Share This Page
Drug Price Trends for HEAD CONGESTION-MUCUS CAPLET
✉ Email this page to a colleague
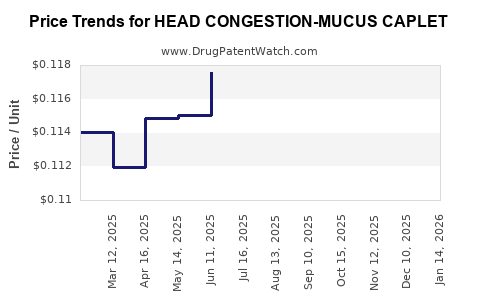
Average Pharmacy Cost for HEAD CONGESTION-MUCUS CAPLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HEAD CONGESTION-MUCUS CAPLET | 70000-0083-01 | 0.11201 | EACH | 2026-03-18 |
| HEAD CONGESTION-MUCUS CAPLET | 70000-0083-01 | 0.11211 | EACH | 2026-02-18 |
| HEAD CONGESTION-MUCUS CAPLET | 70000-0083-01 | 0.11401 | EACH | 2026-01-21 |
| HEAD CONGESTION-MUCUS CAPLET | 70000-0083-01 | 0.11319 | EACH | 2025-12-17 |
| HEAD CONGESTION-MUCUS CAPLET | 70000-0083-01 | 0.11466 | EACH | 2025-11-19 |
| HEAD CONGESTION-MUCUS CAPLET | 70000-0083-01 | 0.11419 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HEAD CONGESTION-MUCUS CAPLET Market Analysis and Financial Projection
What is the Current Market for Head Congestion-Mucus Caplet?
The market for combination cold and congestion medications, including head congestion-mucus caplets, is sizable within the broader OTC and prescription drug sectors. These products primarily target adult cold relief, with a secondary segment for pediatric use. The global cold and flu remedy market was valued at approximately $17 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 4-5% through 2028 [1].
Market share is concentrated among established brands like NyQuil, DayQuil, Mucinex, and Sudafed. These products typically contain active ingredients such as phenylephrine, pseudoephedrine, guaifenesin, or chlorpheniramine to target nasal congestion, mucus proliferation, and associated symptoms.
How is the Product Positioned in the Market?
The head congestion-mucus caplet competes primarily in the OTC segment, which accounts for over 65% of the total cold relief revenue. Prescription options are limited mostly to severe cases or comorbid conditions; thus, OTC dominance is significant. The target demographic includes adults age 18–65, with some products marketed toward seniors. Pediatric formulations are less prevalent, focusing on separate formulations with age-appropriate dosing.
Market differentiation hinges on formulations offering quick relief, fewer side effects, and combination of multiple active ingredients. Drug manufacturers are increasingly emphasizing natural ingredients or multi-symptom approaches to capture consumer attention.
What Are the Key Active Ingredients and Their Regulatory Status?
Common active ingredients:
- Pseudoephedrine: decongestant, restricted sales in some regions due to methamphetamine precursor status.
- Phenylephrine: alternative decongestant, with mixed evidence regarding efficacy.
- Guaifenesin: expectorant, widely used for mucus thinning.
- Chlorpheniramine: antihistamine, used for nasal allergy symptoms.
The regulatory landscape influences product formulation and marketing. For example, in the U.S., the Combat Methamphetamine Epidemic Act restricts pseudoephedrine sales, prompting manufacturers to pivot toward phenylephrine or develop combination products with alternative active ingredients [2].
What Are the Pricing Dynamics?
Pricing for head congestion-mucus caplets varies widely. Retail prices for a 20-count bottle range from $4 to $12, depending on brand, formulation, and geographic location. Private-label/store brands offer lower prices, generally around $4–6, while established brands can command higher premiums due to brand recognition and perceived efficacy.
Market entry costs include regulatory approval, manufacturing scale, and marketing. Brand premiums can reach 50-100% over generic equivalents. The presence of OTC giants and patent expirations influences price competition, favoring generic versions.
What Are the Price Projections for the Next 3–5 Years?
Based on current market dynamics:
| Year | Average Retail Price Range (per 20-count bottle) | Expected Trends |
|---|---|---|
| 2023 | $4–$12 | Stable, with minor increases aligned with inflation |
| 2024 | $4.25–$12.50 | Slight price hike, driven by increased raw material costs |
| 2025 | $4.50–$13 | Market saturation and generic competition curb major hikes |
| 2026 | $4.75–$13.50 | Moderate inflation influence |
| 2027 | $5–$14 | Adjustments for regulatory and supply chain costs |
No significant patent protections are currently in place, which favors price erosion via generics. Consumers may shift toward less costly alternatives, further pressuring pricing models.
What Are the Key Market and Regulatory Risks?
Regulatory shifts, such as stricter OTC drug regulations or ingredient bans, can impact formulation strategies. Supply chain disruptions, especially for active ingredients like pseudoephedrine, could restrict product availability and influence prices. Consumer health trends favoring natural or organic formulations may reduce demand for traditional caplets, pressuring traditional brands.
What Are the R&D and Product Innovation Opportunities?
Innovation focus areas include:
- Formulations with faster absorption profiles.
- Non-medicinal ingredients aimed at reducing side effects.
- Combining OTC medications with natural extracts or herbal components.
- Developing pediatric-specific formulations and flavors.
Regulatory approval remains straightforward for reformulations that do not change active ingredients but requires new safety and efficacy data for novel compositions.
Final Insights
The head congestion-mucus caplet market remains competitive, with dominant OTC brands and increasing generic penetration. Prices are likely to rise modestly, constrained by price-sensitive consumers and regulatory pressures. Product differentiation through formulation improvements offers growth avenues, but regulatory navigation remains a challenge.
Key Takeaways
- The market for head congestion-mucus caplets is driven by OTC demand, with established brands leading.
- Prices range broadly, with generics exerting downward pressure; prices are projected to increase slightly over the next five years.
- Active ingredients are influenced by regulatory restrictions, affecting formulation choices.
- Innovation opportunities focus on faster relief, natural ingredients, and pediatric formulations.
- Supply chain stability and regulatory changes represent key risks.
FAQs
1. How do regulatory restrictions impact pricing?
Restrictions on ingredients like pseudoephedrine limit supply options, often leading to shortages and potential price increases for certain formulations.
2. Are there significant patent expirations affecting this market?
Most OTC formulations rely on active ingredients whose patents have expired, leading to a proliferation of generic brands and downward price pressure.
3. What are consumer preferences influencing market dynamics?
Consumers increasingly favor products with natural ingredients, fewer side effects, and multi-symptom relief, driving innovation and reformulation.
4. How does the rise of natural or herbal alternatives affect prices?
Natural formulations often command premium pricing due to perceived safety and efficacy but may face regulatory hurdles.
5. What impact does supply chain disruption have on prices?
Supply chain issues can restrict product availability, temporarily increasing prices and prompting shifts toward alternative products or formulations.
Sources:
[1] MarketWatch, "Cold and Flu Remedy Market Size, Growth," 2022
[2] U.S. FDA, "Pseudoephedrine Regulations," 2021
More… ↓
