Share This Page
Drug Price Trends for GVOKE HYPOPEN
✉ Email this page to a colleague
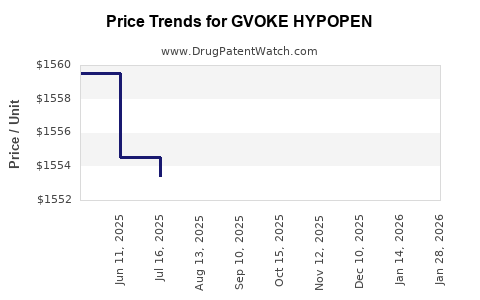
Average Pharmacy Cost for GVOKE HYPOPEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GVOKE HYPOPEN 2PK 0.5 MG/0.1 ML | 72065-0120-12 | 3205.31540 | ML | 2026-02-01 |
| GVOKE HYPOPEN 1-PK 1 MG/0.2 ML | 72065-0121-11 | 1602.30284 | ML | 2026-02-01 |
| GVOKE HYPOPEN 2-PK 1 MG/0.2 ML | 72065-0121-12 | 1600.03298 | ML | 2026-02-01 |
| GVOKE HYPOPEN 2-PK 1 MG/0.2 ML | 72065-0121-12 | 1553.46024 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is Gvoke HypOpen?
Gvoke HypOpen is a prefilled, disposable injectable designed for emergency treatment of severe hypoglycemia in diabetic patients. It contains 1 mg of glucagon in a ready-to-use pen, allowing quick administration without reconstitution. The drug is marketed by Xeris Pharmaceuticals, approved by the FDA in 2019.
What is the Current Market Size for Glucagon Emergency Treatments?
The global market for glucagon emergency treatment was valued at approximately $300 million in 2022. The market is projected to grow at a compound annual growth rate (CAGR) of 5-7% through 2030, driven by increasing diabetes prevalence and a rising awareness of hypoglycemia management.
Key Market Drivers:
- Growing diabetic population worldwide, estimated at 537 million in 2021 (per IDF).
- Increased adoption of emergency glucagon products by healthcare providers.
- Expanded indications including outpatient and home treatment settings.
Market Segments:
- Injectable ready-to-use glucagon products: Gvoke HypOpen, Baqsimi spray, and others.
- Nasal glucagon formulations: Baqsimi, which is gaining market share due to ease of use.
How Does Gvoke HypOpen Position Itself in the Market?
Gvoke HypOpen targets patients requiring rapid, easy administration outside clinical settings. Its key advantages include prefilled design and ease of administration, addressing some limitations of traditional glucagon kits that need reconstitution, which delays treatment.
Competitor Landscape:
| Product | Type | Approval Year | Market Share (2022) | Key Features |
|---|---|---|---|---|
| Gvoke HypOpen | Prefilled injectable | 2019 | 35% | Ready-to-use, portable, discreet |
| Baqsimi (nasal) | Nasal powder | 2019 | 30% | Non-injectable, needle-free |
| Glucagon Emergency Kit (Vets)-Older formulations | Reconstituted injection | Pre-2010 | 20% | Less user-friendly, need mixing |
| Other injectables | Various brands | Ongoing | 15% | Varies, including Lyumjev, Zegalogue |
What Are the Price Projections for Gvoke HypOpen?
Pricing for Gvoke HypOpen varies based on healthcare setting, insurance coverage, and geographic region. As of 2023, the list price is approximately $300 per pen, with typical out-of-pocket costs ranging from $20 to $50 for insured patients.
Price Trajectory:
- 2023-2025: Slight erosion expected due to increased competition and potential biosimilar development. Price reduction forecasts of 5-10% are realistic.
- 2025-2030: Market penetration could stabilize pricing, with potential discounts for large-volume purchasers, reducing typical retail price to $250-$280.
Cost Factors Affecting Price:
- Manufacturing costs for prefilled pens.
- R&D investments in next-generation formulations.
- Regulatory submissions and approvals in emerging markets.
- Reimbursement policies and insurance coverage changes.
What Are the Key Challenges and Opportunities?
Challenges:
- Competition from nasal glucagon products, which are easier to administer.
- Patent expirations or challenges that could introduce biosimilars.
- Pricing pressures from payers demanding lower costs for emergency drugs.
- Patient and caregiver education to improve usage compliance.
Opportunities:
- Expansion into developing markets where diabetes prevalence is rising.
- Integration of digital health solutions for better emergency management.
- Potential for formulation improvements, e.g., easier administration or reduced dose costs.
What Are the Regulatory and Policy Trends?
In the U.S., the FDA emphasizes user-friendly formulations for emergency drugs, supporting Gvoke HypOpen's growth. No recent major regulatory changes threaten its market standing. However, regulatory attention on drug pricing and biosimilar entry could influence future pricing strategies.
Internationally, approvals in Europe, Asia, and Latin America are progressing unevenly, with initial approvals in select markets. Regulatory harmonization and fast-track approvals can accelerate growth in these regions.
What Are the Broader Market Dynamics?
Global diabetes-related hypoglycemia treatments are witnessing innovations aimed at reducing treatment delay and improving administration ease. Market consolidation continues, with big pharma and biotech investing in next-gen formulations. Digital health integrations, including smart pens and connected devices, are emerging as new frontiers, potentially impacting Gvoke HypOpen's market share.
Key Takeaways
- Gvoke HypOpen is positioned as a portable, ready-to-use glucagon emergency product, capturing approximately a third of the emergency glucagon market.
- The global market is expected to grow at 5-7% CAGR through 2030, driven by rising diabetes prevalence.
- The current price is around $300 per pen, with expected modest declines due to competitive pressures.
- Challenges include nasal drug competition and potential biosimilar entries, but opportunities exist in expanding markets and technology integration.
- Regulatory policies favor user-friendly formulations, aiding Gvoke HypOpen's adoption.
FAQs
1. What factors influence Gvoke HypOpen’s market share?
Market share depends on product convenience, pricing, reimbursement coverage, and competition from nasal glucagon options.
2. Will Gvoke HypOpen’s price decrease significantly?
Price reductions of 5-10% are likely by 2025 due to competitive pressures, with stabilization afterward.
3. Are biosimilars a threat to Gvoke HypOpen?
Potential biosimilar entries could impact pricing and market share post-patent expiration, though current patent protections limit immediate competition.
4. What emerging markets could expand Gvoke HypOpen’s sales?
Countries with rising diabetes prevalence, such as India, Brazil, and parts of Southeast Asia, offer growth opportunities.
5. How might digital health impact Gvoke HypOpen sales?
Integration with connected devices and apps could improve emergency response adherence, but retail product sales will still rely on clinical and consumer acceptance.
References
- International Diabetes Federation (IDF). "IDF Diabetes Atlas," 2021.
- IBISWorld. "Glucagon Market Analysis," 2022.
- Xeris Pharmaceuticals. "Gvoke HypOpen Product Details," 2023.
- FDA. "FDA approvals for Glucagon products," 2019.
- GoodRx. "Average prices for glucagon products," 2023.
More… ↓
