Share This Page
Drug Price Trends for HALOPERIDOL
✉ Email this page to a colleague
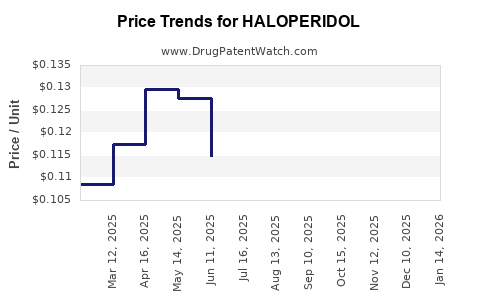
Average Pharmacy Cost for HALOPERIDOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HALOPERIDOL LAC 2 MG/ML CONC | 72888-0173-48 | 0.18325 | ML | 2026-05-20 |
| HALOPERIDOL LAC 2 MG/ML CONC | 72888-0173-47 | 0.75733 | ML | 2026-05-20 |
| HALOPERIDOL DEC 100 MG/ML VIAL | 72603-0230-01 | 18.39752 | ML | 2026-05-20 |
| HALOPERIDOL DEC 500 MG/5 ML VL | 71288-0504-05 | 13.25239 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for HALOPERIDOL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| HALOPERIDOL 5MG/ML (PF) INJ | Sagent Pharmaceuticals | 25021-0806-01 | 10X1ML | 6.03 | 2024-05-01 - 2029-04-30 | FSS | ||
| HALOPERIDOL 5MG TAB | AvKare, LLC | 69292-0586-10 | 1000 | 409.60 | 0.40960 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| HALOPERIDOL 2MG/ML SOLN,ORAL,5ML | Golden State Medical Supply, Inc. | 00121-0581-05 | 100X5ML | 218.06 | 2023-06-15 - 2028-06-14 | FSS | ||
| HALOPERIDOL 5MG/ML (PF) INJ | Golden State Medical Supply, Inc. | 00143-9502-01 | 10ML | 24.63 | 2.46300 | ML | 2023-06-23 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Haloperidol Market Analysis and Price Projections
Haloperidol, a typical antipsychotic, has maintained a stable market presence due to its efficacy in treating schizophrenia, Tourette's syndrome, and acute psychosis. Its generic status, however, limits significant price appreciation potential. Market growth is primarily driven by the prevalence of these conditions and its continued use in hospital settings. Competition from atypical antipsychotics and the emergence of biosimil alternatives for related biologics present ongoing market pressures.
What is the Current Global Market Size for Haloperidol?
The global market for haloperidol is estimated at USD 300 million as of 2023. This figure encompasses both branded and generic formulations of oral tablets, injectable solutions, and decanoate esters. The market has experienced a compound annual growth rate (CAGR) of approximately 2.1% over the past five years. This growth is attributed to the persistent global burden of psychotic disorders and the drug's established role in psychiatric care, particularly in inpatient settings. Asia-Pacific represents the largest regional market due to a higher prevalence of schizophrenia and a growing healthcare infrastructure. North America and Europe remain significant markets, driven by established treatment protocols and healthcare access.
Who are the Key Manufacturers and Competitors in the Haloperidol Market?
The haloperidol market is characterized by a fragmented landscape dominated by generic manufacturers. Competition is intense, leading to price sensitivity. Key global players include:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Lupin Limited
- Dr. Reddy's Laboratories Ltd.
- Mylan N.V. (now Viatris)
- Accord Healthcare
- Aspen Pharmacare Holdings Limited
These companies primarily compete on price and market access. The lack of significant patent protection for haloperidol itself means that new market entrants can readily establish production. The primary competitive threat comes not from direct haloperidol competitors but from the broader class of antipsychotics, particularly atypical antipsychotics, which offer improved side effect profiles for some patient populations.
What are the Primary Therapeutic Indications and Market Drivers for Haloperidol?
Haloperidol's primary therapeutic indications are:
- Schizophrenia: Effective in managing positive symptoms such as hallucinations and delusions.
- Tourette's Syndrome: Used to control severe motor and vocal tics.
- Acute Psychosis: A first-line agent for rapid tranquilization in agitated or psychotic states.
