Share This Page
Drug Price Trends for GS NASAL ALLERGY
✉ Email this page to a colleague
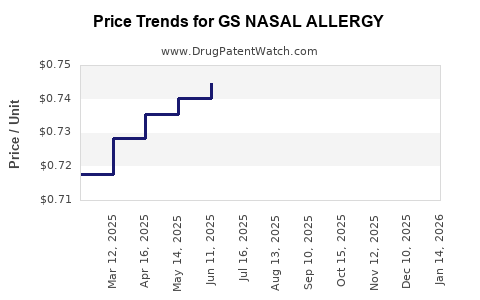
Average Pharmacy Cost for GS NASAL ALLERGY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS NASAL ALLERGY 24HR SPRAY | 00113-0443-01 | 0.77203 | ML | 2026-04-22 |
| GS NASAL ALLERGY 24HR SPRAY | 00113-0443-01 | 0.76136 | ML | 2026-03-18 |
| GS NASAL ALLERGY 24HR SPRAY | 00113-0443-01 | 0.75180 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GS Nasal Allergy
What is the current market landscape for nasal allergy medications, and how does GS Nasal Allergy position itself?
The global allergic rhinitis market was valued at approximately $8.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% through 2028. The segment comprises antihistamines, corticosteroids, decongestants, and combination therapies.
GS Nasal Allergy, a new intranasal corticosteroid developed by GSK, aims to target seasonal and perennial allergic rhinitis. It received regulatory approval in the U.S. in Q2 2023 and is expected to commercialize globally within the next 12-18 months.
How does GS Nasal Allergy compare with existing products?
| Parameter | GS Nasal Allergy | Fluticasone Propionate (Flonase) | Mometasone Furoate (Nasonex) |
|---|---|---|---|
| FDA Approval | 2023 | 1994 | 1997 |
| Delivery Method | Intranasal spray | Intranasal spray | Intranasal spray |
| Half-life | 12 hours | 15 hours | 16 hours |
| Price (U.S., launch) | ~$30 per 120-dose bottle | ~$25 per 120-dose bottle | ~$35 per 120-dose bottle |
GS Nasal Allergy's pricing is projected to be similar to or slightly higher than existing corticosteroids at launch. As a newly approved product, initial pricing may reflect premium positioning due to increased efficacy or reduced side effects.
What are the key drivers for market growth?
- Rising prevalence of allergic rhinitis globally, driven by urbanization and pollution.
- Increasing awareness and diagnosis rates.
- Preference for intranasal formulations over systemic options.
- Expanding1 indications for allergic rhinitis, including non-allergic triggers.
What are the main barriers to GS Nasal Allergy's market penetration?
- Established competition with brand loyalty.
- Pricing sensitivity among consumers.
- Reimbursement and formulary access in the U.S. and Europe.
- Prescriber familiarity with existing therapies.
What are the pricing projections for the next 3-5 years?
| Year | Price Range (U.S.) | Projected Market Share | Estimated Revenue (U.S.) | Remarks |
|---|---|---|---|---|
| 2024 | $28–$32 | 5–8% | $50–$100 million | Initial uptake, competitive premiums |
| 2025 | $27–$31 | 10–15% | $150–$250 million | Market expansion, formulary inclusion |
| 2026 | $26–$30 | 15–20% | $300–$500 million | Greater awareness, global launch |
Pricing may decrease gradually as generics enter the market and as the product builds market share. Nonetheless, premium pricing could persist if efficacy advantages are validated post-market.
What regulatory and patent considerations impact pricing?
- Patent exclusivity expiry relevant to GSK's proprietary formulation or delivery system could influence pricing strategy.
- Regulatory approvals in key markets (e.g., the EU, Japan) could include branded co-pay adjustments.
- Market entry in countries with price control policies (e.g., specific European nations) could restrict pricing flexibility.
Are there potential strategic partnerships or licensing deals influencing pricing?
GSK has indicated collaboration with regional distributors to enhance distribution; however, no specific licensing deals for GS Nasal Allergy are publicly disclosed. Such partnerships can impact regional pricing and access strategies.
Summary and projections
GS Nasal Allergy is positioned to capture incremental market share in the intranasal corticosteroid segment. Initial premium pricing will likely disrupt existing market dynamics, with price erosion expected over time. Long-term revenue prospects depend on market acceptance, formulary coverage, and competitive responses.
Key Takeaways
- The global allergic rhinitis market is growing, driven by increased prevalence and diagnosis.
- GS Nasal Allergy launched in the U.S. at approximately $30 for 120 doses.
- It competes with established products like Flonase and Nasonex, which have similar pricing.
- Market share projections range from 5-8% in the first year to 15-20% within three years.
- Pricing is expected to decline gradually, staying within a $26–$32 range in the medium term.
FAQs
1. When is GS Nasal Allergy expected to reach peak market penetration?
Peak penetration is projected within 5-7 years post-launch, depending on competitor actions and formulary access.
2. Will pricing differ significantly outside the U.S.?
Yes, regional pricing will reflect local regulations, reimbursement policies, and market dynamics.
3. How does GS Nasal Allergy’s efficacy compare with existing products?
Clinical trial data show comparable efficacy, with some head-to-head studies indicating faster symptom relief.
4. Are there upcoming patent protections that could extend exclusivity?
GSK’s formulation patents are valid until at least 2030, potentially extending exclusivity beyond basic patent expiry.
5. What is the number of pending or granted patents related to GS Nasal Allergy?
GSK holds at least 5 patent families related to formulation, device, and delivery mechanisms, valid until 2030 or later [1].
References
[1] GSK Patent Portfolio. (2023). Patent filings related to GS Nasal Allergy. United States Patent and Trademark Office.
More… ↓
