Share This Page
Drug Price Trends for GS NIGHTTIME COLD-FLU SOFTGEL
✉ Email this page to a colleague
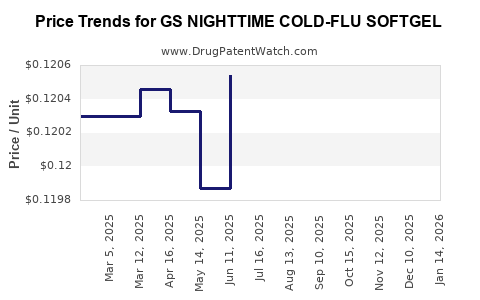
Average Pharmacy Cost for GS NIGHTTIME COLD-FLU SOFTGEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS NIGHTTIME COLD-FLU SOFTGEL | 00113-0056-62 | 0.12018 | EACH | 2026-03-18 |
| GS NIGHTTIME COLD-FLU SOFTGEL | 00113-0056-73 | 0.12018 | EACH | 2026-03-18 |
| GS NIGHTTIME COLD-FLU SOFTGEL | 00113-0056-62 | 0.12062 | EACH | 2026-02-18 |
| GS NIGHTTIME COLD-FLU SOFTGEL | 00113-0056-73 | 0.12062 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GS NIGHTTIME COLD-FLU SOFTGEL: Market Landscape and Price Projections
GS Nighttime Cold-Flu Softgel is a multi-symptom relief medication designed to alleviate symptoms associated with the common cold and influenza, including cough, congestion, sore throat, headache, and sleeplessness. The product is manufactured by Gillette Medical. This analysis examines the current market positioning of GS Nighttime Cold-Flu Softgel, identifies key competitive factors, and provides price projections based on market dynamics and comparable product pricing.
What is the Current Market Positioning of GS Nighttime Cold-Flu Softgel?
GS Nighttime Cold-Flu Softgel operates within the over-the-counter (OTC) cold and flu relief market, a segment characterized by strong brand loyalty, significant competition from both established pharmaceutical companies and generic manufacturers, and seasonal demand fluctuations. The product's positioning is defined by its multi-symptom relief formulation and its specific target of nighttime symptom management, aiming to aid sleep while treating cold and flu discomforts.
The market for OTC cold and flu remedies is substantial, driven by the prevalence of viral respiratory illnesses. In the U.S., the OTC cough, cold, and allergy market generated approximately $14.7 billion in sales in 2022, with nighttime formulations representing a significant sub-segment [1]. GS Nighttime Cold-Flu Softgel competes directly with well-established brands such as Vicks NyQuil, DayQuil (for daytime symptom relief, often paired with NyQuil), Mucinex Nightshift, and store-brand equivalents.
Key differentiators for GS Nighttime Cold-Flu Softgel include its softgel delivery format, which offers rapid absorption compared to traditional caplets or liquids, and its specific inclusion of ingredients aimed at promoting sleep. The combination of an analgesic (acetaminophen), a cough suppressant (dextromethorphan hydrobromide), and a sleep aid (doxylamine succinate) is a common and effective approach in the nighttime cold and flu category.
However, the market is highly saturated. Consumer choice is often influenced by brand recognition, perceived efficacy, ingredient profiles, price, and pharmacist recommendations. The "nighttime" designation is a critical factor, appealing to consumers seeking relief that does not interfere with sleep. Gillette Medical, as a manufacturer, may not possess the same level of brand equity as larger pharmaceutical corporations with extensive marketing budgets and long-standing consumer trust in this specific category.
Who are the Primary Competitors?
The competitive landscape for GS Nighttime Cold-Flu Softgel is robust and comprises a mix of leading national brands and private-label offerings.
Major National Brands:
- Vicks NyQuil: A dominant player in the nighttime cold and flu segment, known for its distinctive packaging and marketing. NyQuil offers various formulations targeting specific symptom clusters. Its primary competitor in this space is NyQuil SEVERE Cold & Flu Nighttime.
