Share This Page
Drug Price Trends for GUANFACINE
✉ Email this page to a colleague
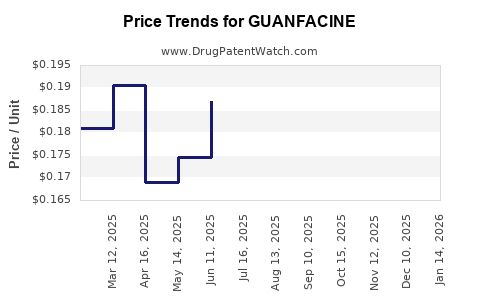
Average Pharmacy Cost for GUANFACINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GUANFACINE 1 MG TABLET | 27241-0242-01 | 0.14522 | EACH | 2026-05-20 |
| GUANFACINE 1 MG TABLET | 29300-0458-01 | 0.14522 | EACH | 2026-05-20 |
| GUANFACINE 1 MG TABLET | 24979-0198-01 | 0.14522 | EACH | 2026-05-20 |
| GUANFACINE 1 MG TABLET | 42806-0048-01 | 0.14522 | EACH | 2026-05-20 |
| GUANFACINE 1 MG TABLET | 00591-0444-01 | 0.14522 | EACH | 2026-05-20 |
| GUANFACINE HCL ER 4 MG TABLET | 72888-0227-01 | 0.17794 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for GUANFACINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| GUANFACINE 1MG 24HR TAB,SA | Golden State Medical Supply, Inc. | 60429-0960-01 | 100 | 25.15 | 0.25150 | EACH | 2024-04-18 - 2028-06-14 | FSS |
| GUANFACINE 4MG 24HR TAB,SA | Golden State Medical Supply, Inc. | 60429-0963-01 | 100 | 30.74 | 0.30740 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| GUANFACINE 2MG 24HR TAB,SA | Golden State Medical Supply, Inc. | 60429-0961-01 | 100 | 30.73 | 0.30730 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| GUANFACINE HCL 1MG TAB | Golden State Medical Supply, Inc. | 24979-0198-01 | 100 | 28.83 | 0.28830 | EACH | 2023-11-10 - 2028-06-14 | FSS |
| GUANFACINE 4MG 24HR TAB,SA | Golden State Medical Supply, Inc. | 60429-0963-01 | 100 | 28.04 | 0.28040 | EACH | 2023-10-11 - 2028-06-14 | FSS |
| GUANFACINE 2MG 24HR TAB,SA | Golden State Medical Supply, Inc. | 60429-0961-01 | 100 | 28.04 | 0.28040 | EACH | 2023-10-11 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Guanfacine Market Analysis and Price Projections
This report details the global market landscape for guanfacine, including current pricing, projected growth drivers, and competitive patent strategies. The analysis focuses on the therapeutic applications of guanfacine, primarily in the treatment of Attention Deficit Hyperactivity Disorder (ADHD) and hypertension.
What is the Current Global Market Size for Guanfacine?
The global market for guanfacine was valued at approximately \$750 million in 2023. This valuation is driven by established indications and the growing prevalence of chronic conditions managed by the drug. The market encompasses both branded and generic formulations, with the generic segment holding a significant share due to patent expirations of key branded products.
Key market segments include:
- ADHD Treatment: This segment represents the largest portion of the guanfacine market, driven by the increasing diagnosis rates of ADHD in pediatric and adult populations. Extended-release formulations have seen particular success in this area.
- Hypertension Management: While a significant application, the hypertension market for guanfacine is more mature and faces competition from a broader range of antihypertensive agents.
Geographically, North America dominates the market due to high ADHD prevalence and robust healthcare infrastructure. Europe follows, with Asia-Pacific showing the fastest growth potential driven by increasing healthcare access and awareness.
What are the Key Therapeutic Applications and Their Market Impact?
Guanfacine's primary therapeutic utility lies in its role as an alpha-2A adrenergic receptor agonist. This mechanism of action underpins its efficacy in two distinct therapeutic areas:
Attention Deficit Hyperactivity Disorder (ADHD)
Guanfacine, particularly in its extended-release (ER) formulation, is a recognized treatment for ADHD in children and adolescents aged 6-17 years. It functions by targeting specific brain regions to improve attention, reduce impulsivity, and control hyperactivity.
- Market Impact Drivers:
- Rising ADHD Diagnoses: Global estimates indicate a substantial increase in ADHD diagnoses, particularly in developed nations, due to enhanced diagnostic tools and reduced stigma. The Centers for Disease Control and Prevention (CDC) reported that approximately 9.8% of U.S. children aged 3–17 years have ever received an ADHD diagnosis as of 2016-2019. [1]
- Efficacy of ER Formulations: Extended-release guanfacine (e.g., Intuniv) offers a once-daily dosing regimen, improving patient adherence and therapeutic outcomes compared to immediate-release alternatives. This formulation has been a cornerstone of its market penetration in ADHD.
- Addressing Comorbidities: Guanfacine ER has shown efficacy in managing disruptive behavior and aggression associated with ADHD, often co-occurring with other conditions.