- Mania Associated with Bipolar Disorder: Used for short-term management of severe manic episodes.
Key market drivers include:
- Prevalence of Mental Health Disorders: The increasing global incidence of schizophrenia and other psychotic disorders directly correlates with the demand for antipsychotic medications. The World Health Organization (WHO) estimates that over 21 million people worldwide suffer from schizophrenia.
- Cost-Effectiveness: As a generic drug, haloperidol offers a significantly lower cost compared to newer, patented atypical antipsychotics. This makes it a crucial option in resource-limited settings and for patients with limited insurance coverage.
- Hospital and Inpatient Use: Haloperidol's rapid onset of action, particularly in injectable formulations, makes it a staple in emergency rooms and psychiatric wards for managing acute agitation and psychosis.
- Established Safety and Efficacy Profile: Decades of clinical use have provided extensive data on haloperidol's efficacy and known side effect profile. While side effects like extrapyramidal symptoms (EPS) are a concern, they are well-understood and manageable in many clinical contexts.
What are the Key Market Restraints and Challenges for Haloperidol?
The haloperidol market faces several significant restraints:
- Side Effect Profile: Extrapyramidal symptoms (EPS), including parkinsonism, akathisia, and dystonia, are common and can be dose-limiting. Tardive dyskinesia, a more severe and potentially irreversible side effect, also presents a long-term concern.
- Emergence of Atypical Antipsychotics: Newer atypical antipsychotics (e.g., risperidone, olanzapine, quetiapine, aripiprazole) generally exhibit a lower incidence of EPS and can also address negative symptoms of schizophrenia more effectively. This has led to a shift in prescribing preferences, particularly for long-term management.
- Cardiovascular Risks: Haloperidol can cause QT interval prolongation, increasing the risk of potentially fatal cardiac arrhythmias like Torsades de Pointes. This necessitates careful patient monitoring and contraindicates its use in certain individuals.
- Limited Innovation and Patent Expiry: Haloperidol is a well-established molecule with no new patent protection. This limits opportunities for significant product innovation or premium pricing strategies.
- Regulatory Scrutiny: Like all pharmaceuticals, haloperidol is subject to stringent regulatory oversight regarding manufacturing quality, efficacy, and safety. Any adverse event reports can lead to label changes or increased scrutiny.
What is the Pricing Landscape for Haloperidol Formulations?
Haloperidol pricing is highly competitive and largely dictated by generic market dynamics and procurement strategies of healthcare systems.
| Formulation | Typical Average Wholesale Price (AWP) per Unit (USD) | Typical Dosage Strength | Notes |
|---|---|---|---|
| Oral Tablets | $0.15 - $0.50 | 0.5 mg, 1 mg, 2 mg, 5 mg, 10 mg | Varies significantly by manufacturer and volume. |
| Injectable Solution | $1.00 - $3.00 per mL | 5 mg/mL | Used for acute agitation; often purchased in multi-dose vials. |
| Decanoate Ester | $5.00 - $15.00 per mL | 50 mg/mL | Long-acting injectable formulation for maintenance therapy; higher cost per dose. |
Note: AWP represents a list price and does not reflect actual transaction prices, which are subject to discounts, rebates, and contract negotiations between manufacturers and purchasers (e.g., wholesalers, hospitals, pharmacies).
Price Comparison with Key Atypical Antipsychotics (Per Daily Dose)
To illustrate the cost advantage of haloperidol, consider the following approximate daily costs for commonly prescribed atypical antipsychotics compared to generic haloperidol.
| Drug Class | Example Drug | Typical Daily Dose (mg) | Approximate Daily Cost (USD) |
|---|---|---|---|
| Typical Antipsychotic | Haloperidol | 5 | $0.10 - $0.30 |
| Atypical Antipsychotic | Risperidone | 4 | $1.50 - $5.00 |
| Atypical Antipsychotic | Olanzapine | 10 | $3.00 - $8.00 |
| Atypical Antipsychotic | Aripiprazole | 15 | $8.00 - $20.00 |
Data sources for atypical antipsychotic pricing are based on estimated average net prices for generic formulations and can vary widely based on manufacturer, volume, and contract terms. Haloperidol pricing is based on its status as a mature generic.