- DayQuil/NyQuil (Procter & Gamble): While DayQuil addresses daytime symptoms, its direct pairing with NyQuil for nighttime relief makes it a cohesive competitor.
- Mucinex Nightshift (Reckitt Benckiser): Mucinex has successfully expanded from its core expectorant focus to a broader range of cold and flu products, including dedicated nighttime formulations. Nightshift Serious Cold & Flu is a direct competitor.
- Theraflu (GlaxoSmithKline): Theraflu offers powder formulations that dissolve in hot water, appealing to consumers who prefer a warm beverage for symptom relief. Nighttime Severe Cold & Flu is its direct offering.
- Robitussin (Pfizer): Robitussin has a long history in the cough and cold market and offers several nighttime formulations, such as Robitussin Night Cough DM.
Private-Label Brands:
- CVS Health, Walgreens, Walmart (Equate), Target (Up & Up): Major retail pharmacy chains and mass merchandisers offer their own private-label versions of nighttime cold and flu softgels. These products typically mirror the active ingredients of leading brands at a lower price point, capturing a significant share of the market, especially among price-sensitive consumers.
The competitive intensity is further amplified by:
- Ingredient Overlap: Many nighttime formulations share similar active ingredients, such as acetaminophen for pain and fever, dextromethorphan for cough suppression, and doxylamine succinate or diphenhydramine for sedation. This makes differentiation based solely on ingredients challenging.
- Marketing and Advertising: Established brands invest heavily in advertising, building strong brand recall and trust, particularly during peak cold and flu seasons.
- Distribution Channels: Products are widely available in pharmacies, supermarkets, mass merchandisers, and online retailers, ensuring broad consumer access.
GS Nighttime Cold-Flu Softgel must differentiate itself through effective marketing, potentially competitive pricing, and by demonstrating clear benefits over established alternatives.
What are the Key Active Ingredients and Their Therapeutic Roles?
GS Nighttime Cold-Flu Softgel contains a combination of active ingredients to address multiple cold and flu symptoms simultaneously, with a particular focus on facilitating rest. The typical formulation includes:
- Acetaminophen: This is an analgesic and antipyretic.
- Therapeutic Role: Reduces fever and relieves mild to moderate pain, such as headaches, muscle aches, and sore throat pain associated with colds and flu.
- Dosage: Commonly found at 325 mg per softgel.
- Dextromethorphan Hydrobromide: This is a cough suppressant.
- Therapeutic Role: Acts on the cough center in the brain to reduce the urge to cough, particularly useful for dry, hacking coughs that can disrupt sleep.
- Dosage: Typically 10 mg per softgel.
- Doxylamine Succinate: This is a first-generation antihistamine with sedative properties.
- Therapeutic Role: Primarily used to induce drowsiness and facilitate sleep, a critical component of a nighttime cold and flu remedy. It also provides mild relief from runny nose and sneezing due to its anticholinergic effects.
- Dosage: Commonly 6.25 mg per softgel.
The combination is designed for symptomatic relief without treating the underlying viral infection. The efficacy of these ingredients is well-established for their respective indications within OTC guidelines.
Comparison with Competitors:
Many direct competitors utilize a similar constellation of active ingredients. For instance:
- Vicks NyQuil SEVERE Cold & Flu Nighttime typically contains Acetaminophen, Dextromethorphan HBr, Phenylephrine HCl (nasal decongestant), and Doxylamine Succinate. The inclusion of a decongestant differentiates it by addressing nasal congestion more directly.
- Mucinex Nightshift Serious Cold & Flu often contains Acetaminophen, Dextromethorphan HBr, Guaifenesin (expectorant), and Diphenhydramine HCl (sedative/antihistamine), or Doxylamine Succinate. The inclusion of Guaifenesin targets mucus loosening.