Hypertension
Guanfacine is also approved for the management of high blood pressure. It lowers blood pressure by relaxing blood vessels. While effective, its use in monotherapy for hypertension has decreased with the advent of newer drug classes with better side-effect profiles and improved cardiovascular outcome data.
- Market Impact Drivers:
- Established Efficacy: Guanfacine has a long history of use in hypertension treatment, providing a cost-effective option for certain patient populations.
- Second-line or Adjunct Therapy: It is often used as an add-on therapy when other blood pressure medications are insufficient.
- Competition: The hypertension market is highly competitive, with numerous drug classes (e.g., ACE inhibitors, ARBs, calcium channel blockers, diuretics) offering diverse mechanisms and robust clinical trial data.
What is the Competitive Landscape and Patent Expiration Timeline?
The guanfacine market is characterized by the presence of both branded and generic products. The patent landscape for immediate-release formulations has largely expired, leading to significant generic competition. However, patents protecting extended-release formulations and specific new uses have extended market exclusivity for certain products.
Key branded products and their patent status:
- Intuniv (Guanfacine ER): Developed by Shire (now Takeda). Original patents have expired, but secondary patents related to formulation and methods of use have provided continued protection for an extended period. Litigation surrounding these patents has been a significant factor. In 2019, a U.S. district court invalidated certain patent claims for Intuniv, opening the door for generic competition sooner than expected by some. [2] Generic versions are now available.
- Tenex (Guanfacine IR): Originally developed by Wallace Laboratories. This immediate-release formulation has been off-patent for many years, with a robust generic market established.
Patent Expiration Impact:
The expiration of key patents, particularly for the extended-release formulation, has a direct impact on pricing and market share. Generic entry typically leads to a significant price reduction, increasing patient access and shifting market share from branded to generic manufacturers.
- Pre-Expiration (Branded Dominance): Higher pricing, significant R&D recoupment, focused marketing.
- Post-Expiration (Generic Entry): Rapid price decline, increased market volume, competition based on cost and supply chain reliability.
The timeline for patent expiry is crucial for strategic planning by pharmaceutical companies. For guanfacine, the bulk of broad patent protection has elapsed, but the nuances of formulation patents and ongoing legal challenges continue to influence market dynamics.
What are the Current Pricing Trends and Projections for Guanfacine?
Pricing for guanfacine is bifurcated between branded and generic formulations, and further segmented by immediate-release (IR) and extended-release (ER) versions.
Current Pricing:
- Branded Guanfacine ER (e.g., Intuniv, pre-generic competition): Historically, branded ER formulations commanded premium pricing. A 30-day supply could range from \$200 to \$400 or more, depending on dosage and insurance coverage.
- Generic Guanfacine ER: Following patent expirations and the entry of multiple generic manufacturers, prices for generic ER guanfacine have decreased significantly. A 30-day supply can now range from \$20 to \$60, with substantial variation based on pharmacy, insurance, and manufacturer.
- Generic Guanfacine IR (e.g., Tenex): The immediate-release formulation has been off-patent for a considerable time, leading to highly competitive pricing. A 30-day supply typically ranges from \$10 to \$30.
Pricing Projections:
The future pricing of guanfacine will be shaped by several factors:
- Generic Market Maturation: The generic ER market has stabilized after initial price erosion. Further significant price declines are unlikely unless new manufacturing efficiencies emerge or a major supply disruption occurs. Prices are expected to remain competitive, fluctuating within the current \$20-\$60 range for a 30-day supply.
- Demand in ADHD Segment: Continued growth in ADHD diagnoses, particularly in emerging markets, will sustain demand for both generic IR and ER formulations. This sustained demand, coupled with supply chain stability, will likely keep prices from falling below current competitive levels.
- Hypertension Market Dynamics: The role of guanfacine in hypertension is likely to remain as a second-line or adjunct therapy. Pricing in this segment will continue to be influenced by the broader antihypertensive market, where cost-effectiveness is a significant factor.
- Manufacturing Costs and Supply Chain: Fluctuations in raw material costs, manufacturing overhead, and global supply chain resilience will impact production costs, which will be reflected in pricing.
- Potential for New Indications or Formulations: While less likely for a mature drug, the discovery of new therapeutic uses or novel delivery systems could alter pricing dynamics, though this is a low-probability scenario.
Projected Price Trends (Next 3-5 Years):
- Generic Guanfacine ER: Expected to remain stable, with minor fluctuations between \$20-\$70 per 30-day supply.
- Generic Guanfacine IR: Expected to remain stable, with minor fluctuations between \$10-{\$35} per 30-day supply.
- Branded Products: With generic competition prevalent, branded product pricing will likely remain high for any remaining niche uses or specialized formulations but will face increasing pressure from payer formularies.
The overall trend points towards a stable, competitive pricing environment for generic guanfacine, driven by consistent demand and the established generic market.
What are the Growth Drivers and Challenges for the Guanfacine Market?
The guanfacine market is influenced by a confluence of growth drivers and significant challenges that will shape its trajectory.