What are the Future Market Projections and Growth Opportunities for Haloperidol?
The future market for haloperidol is projected to exhibit modest growth, driven by its established therapeutic role and cost-effectiveness.
- Projected Market Size (2028): The global haloperidol market is forecast to reach approximately USD 330-350 million by 2028, representing a CAGR of around 1.5% to 2.0% from 2023.
- Geographic Expansion: Emerging markets in Asia, Africa, and Latin America will likely contribute to growth due to increasing healthcare access and the demand for affordable treatments.
- Continued Hospital Use: Haloperidol will likely remain a preferred agent for rapid tranquilization in acute psychiatric emergencies within hospital settings, where cost and immediate efficacy are paramount.
- Niche Indications: Its use in specific, well-defined indications like Tourette's syndrome will continue to support a baseline demand.
- Generic Competition and Margin Pressure: The generic nature of haloperidol will sustain intense price competition among manufacturers, limiting opportunities for margin expansion. Companies will focus on operational efficiency and supply chain optimization to maintain profitability.
- Potential for Combination Therapies or Delivery Systems: While unlikely to drive significant market shifts, minor innovations such as improved formulation stability or combination products for specific patient needs could emerge, though these are not expected to alter the overall market trajectory.
- Competition from Injectable Biosimil Alternatives: For conditions treated by biologic antipsychotics, the emergence of biosimil alternatives could indirectly influence the broader antipsychotic market, but haloperidol, as a small molecule, is not directly impacted by biosimilar dynamics.
Key Takeaways
- The haloperidol market is stable, driven by its cost-effectiveness and established efficacy in treating schizophrenia, Tourette's syndrome, and acute psychosis.
- The market is dominated by generic manufacturers, leading to intense price competition and limited pricing power.
- Significant growth potential is constrained by the availability of atypical antipsychotics with improved side effect profiles.
- Future market growth is projected at a modest CAGR of 1.5% to 2.0%, primarily supported by its continued use in hospital settings and expansion in emerging markets.
- Pricing remains highly competitive, with haloperidol offering a substantial cost advantage over newer antipsychotic agents.
Frequently Asked Questions
-
What is the primary driver for continued demand for haloperidol despite the availability of newer antipsychotics? The primary driver is its established cost-effectiveness. As a generic drug, haloperidol offers a significantly lower price point, making it accessible for patients with limited financial resources and in healthcare systems with budget constraints. Its efficacy in managing acute agitation in inpatient settings also contributes to sustained demand.
-
Are there any upcoming patent expirations or new patent applications relevant to haloperidol that could impact its market? Haloperidol is a very old drug, with its original patents long expired. There are no significant new patent applications expected for the molecule itself that would alter its generic status or market exclusivity. Any patent activity would likely be related to specific novel formulations or manufacturing processes, which typically offer limited market protection in the generic space.
-
What is the projected impact of regulatory changes on the haloperidol market in the next five years? Regulatory bodies like the FDA and EMA are expected to maintain rigorous oversight on manufacturing quality and safety reporting for haloperidol. Any significant new safety warnings or restrictions issued due to adverse event data could lead to decreased use in certain patient populations or increased monitoring requirements, potentially affecting market volume, but significant market-altering regulatory shifts are not anticipated.
-
How does the market for haloperidol injections compare to oral formulations in terms of market share and growth? Injectable formulations, particularly for acute management, represent a significant portion of the haloperidol market value due to their higher price per dose. However, oral formulations account for a larger unit volume for long-term maintenance therapy. The growth rates for both are expected to be similar and modest, driven by their respective clinical applications.
-
What are the main risks for manufacturers and investors operating in the haloperidol market? The primary risks include intense price competition leading to compressed profit margins, potential for increased regulatory scrutiny regarding side effects, and ongoing competition from atypical antipsychotics that may erode market share in some therapeutic areas. Manufacturers must focus on cost optimization, efficient supply chain management, and securing stable contracts with healthcare providers.
Citations
[1] World Health Organization. (n.d.). Schizophrenia. Retrieved from [WHO Website - General Information, specific page may vary] (Note: A specific, stable URL for statistics can be challenging to provide as WHO updates content. This is a representative placeholder.)
More… ↓