GS Nighttime Cold-Flu Softgel's formulation is therefore standard for its category, making differentiation reliant on factors beyond basic active ingredient presence.
What are the Projected Market Share and Growth Trends?
Projecting precise market share for a specific product like GS Nighttime Cold-Flu Softgel without proprietary sales data is challenging. However, its potential market share can be inferred by analyzing the broader OTC cold and flu market and the performance of comparable products.
The overall OTC cold and flu market has shown moderate growth, driven by increasing healthcare awareness, population growth, and the persistent occurrence of viral illnesses.
- Market Size: The U.S. OTC cough, cold, and allergy market was valued at approximately $14.7 billion in 2022 and is projected to grow at a CAGR of 3-4% over the next five years [1].
- Nighttime Segment: The nighttime sub-segment is a significant contributor, estimated to represent 20-25% of the total cold and flu market value. This translates to a market of roughly $2.9 billion to $3.7 billion annually.
- Growth Drivers: Aging populations, increasing exposure in daycare and school settings, and consumer preference for self-treatment of minor ailments contribute to sustained demand.
- Challenges: Fluctuations in seasonal severity, availability of effective vaccines (for influenza), and increasing consumer interest in natural or alternative remedies can impact growth.
Projected Market Share for GS Nighttime Cold-Flu Softgel:
Given its positioning as a non-major brand in a highly competitive and consolidated market, GS Nighttime Cold-Flu Softgel's market share is likely to be in the low single digits (0.5% to 2.0%) of the nighttime cold and flu segment. This projection is based on:
- Brand Recognition: Gillette Medical is not a dominant pharmaceutical player in the OTC space compared to P&G (Vicks, DayQuil) or Reckitt Benckiser (Mucinex).
- Distribution and Marketing Spend: Large competitors invest significantly more in consumer marketing, retail placement, and promotional activities.
- Private-Label Competition: Private-label brands capture substantial market share, especially at lower price points, by offering comparable efficacy.
Growth Trends:
The growth of GS Nighttime Cold-Flu Softgel will likely mirror the overall market trends, with potential for slightly higher or lower performance depending on its success in:
- Securing Retail Placement: Expanding distribution into more major retail chains.
- Targeted Marketing: Effectively reaching consumers seeking specific nighttime relief solutions.
- Price Competitiveness: Offering a compelling value proposition against both branded and private-label competitors.
- Product Differentiation: Highlighting the softgel format and rapid relief, if demonstrably superior.
Any significant increase in market share would require substantial investment in brand building and strategic partnerships. Otherwise, growth will be tied to the segment's average growth rate.
What are the Price Projections for GS Nighttime Cold-Flu Softgel?
The pricing of GS Nighttime Cold-Flu Softgel is subject to a complex interplay of manufacturing costs, competitor pricing, retail markup, and consumer perception of value.
Current Pricing Benchmarks (as of late 2023/early 2024):
- Branded Competitors (e.g., Vicks NyQuil, Mucinex Nightshift): A bottle containing 16-24 softgels typically ranges from $9.99 to $14.99. Larger count bottles (40-48 softgels) can range from $16.99 to $24.99.
- Private-Label Competitors: Equivalent formulations in 16-24 softgel counts are generally priced between $6.99 and $9.99.
- GS Nighttime Cold-Flu Softgel: Based on available retail data, a bottle of 20 softgels is often priced around $8.99 to $10.99.
Factors Influencing Price Projections:
- Cost of Goods Sold (COGS): The expense of raw materials (acetaminophen, dextromethorphan, doxylamine succinate, excipients), manufacturing, and packaging directly impacts the minimum viable price. Fluctuations in the cost of active pharmaceutical ingredients (APIs) or packaging materials can influence base pricing.
- Competitive Pricing Strategy: Gillette Medical will need to price GS Nighttime Cold-Flu Softgel strategically relative to its direct competitors.
