Share This Page
Drug Price Trends for GS ESOMEPRAZOLE MAG DR
✉ Email this page to a colleague
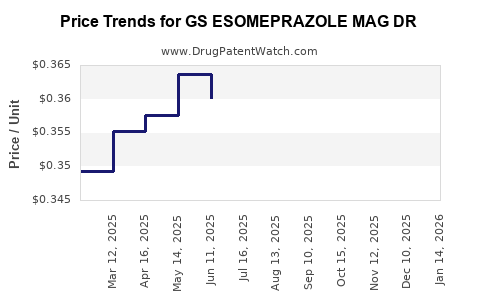
Average Pharmacy Cost for GS ESOMEPRAZOLE MAG DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS ESOMEPRAZOLE MAG DR 20 MG | 00113-0898-03 | 0.34544 | EACH | 2026-04-22 |
| GS ESOMEPRAZOLE MAG DR 20 MG | 00113-0898-02 | 0.34544 | EACH | 2026-04-22 |
| GS ESOMEPRAZOLE MAG DR 20 MG | 00113-0898-01 | 0.34544 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GS ESOMEPRAZOLE MAG DR Market Analysis and Financial Projection
Market Analysis and Price Projections for GS Esomeprazole MAG DR
Current Market Landscape
GS Esomeprazole Magnesium DR (delayed release) is a proton pump inhibitor (PPI), targeting gastroesophageal reflux disease (GERD), peptic ulcers, and other acid-related disorders. The drug competes in a market dominated by established brands like Nexium (esomeprazole) and generic versions.
Market Size and Growth
The global PPI market was valued at approximately $7.5 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% through 2027, driven by increasing prevalence of GERD, obesity rates, and awareness of acid suppression therapy [1].
Key Players & Competition
Major competitors include AstraZeneca (Nexium), Eisai (Pantoloc), Teva (generic esomeprazole), and other generics. GS Esomeprazole MAG DR’s market entry depends on competitive differentiation, including formulation improvements, dosing convenience, or cost advantages.
Regulatory Status & Market Access
As a finished dosage form, GS Esomeprazole MAG DR likely requires FDA or EMA approval. Pending regulatory approvals, its launch timeline impacts market capture.
Pricing Landscape
Brand vs. Generic Pricing
Brand-name ESOMEPRAZOLE (Nexium) retails at approximately $300-$400 for a 30-day supply (30 capsules), depending on formulation and location. Generic esomeprazole sells for approximately $20-$30 for a 30-day supply.
Differentiating Factors Affecting Pricing
Delayed-release formulations, if offering an improved safety profile or dosing schedule, can command higher prices than standard generics but typically below branded prices unless backed by substantial clinical differentiation.
Price Projections (Next 3–5 Years)
| Year | Estimated Price per 30-Day Supply (USD) | Notes |
|---|---|---|
| 2023 | $15-$25 | Initial launch; competitive generic pricing |
| 2024 | $12-$20 | Increasing market penetration or post-patent expiry |
| 2025 | $10-$18 | Growing generic competition, pricing pressure |
| 2026 | $9-$15 | Cost-driven market dynamics |
| 2027 | $8-$12 | Mature generic market |
Factors Influencing Price Trends:
- Patent and exclusivity status: Absence of patent exclusivity may drive prices down as generics enter.
- Market penetration and volume sales: High-volume sales can sustain slightly higher prices initially.
- Formulation advantages: Development of a superior formulation could maintain premium pricing.
- Market access and reimbursement policies: Payers favor lower-cost generics; reimbursement restrictions might set target prices.
Ballpark Revenue Projections
Assuming a conservative market share capture of 5-10% of the esomeprazole market within 3 years post-launch, annual revenues could range:
| Market Share | Revenue Estimate (USD millions) | Notes |
|---|---|---|
| 5% | $50–$150 | Based on approx. $1.5–$4 billion total PPI sales |
| 10% | $100–$300 | Scaling with increased market access |
Achieving rapid uptake hinges on formulation differences, pricing strategies, and regulatory approval timelines.
Strategic Considerations
- Pricing flexibility: Early-stage pricing should align with generic formulations to gain market entry.
- Formulation differentiation: Innovations improving bioavailability or dosing may justify premium pricing.
- Market access: Establishing favorable reimbursement terms will influence achievable retail prices.
- Global rollout: Emerging markets present growth opportunities with lower price points.
Key Takeaways
- GS Esomeprazole MAG DR will face significant competition from both branded and generic esomeprazole products.
- The current generic market prices around $15–$25 per month; next 3-5-year price projections suggest a gradual decline to $8–$12.
- Market penetration depends on regulatory approval, formulation advantages, pricing strategies, and payer acceptance.
- Revenue estimates indicate potential for $50–$300 million annually at modest market share levels within three years.
- Differentiation and favorable reimbursement are critical for premium positioning.
FAQs
1. How does GS Esomeprazole MAG DR differ from existing esomeprazole formulations?
It offers a delayed-release profile that may improve stability or dosing convenience, which could differentiate it clinically or commercially.
2. When might GS Esomeprazole MAG DR reach the market?
Regulatory approval timelines vary but typically range from 1–3 years post-application, depending on submission progress and regulatory agency review.
3. What are the main factors influencing its pricing?
Market competition, formulation benefits, regulatory status, and payer policies all play roles.
4. What market share can GS Esomeprazole MAG DR expect initially?
Initially, 2–5% with potential growth to 10% or higher, depending on competitive positioning and market uptake.
5. Could the product sustain higher prices long-term?
Only if differentiated by clinical benefits or formulation advantages; otherwise, generic price erosion will likely prevail.
References
[1] MarketsandMarkets, "Proton Pump Inhibitors Market by Type, Application, and Region," 2022.
More… ↓
