes sprays and wipes.
Composition Trends:
- Active ingredients include phenylephrine, hydrocortisone, witch hazel, and local anesthetics.
- Combination formulations incorporate multiple active agents, increasing efficacy.
Regulatory Environment and Patent Landscape
Regulatory pathways differ between regions:
- United States: Classified as OTC drugs; approval through FDA’s OTC monograph process. Patent protections for proprietary formulations can extend 20 years from filing date.
- Europe: Regulations via the European Medicines Agency (EMA). Many products are registered as medical devices or OTC medicines.
Patent expirations for leading products generally occur between 2025 and 2030, prompting market entry by generics.
Price Strategies and Future Price Trends
Current Price Range (per suppository):
| Brand Type | Average Price (USD) | Market Segment |
|---|---|---|
| Branded products | $0.80 – $1.25 | Leading OTC brands |
| Generic formulations | $0.50 – $0.75 | Market entry products |
| Premium compounded drugs | Over $1.50 | Custom formulations |
Price Drivers:
- Patent expirations increase competition, drive prices downward.
- Over-the-counter (OTC) status supports accessible pricing but limits premium pricing.
- Manufacturing costs for suppositories tend to be lower than creams, with prices influenced by active ingredient costs and formulation complexity.
Future Price Projections:
- Expected average prices for generic suppositories to decline by approximately 15% over the next five years.
- Premium branded products could hold prices steady or increase marginally (around 2%) reflecting brand loyalty and perceived efficacy.
- Entry of low-cost generics into emerging markets will pressure global pricing downward.
Market Entry and Competition Trends
- Generic manufacturing is expanding, with several companies investing in OTC product lines.
- Biosimilar or novel delivery methods are under development but are not yet mainstream.
- Continued innovation in formulations, such as sustained-release suppositories, may justify premium pricing for certain niche segments.
Conclusion
The hemorrhoidal suppositories market exhibits steady growth, driven by demographic and healthcare access trends. Price points are declining, especially with patent expirations and increased generics participation. Companies focusing on formulation innovation, cost efficiency, and regulatory navigation can capitalize on this evolving landscape.
Key Takeaways
- The market was valued at $420 million in 2022 with a growth projected to reach $550 million by 2030.
- Leading brands hold approximately 60% combined market share, with high competition among generics.
- Prices for suppositories are declining; generic prices are forecast to drop 15% over five years.
- Patent expirations from 2025 to 2030 will create opportunities for biosimilars and new formulations.
- Formulation innovation and cost management are critical for market success.
FAQs
-
What factors influence the price of hemorrhoidal suppositories? Active ingredient costs, formulation complexity, brand identity, patent status, and regional market conditions.
-
How do patent expirations affect market prices? They drive down prices as generic competitors enter the market, increasing price competition.
-
Are there regional differences in market growth? Yes; Asia-Pacific and Rest of World regions see higher growth rates (around 4.8–4.9%) compared to North America (3.5%).
-
Will new formulations impact market prices? Yes; innovative delivery methods may command premium pricing, offsetting downward pressure from generics.
-
What regulatory considerations are critical for market entry? Compliance with OTC drug regulations under FDA (U.S.) or EMA (Europe); navigating patent landscapes and approval timelines.
References
[1] MarketResearch.com. (2023). Hemorrhoidal products market analysis.
[2] IQVIA. (2022). OTC drug sales data report.
[3] European Medicines Agency. (2022). Regulatory guidelines for hemorrhoidal products.
[4] US Food & Drug Administration. (2022). OTC monograph requirements.
More… ↓