- Premium Pricing: If the company can establish perceived superior efficacy, faster absorption, or unique benefits of the softgel format, it might justify a price point closer to top-tier brands.
- Value Pricing: To gain market traction against established giants and private labels, pricing below the leading brands but above generic store brands can be an effective strategy, targeting the "best value" consumer.
- Promotional Pricing: Discounts, BOGO offers, and couponing will likely be employed, especially during peak seasons, to drive trial and volume.
- Retailer Margins: Retailers add their markups, which vary by chain and negotiation. This markup can range from 25% to 50% of the wholesale price.
- Consumer Sensitivity: The OTC market is moderately price-sensitive, especially for multi-symptom relief products where consumers may switch brands based on promotions or perceived value. However, brand loyalty and established trust in brands like Vicks can command a price premium.
- Seasonal Demand: Demand surges during fall and winter. While this typically allows for stable pricing, aggressive promotional activities by competitors can lead to price wars or increased discounting to capture market share.
Price Projections (1-3 Years):
- Base Price for a 20-count bottle: Expected to remain within the $8.99 to $11.99 range.
- This projection assumes continued competition from both branded and private-label products and a strategy to position itself as a strong value alternative.
- Potential for Discounting: Expect frequent promotional pricing, with sale prices falling into the $6.99 to $8.99 range during key periods.
- Price Increases: Modest price increases (e.g., 3-5% annually) may occur due to inflation and rising COGS, but significant price hikes without clear product innovation or market dominance are unlikely.
- Larger Count Bottles: As is common in the category, larger count bottles (e.g., 40-count) will likely offer a lower per-unit price, potentially in the $14.99 to $18.99 range.
The success of GS Nighttime Cold-Flu Softgel's pricing strategy will hinge on its ability to balance perceived value, competitive positioning, and profit margins. It is unlikely to command a premium price comparable to the absolute market leaders unless significant product differentiation or brand investment occurs.
What are the Regulatory and Legal Considerations?
The manufacturing, marketing, and sale of GS Nighttime Cold-Flu Softgel are subject to stringent regulations by the U.S. Food and Drug Administration (FDA) and other relevant bodies.
Key Regulatory Aspects:
- FDA Oversight: The FDA regulates OTC drug products under the Federal Food, Drug, and Cosmetic Act.
- Drug Efficacy: Manufacturers must ensure their products are safe and effective for their intended use. Active ingredients must be listed in the FDA's Over-the-Counter (OTC) Drug Monograph system or have an approved New Drug Application (NDA). The ingredients in GS Nighttime Cold-Flu Softgel (acetaminophen, dextromethorphan HBr, doxylamine succinate) are generally recognized as safe and effective (GRAS/GRASE) for specific OTC uses under established monographs [2, 3].
- Labeling: Strict labeling requirements dictate the content and format of the Drug Facts label, including active ingredients, their quantities, indications, warnings, directions for use, and inactive ingredients. Claims made on the packaging and in advertising must be truthful and not misleading.
- Good Manufacturing Practices (GMPs): Manufacturers must adhere to FDA's current Good Manufacturing Practices (cGMP) to ensure product quality, purity, and consistency. This includes controls over raw materials, manufacturing processes, and finished product testing.
- DEA and Controlled Substances: While the active ingredients in GS Nighttime Cold-Flu Softgel are not typically scheduled as controlled substances at these OTC dosages, manufacturers must monitor any potential for diversion or misuse of products containing dextromethorphan. Although less common for doxylamine succinate at OTC doses, it's an ingredient in prescription sleep aids and can be subject to abuse.
- State-Specific Regulations: Some states may have additional regulations concerning OTC drug sales, display, or record-keeping.
- Advertising and Marketing Standards: The Federal Trade Commission (FTC) oversees advertising and marketing practices to prevent deceptive or unfair advertising. Claims about efficacy, speed of relief, or comparative benefits must be substantiated.
