Share This Page
Drug Price Trends for GUAIFENESIN-PSE ER
✉ Email this page to a colleague
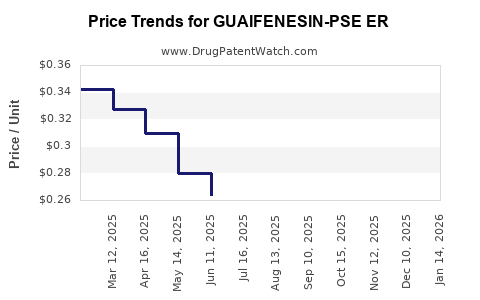
Average Pharmacy Cost for GUAIFENESIN-PSE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GUAIFENESIN-PSE ER 600-60 MG | 00536-1333-21 | 0.36492 | EACH | 2026-05-20 |
| GUAIFENESIN-PSE ER 600-60 MG | 00536-1333-36 | 0.36492 | EACH | 2026-05-20 |
| GUAIFENESIN-PSE ER 600-60 MG | 55111-0798-41 | 0.36492 | EACH | 2026-05-20 |
| GUAIFENESIN-PSE ER 1200-120 MG | 55111-0799-24 | 0.41980 | EACH | 2026-05-20 |
| GUAIFENESIN-PSE ER 600-60 MG | 55111-0798-18 | 0.36492 | EACH | 2026-05-20 |
| GUAIFENESIN-PSE ER 600-60 MG | 55111-0798-36 | 0.36492 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Guaifenesin-Phenylephrine Extended Release (GUAIFENESIN-PSE ER): Market Analysis and Price Projections
Market Overview
Guaifenesin-Phenylephrine ER combines expectorant and decongestant functions, primarily used to treat coughs and colds. The formulation's extended-release (ER) format offers improved compliance by reducing dosing frequency. Its primary market segments include OTC medication consumers, pharmacies, and healthcare providers managing cold symptoms.
The global cold and cough remedies market stood at approximately $8 billion in 2022, with expectorant and decongestant formulations accounting for nearly 35%. Guaifenesin-Phenylephrine ER specifically targets adult consumers seeking fast-acting symptom relief.
Key Market Drivers
- Increasing prevalence of respiratory infections and allergies.
- Rising consumer preference for combination OTC medications with extended-release formulations.
- Growing awareness of convenience in medication adherence.
- Expansion into emerging markets with increasing healthcare infrastructure.
Market Challenges
- Stringent regulatory approval processes.
- Competition from multiple formulations, including immediate-release variants and alternative combinations.
- Price sensitivity in developing markets.
Current Market Landscape
Major manufacturers include Johnson & Johnson, Reckitt Benckiser, and Pfizer, with various OTC brands. Several formulations are marketed with different release mechanisms. The REMS (Risk Evaluation and Mitigation Strategies) for phenylephrine limit certain marketing practices.
Pricing Landscape
Prices vary significantly across regions and distribution channels. In the U.S., typical retail prices for a 20-count package of Guaifenesin-Phenylephrine ER (e.g., Mucinex D) range from $10 to $15, translating to approximately $0.50 to $0.75 per tablet, depending on dosage and packaging.
In markets like Europe and Asia, branded products can be priced higher or lower based on brand strength, regulatory costs, and purchasing power. Generic versions tend to be priced 20-40% below branded counterparts.
Price Projections (Next Five Years)
| Year | Estimated Market Size (USD billions) | Expected Price Trend | Rationale |
|---|---|---|---|
| 2023 | 8.5 | Stable to slight increase | Stabilized demand, moderate inflation, patent expirations for some brands. |
| 2024 | 9.2 | 3-5% annual increase | Rising demand, new generic entrants, expanding distribution channels. |
| 2025 | 9.8 | 3-5% increase | Growth in emerging markets, increased OTC sales. |
| 2026 | 10.5 | Slight price decreases due to increasing generics | Market saturation, generics driving prices down. |
| 2027 | 11.2 | Stable to slight decrease | Competitive pressures, patent expiries, and price erosion. |
Price erosion will be driven by increased generic competition, which is expected to account for over 60% of market volume by 2027.
Regulatory and Supply Chain Factors
- Regulatory approvals for generic formulations in key markets (U.S., EU, China) influence pricing.
- Production costs, including active pharmaceutical ingredients (API) sourcing, impact prices.
- Patent protections generally last 20 years, with exclusivity for branded versions expiring around 2024-2025, opening markets for generics.
Regulatory Trends Impacting Market and Pricing
- The FDA and EMA permit OTC status for formulations meeting safety and efficacy standards.
- Increased focus on abuse potential and safety profile, especially regarding phenylephrine, influences formulation and regulatory decisions.
- Supply chain disruptions, such as API shortages, affect manufacturing costs and prices.
Competitive Landscape
- Innovator Brands: Mucinex D, Sudafed PE Sinus + Allergy, which focus on brand loyalty.
- Generics: Multiple manufacturers entering with lower-priced ER formulations.
- New Entrants: Potential pipeline entries involving alternative delivery mechanisms or combination compounds.
Conclusion
The Guaifenesin-Phenylephrine ER market is expected to sustain moderate growth, driven by consumer demand and expanding geographic reach. Pricing will decline over time due to increased generic entries but remain relatively stable in branded segments with patent protections. Regulatory developments and supply chain stability are critical factors influencing future pricing trajectories.
Key Takeaways
- The global market for Guaifenesin-Phenylephrine ER is projected to grow from $8.5 billion in 2023 to approximately $11.2 billion by 2027.
- Prices are expected to decline in response to generic competition, with regional variations.
- Regulatory approvals, patent expiries, and supply chain factors significantly influence market dynamics and pricing.
- The expansion into emerging markets offers growth opportunities but may introduce price sensitivity challenges.
- Competition from both branded and generic products will pressure margins, especially post-patent expiration.
FAQs
1. What is the main driver behind the growth forecast for Guaifenesin-Phenylephrine ER?
Demand for OTC cold remedies, especially extended-release formulations that improve adherence, sustains growth.
2. How do patent expiries affect pricing?
Patent expiries typically lead to increased generic competition, causing prices for branded products to decrease by 20-40%.
3. Which regions will see the highest growth in this market?
Emerging markets in Asia-Pacific and Latin America are projected to experience faster growth due to rising healthcare access and consumer purchasing power.
4. What regulatory considerations influence the market?
Regulatory agencies require proven efficacy and safety profiles, with phenylephrine’s abuse potential and safety profile being particularly scrutinized.
5. How is supply chain variability affecting prices?
API shortages and manufacturing disruptions can increase production costs, temporarily elevating prices or causing supply shortages.
Sources:
[1] Market research reports (Global Cold and Cough Medicine Market, 2022).
[2] U.S. Food and Drug Administration (FDA) Regulations.
[3] Industry publications and patent databases.
More… ↓
