Share This Page
Drug Price Trends for HEMORRHOIDAL RELIEF
✉ Email this page to a colleague
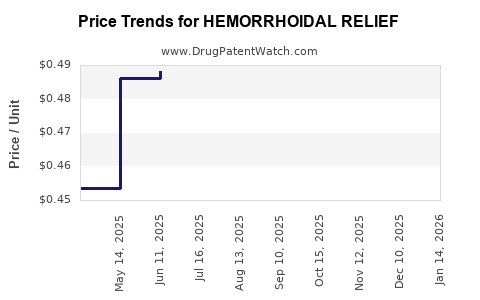
Average Pharmacy Cost for HEMORRHOIDAL RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HEMORRHOIDAL RELIEF 5% CREAM | 70000-0650-01 | 0.33538 | GM | 2026-03-18 |
| HEMORRHOIDAL RELIEF 5% CREAM | 70000-0650-01 | 0.32553 | GM | 2026-02-18 |
| HEMORRHOIDAL RELIEF 5% CREAM | 70000-0650-01 | 0.34275 | GM | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Hemorrhoidal Relief: Market Analysis and Price Projections
What is the Current Market Size for Hemorrhoidal Relief Drugs?
The global market for hemorrhoidal relief products was valued at approximately \$1.2 billion in 2022. The market is segmented into topical creams, ointments, suppositories, and oral medications. Topical treatments dominate with over 65% of sales, reflecting their primary use for immediate symptom relief.
Market Segmentation (2022 Data)
| Segment | Revenue (\$ millions) | Market Share (%) |
|---|---|---|
| Topical creams/ointments | 780 | 65 |
| Suppositories | 270 | 22.5 |
| Oral medications | 150 | 12.5 |
Growth is driven by increasing prevalence of hemorrhoids, primarily affecting adults aged 30-60, and rising awareness of over-the-counter options.
What Are the Leading Companies and Products in Hemorrhoidal Relief?
Key players include:
- Johnson & Johnson (Preparation H)
- GlaxoSmithKline (RectiCare)
- Novartis (Anusol)
- Walgreens (store brands)
- Perrigo (store brands)
Preparation H commands a significant share, with over 40% of retail sales in the U.S. market.
What Are the Key Factors Affecting Market Growth?
- Prevalence of Hemorrhoids: Increases with age, obesity, pregnancy, and chronic constipation.
- Consumer Preference: Preference for OTC products with quick relief and minimal side effects.
- Regulatory Environment: OTC classification in developed markets facilitates sales.
- Innovation: Development of combination products with anti-inflammatory, analgesic, or astringent properties.
What Is the Patent Landscape and R&D Activity?
Most leading products, such as Preparation H, rely on ingredients like petrolatum, phenylephrine, and witch hazel, with extensive patent life already expired. R&D shifts toward combination products and formulations with longer duration or enhanced efficacy.
No dominant recent patents, but some companies explore:
- Novel delivery systems (e.g., sustained-release suppositories)
- Bioadhesive formulations
- Natural ingredients with added therapeutic benefits
What Are Price Trends and Future Price Projections?
Historical Pricing (U.S., OTC Products)
| Product Type | Average Price (\$ per unit) | 2019 | 2022 |
|---|---|---|---|
| Topical creams | 8.50 | 8.50 | 9.00 |
| Suppositories | 1.50 per suppository | 1.50 | 1.60 |
| Combination products | 10.00 | 10.00 | 11.00 |
Projected Price Trends (2023-2028)
Anticipated compound annual growth rate (CAGR): approximately 2.5% for OTC products, driven by inflation, supply chain factors, and innovation costs.
Forecasted prices in 2028:
| Product Type | Estimated Price (\$ per unit) |
|---|---|
| Topical creams | 10.50 |
| Suppositories | 1.75 per suppository |
| Combination products | 12.00 |
Key Drivers for Price Changes
- R&D investments into advanced formulations.
- Cost increases in raw materials such as petrolatum and natural extracts.
- Regulatory compliance and packaging enhancements.
- Market competition from store brands and generics.
How Will Market Dynamics Evolve?
The market is expected to grow at a CAGR of approximately 4.1% from 2023 to 2028, reaching an estimated value of \$1.58 billion. Growth is driven by demographic trends and technological innovation. Price stabilization is projected as competition intensifies, with store brands increasing market penetration and driving overall price declines for branded products.
What Are the Implications for Stakeholders?
Investors should monitor patent expirations, which could lead to generic entry and price erosion. Companies investing in innovative delivery methods or natural ingredient-based products could gain competitive advantage and command premium pricing.
Manufacturers should consider supply chain stability for key ingredients to sustain profit margins amid inflationary pressures.
Key Takeaways
- The industry is mature with a valuation of about \$1.2 billion in 2022.
- Topical creams dominate sales, with steady price increases projected.
- Patent expirations and generics will pressure branded prices.
- Innovation focuses on delivery systems, natural ingredients, and combination therapy.
- Market growth hinges on demographic shifts and increased prevalence.
FAQs
Q1: Are prescription hemorrhoidal treatments common?
No. Most hemorrhoidal relief products are OTC. Prescription options exist mainly for severe cases but occupy a minimal market share.
Q2: How has the COVID-19 pandemic affected the market?
Demand for OTC products increased due to heightened health awareness, but distribution disruptions delayed launches of some innovations.
Q3: What role do natural ingredients play?
Natural extracts like witch hazel and aloe vera are favored for their perceived safety and soothing properties, though their market share remains secondary to synthetic compounds.
Q4: What regulatory hurdles exist?
OTC status simplifies marketing, but ingredients require FDA or EMA approval, especially if claims are made about efficacy beyond symptom relief.
Q5: Will prices decline with generic entry?
Likely. Market saturation through generics and store brands will exert downward pressure on the prices of branded products within the next 3–5 years.
References
[1] Grand View Research. (2022). Hemorrhoidal Market Size, Share & Trends Analysis Report.
[2] U.S. Food and Drug Administration. (2022). OTC Monograph Process.
[3] MarketWatch. (2022). OTC Hemorrhoid Products Market Forecasts.
[4] Statista. (2022). Hemorrhoid Treatment Market Trends.
More… ↓