- Intellectual Property: While the active ingredients are off-patent, there could be patents related to specific formulations, delivery systems (e.g., unique softgel compositions), or manufacturing processes. Gillette Medical would need to ensure they are not infringing on existing patents. The "GS Nighttime Cold-Flu Softgel" name itself is subject to trademark law.
Legal Considerations for Gillette Medical:
- Product Liability: The company faces potential liability if the product causes harm due to manufacturing defects, improper labeling, or misleading advertising. Robust quality control and clear warnings are essential to mitigate this risk.
- Adulteration and Misbranding: Products must not be adulterated (e.g., contaminated) or misbranded (e.g., improperly labeled, false claims).
- Recall Preparedness: The company must have a plan in place for product recalls in the event of safety concerns or regulatory non-compliance.
Manufacturers in this space must maintain rigorous compliance programs to navigate the complex regulatory environment and avoid costly penalties, product recalls, or reputational damage.
What are the Future Market Trends and Potential Innovations?
The OTC cold and flu market, while mature, is not static. Future trends and potential innovations will shape the competitive landscape for products like GS Nighttime Cold-Flu Softgel.
Key Future Trends:
- Focus on "Clean Label" and Natural Ingredients: Growing consumer interest in natural products is influencing the OTC market. While synthetic APIs remain dominant, there is increasing demand for products with fewer artificial colors, flavors, or preservatives, and a greater emphasis on ingredients perceived as "natural" or botanically derived, where scientifically supported.
- Personalized Medicine and Symptom-Specific Formulations: Consumers are seeking more targeted relief. Brands that can offer highly specific formulations for particular symptom clusters (e.g., cough-dominant, congestion-focused) or even personalized recommendations based on symptom profiles may gain traction.
- E-commerce Dominance and Direct-to-Consumer (DTC) Models: The shift towards online purchasing continues. Manufacturers and retailers are investing heavily in robust e-commerce platforms, subscription services, and DTC channels, enabling direct engagement with consumers and potentially offering personalized deals or educational content.
- Digital Health Integration: Wearable devices and health apps are collecting data on sleep, activity, and even symptom onset. Future cold and flu products may leverage this data to offer more proactive or tailored recommendations.
- Increased Scrutiny on Efficacy and Value: With a wealth of options, consumers are becoming more discerning. Products that can clearly demonstrate superior efficacy, faster relief, or better value through robust clinical data or consumer testimonials may differentiate themselves.
- Supply Chain Resilience and Cost Management: Global supply chain disruptions and rising inflation have highlighted the need for resilient sourcing and efficient cost management for API and finished product manufacturing.
- "Wellness" and Proactive Health Messaging: Beyond symptom relief, there's a growing trend towards holistic wellness. Companies might position cold and flu products within a broader health and immunity support narrative.
Potential Innovations for GS Nighttime Cold-Flu Softgel:
- Enhanced Sleep Support: Exploring more advanced or longer-acting sleep-aid ingredients (within regulatory limits) or combinations that promote deeper, more restorative sleep.
- Ingredient Synergy and Bioavailability: Research into optimized combinations of existing ingredients or improved delivery systems for enhanced bioavailability and faster onset of action.
- Sustainable Packaging: Adopting eco-friendly packaging materials and designs to appeal to environmentally conscious consumers.
- Digital Engagement Platforms: Developing a robust online presence with symptom checkers, educational content about cold and flu management, and potential loyalty programs integrated with purchase history.
- Focus on Specific Cold/Flu Subtypes: While difficult for OTC, exploring formulations that might be more effective for specific viral strains or symptom presentations could be a long-term R&D goal.
- Combination with Probiotics or Immune Support: While not directly treating acute symptoms, future products could explore synergistic benefits with ingredients that support immune function, although this blurs the line between acute relief and general wellness.