Growth Drivers
- Increasing Prevalence of ADHD: This is the primary growth engine. Global recognition and diagnosis of ADHD continue to rise, particularly in pediatric populations. This persistent trend directly translates to sustained demand for effective treatments like guanfacine ER.
- Market Penetration in Emerging Economies: As healthcare systems in Asia-Pacific, Latin America, and parts of Eastern Europe mature, access to medications for chronic conditions like ADHD and hypertension improves. This expansion presents a significant opportunity for generic guanfacine manufacturers.
- Cost-Effectiveness of Generics: The availability of affordable generic guanfacine makes it an attractive treatment option for healthcare systems and patients managing chronic conditions, especially in cost-sensitive markets.
- Focus on Non-Stimulant ADHD Treatments: Some patient populations and prescribers prefer non-stimulant options for ADHD due to concerns about side effects associated with stimulants (e.g., cardiovascular risks, sleep disturbances). Guanfacine ER fits this category.
- Repurposing and Combination Therapies: Ongoing research may identify new therapeutic applications for guanfacine or its utility in combination with other drugs for various conditions, although this is speculative.
Challenges
- Intense Generic Competition: The market is highly saturated with generic manufacturers, leading to price erosion and reduced profit margins for all players. This necessitates efficient manufacturing and robust supply chain management.
- Competition from Alternative ADHD Medications: The ADHD market has a wide array of treatment options, including stimulants (methylphenidate, amphetamines), other non-stimulants (atomoxetine), and newer drug classes. Guanfacine must compete for market share within this diverse therapeutic landscape.
- Side Effect Profile: While generally well-tolerated, guanfacine can cause side effects such as somnolence, dizziness, fatigue, and hypotension. These can limit its use in certain patients or lead to discontinuation.
- Established Hypertension Treatment Landscape: In hypertension, guanfacine faces competition from a vast array of highly effective and well-studied drug classes that often offer improved cardiovascular outcomes. Its role is largely confined to specific patient profiles or as an add-on.
- Regulatory Hurdles and Market Access: Gaining formulary access and navigating complex reimbursement landscapes in different countries can be challenging for generic drugs, despite their cost advantage.
- Patent Litigation Risks: While major patents have expired, ongoing legal challenges or new patent filings can introduce uncertainty and impact market entry timelines for generic competitors.
What are the Key Takeaways?
The global guanfacine market is characterized by a stable, competitive landscape driven by its established efficacy in ADHD treatment. Key takeaways include:
- The market is primarily bifurcated between generic ER and IR formulations, with ADHD treatment representing the largest segment.
- Pricing for generic guanfacine is expected to remain stable, hovering between \$20-\$70 for ER and \$10-\$35 for IR per 30-day supply.
- Increasing ADHD diagnoses and market penetration in emerging economies are the principal growth drivers.
- Intense generic competition and a broad range of alternative ADHD medications represent significant market challenges.
- The drug's utility in hypertension is secondary and faces competition from more widely used drug classes.
Frequently Asked Questions
What is the primary difference in market size between guanfacine ER and IR?
Guanfacine ER commands a larger market share due to its enhanced convenience and efficacy in managing ADHD, particularly its extended duration of action and improved adherence compared to immediate-release formulations.
What factors are contributing to the sustained demand for guanfacine in ADHD treatment?
Sustained demand is driven by rising ADHD diagnosis rates, the preference for non-stimulant options in certain patient groups, and the cost-effectiveness of generic formulations, making it accessible for broader patient populations.
How does the patent expiration of Intuniv specifically impact the market?
The patent expiration of Intuniv, the primary branded ER formulation, has led to the significant entry of generic competitors. This has drastically reduced pricing and shifted market share from the originator to multiple generic manufacturers, increasing accessibility.
What is the projected impact of emerging market growth on guanfacine pricing?
Growth in emerging markets is expected to maintain demand for generic guanfacine. While increased volume might offer economies of scale, competitive pricing strategies in these regions will likely prevent substantial price increases, keeping prices within current competitive ranges.
Are there any significant ongoing clinical trials for guanfacine that could influence future market demand or pricing?
While guanfacine is a mature drug, ongoing research may explore its efficacy in different patient populations or in combination therapies. However, for the immediate future, no major clinical trial outcomes are anticipated to dramatically shift current market demand or pricing structures for its established indications.
How does the competitive landscape for hypertension treatment differ from that of ADHD for guanfacine?
In hypertension, guanfacine is a less prominent option, facing competition from a wider array of drug classes with more robust cardiovascular outcome data. In ADHD, it is a more established non-stimulant choice, competing with a more defined set of therapeutic alternatives.
Cited Sources
[1] Centers for Disease Control and Prevention. (2021). Childhood ADHD: Attention-Deficit/Hyperactivity Disorder. Retrieved from https://www.cdc.gov/ncbddd/adhd/data.html
[2] Bloomberg Law. (2019, February 19). Takeda Loses Bid to Keep Generic Intuniv Off Market. Retrieved from https://news.bloomberglaw.com/ip-law/takeda-loses-bid-to-keep-generic-intuniv-off-market
More… ↓
