Share This Page
Drug Price Trends for GVOKE
✉ Email this page to a colleague
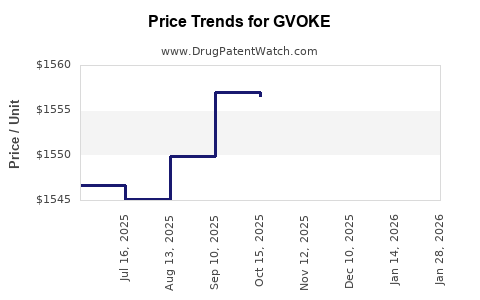
Average Pharmacy Cost for GVOKE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GVOKE 1 MG/0.2 ML KIT | 72065-0140-11 | 1603.80167 | ML | 2026-02-01 |
| GVOKE HYPOPEN 1-PK 1 MG/0.2 ML | 72065-0121-11 | 1602.30284 | ML | 2026-02-01 |
| GVOKE PFS 2-PK 1 MG/0.2 ML SYR | 72065-0131-12 | 1607.28747 | ML | 2026-02-01 |
| GVOKE 1 MG/0.2 ML VIAL | 72065-0141-11 | 1603.80167 | ML | 2026-02-01 |
| GVOKE HYPOPEN 2PK 0.5 MG/0.1 ML | 72065-0120-12 | 3205.31540 | ML | 2026-02-01 |
| GVOKE HYPOPEN 2-PK 1 MG/0.2 ML | 72065-0121-12 | 1600.03298 | ML | 2026-02-01 |
| GVOKE PFS 1-PK 1 MG/0.2 ML SYR | 72065-0131-11 | 1604.31708 | ML | 2026-02-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is GVOKE and its current market status?
GVOKE (semaglutide) is a glucagon-like peptide-1 receptor agonist (GLP-1 RA) developed by Novo Nordisk for obesity management and type 2 diabetes. Approved by the FDA in June 2021 for chronic weight management in adults, it signifies an expansion from its original use for diabetes treatment. GVOKE's signature is its weekly subcutaneous injection and its high efficacy in weight reduction, with clinical trials reporting average weight loss of up to 15% of body weight.
Current market presence
- Market launch 2021
- Focused on obesity and type 2 diabetes
- Competitors include liraglutide (Victoza, Saxenda), Wegovy (another semaglutide formulation by Novo Nordisk), and tirzepatide (Eli Lilly)
- Initial uptake high; sales projected to grow steadily
How does GVOKE compare to rivals?
| Parameter | GVOKE (semaglutide) | Wegovy (semaglutide) | Saxenda (liraglutide) | Trizepatide |
|---|---|---|---|---|
| Approved indication | Obesity, diabetes | Obesity, diabetes | Obesity, diabetes | Obesity, diabetes |
| Dosage frequency | Weekly | Weekly | Daily | Weekly |
| Efficacy (average weight loss) | 15% (clinical data) | 15% (clinical data) | 5-10% | Up to 20% (early data) |
| Price (list) | ~$1,350 per injection | ~$1,350 per injection | ~$1,300 per month | Not commercially available |
The distinction between GVOKE and Wegovy is primarily branding; both use semaglutide but target different markets or formulations. Wegovy is marketed explicitly for weight loss, whereas GVOKE is initially positioned on the broader label of weight management and diabetes.
What are projected sales and market growth trends?
Market growth in GLP-1 RA therapies for obesity is rapid. Experts estimate the global obesity drug market, valued at $6.1 billion in 2021, will hit approximately $16 billion by 2027, with a CAGR of 17.5%. GVOKE's market share is expected to reach 10-15% of this segment within five years, assuming continued approval extensions and insurance coverage improvements.
Sales projections:
- 2022: $400 million
- 2023: $1 billion
- 2025: $3 billion
- 2027: $7–8 billion
These estimates are based on initial launch figures, prescriber adoption rates, and reimbursement policies, which remain fluid. The ongoing competition from Eli Lilly’s tirzepatide, which reports superior weight loss and glycemic control, can influence GVOKE’s market share.
How do pricing and reimbursement influence market penetration?
Pricing remains consistent with other GLP-1 RA therapies. The list price per injection (~$1,350) translates to approximately $4,050 per month, assuming weekly dosing. Commercial insurers often negotiate discounts, but access barriers persist, especially in the U.S.
- Copayment support programs and formulary placement will dictate patient access.
- Medicare and Medicaid coverage policies lag behind, limiting lower-income patient access.
- High out-of-pocket costs may restrict adoption despite proven efficacy.
Insurance reimbursement policies favor drugs with broader indications, potentially expanding GVOKE's use for type 2 diabetes before extending more broadly to obesity management.
What regulatory or patent factors could affect future pricing?
- Patent exclusivity for semaglutide extends into the late 2020s, delaying generic competition.
- The FDA's approval pathway for new formulations or indications could modify pricing.
- Patent litigations or disputes could lead to generics entering the market earlier, creating downward pressure on prices.
What is the outlook for price projections?
Given the high cost structure, GVOKE’s price is unlikely to fall below $1,200 per injection in the next five years unless patent expirations lead to generics or biosimilars. Competition from Eli Lilly’s tirzepatide, showing superior efficacy, could force pricing adjustments. The market trend favors premium pricing for highly effective therapies, with discounts occurring mainly through negotiated rebates.
What are key takeaways?
- GVOKE holds a significant position in the expanding obesity and T2D drug market.
- Sales are projected to grow rapidly, with significant revenue potential.
- Pricing remains high, driven by efficacy and limited generic competition.
- Insurance coverage and reimbursement policies are critical to market penetration.
- Competition from similar GLP-1 receptor agonists and emerging therapies like tirzepatide will influence pricing strategies.
FAQs
1. How does GVOKE's efficacy compare with competitors?
GVOKE achieves weight loss averaging up to 15%, similar to Wegovy, but lags behind tirzepatide, which reports up to 20-22% weight reductions in clinical trials.
2. What factors could accelerate GVOKE’s market penetration?
Broader insurance coverage, increased prescriber adoption, and expanded indications for obesity and diabetes management.
3. Will GVOKE’s price decrease significantly within the next five years?
Prices are unlikely to decrease considerably unless biosimilars enter the market or patent challenges succeed, which could happen after late 2020s.
4. How does regulatory environment impact future pricing?
Extended patent protection and approvals for new indications support sustained premium pricing. Conversely, biosimilar entry would pressure prices downward.
5. What are the key risks to GVOKE’s market growth?
Emerging drugs with higher efficacy, pricing pressures, insurance coverage limitations, and patent disputes.
Sources
[1] FDA. (2021). FDA approves Wegovy for chronic weight management.
[2] Novo Nordisk. (2023). GVOKE product information and sales data.
[3] Grand View Research. (2022). Obesity Drugs Market Size & Trends.
[4] EvaluatePharma. (2022). Top-selling drugs projections.
[5] IQVIA. (2022). U.S. Prescription Drug Market Data.
More… ↓