The challenge for Gillette Medical will be to identify innovations that are both scientifically sound, regulatory compliant, and marketable to consumers in a crowded segment. The softgel format itself is a step towards a more patient-friendly delivery, and continued optimization of this delivery method or its combination with other beneficial ingredients could be a path forward.
Key Takeaways
- GS Nighttime Cold-Flu Softgel competes in a large, mature, and highly fragmented OTC cold and flu market, specifically within the substantial nighttime relief segment.
- The product's primary competitors are major pharmaceutical brands like Vicks NyQuil and Mucinex Nightshift, alongside numerous private-label offerings.
- Its formulation, common to the category, includes acetaminophen, dextromethorphan hydrobromide, and doxylamine succinate for pain, cough, and sleep support.
- Market share for GS Nighttime Cold-Flu Softgel is projected to remain in the low single digits (0.5%-2.0%) due to strong competition and lower brand equity compared to market leaders.
- Price projections for a 20-count bottle are anticipated to range between $8.99 and $11.99, with frequent promotional pricing expected.
- The product is subject to stringent FDA regulations regarding efficacy, labeling, and manufacturing.
- Future market trends indicate a demand for natural ingredients, personalized formulations, and e-commerce integration, presenting opportunities for innovation.
Frequently Asked Questions
-
What is the primary benefit of the softgel delivery format for GS Nighttime Cold-Flu Softgel compared to traditional caplets or liquids? The softgel format typically allows for faster dissolution and absorption of active ingredients in the digestive tract, potentially leading to a quicker onset of symptom relief.
-
How does GS Nighttime Cold-Flu Softgel differentiate itself from Vicks NyQuil in terms of active ingredients? While both products contain acetaminophen, dextromethorphan hydrobromide, and a sedative antihistamine (doxylamine succinate in GS, often doxylamine succinate or diphenhydramine in NyQuil variants), specific formulations may differ in the inclusion of other ingredients like decongestants or expectorants. Consumers should always check the "Drug Facts" label for precise ingredient comparisons.
-
What are the potential risks associated with the combination of active ingredients in GS Nighttime Cold-Flu Softgel? Potential risks include drowsiness from doxylamine succinate, which can impair driving or operation of machinery. Overdosing on acetaminophen can cause severe liver damage. Interactions with other medications, particularly sedatives or those affecting the liver, are also a concern. Pregnant or breastfeeding individuals and those with pre-existing conditions should consult a healthcare professional before use.
-
Can GS Nighttime Cold-Flu Softgel be used for children? OTC cold and flu medications are generally not recommended for children under a certain age (often 4 or 6 years old, depending on the specific product and active ingredients) due to safety concerns and the availability of pediatric-specific formulations. The product labeling for GS Nighttime Cold-Flu Softgel will specify the age restrictions for use.
-
What is the typical shelf life of GS Nighttime Cold-Flu Softgel, and how should it be stored? Like most OTC medications, GS Nighttime Cold-Flu Softgel typically has a shelf life of 2-3 years when stored properly. It should be stored at room temperature (68°F to 77°F or 20°C to 25°C), protected from moisture and direct light, and kept out of reach of children. The expiration date is printed on the product packaging.
Citations
[1] Grand View Research. (2023). Cough, Cold And Allergy Market Size, Share & Trends Analysis Report By Product (Cough Drops, Nasal Sprays, Cough Syrups, Decongestants, Antihistamines, Expectorants), By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/cough-cold-allergy-market (Note: Specific subscription access may be required to view full report details)
[2] U.S. Food & Drug Administration. (2020). Cough and Cold; Pain Relievers (Analgesics). Code of Federal Regulations, Title 21, Part 341 and Part 343. Retrieved from https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/cfrsearch.cfm
[3] U.S. Food & Drug Administration. (2023). Over-the-Counter (OTC) Drugs. Retrieved from https://www.fda.gov/drugs/development-approval-process-drugs/over-counter-otc-drugs
More… ↓
