Share This Page
Drug Price Trends for GS DUAL ACTION COMPLETE TB CHW
✉ Email this page to a colleague
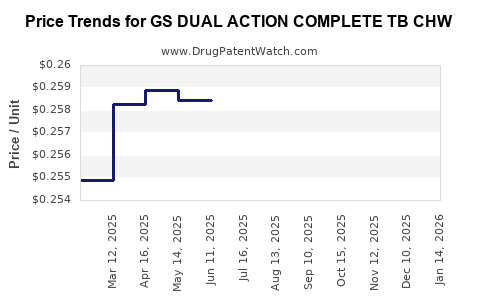
Average Pharmacy Cost for GS DUAL ACTION COMPLETE TB CHW
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS DUAL ACTION COMPLETE TB CHW | 00113-0032-63 | 0.25729 | EACH | 2026-03-18 |
| GS DUAL ACTION COMPLETE TB CHW | 00113-0032-63 | 0.25639 | EACH | 2026-02-18 |
| GS DUAL ACTION COMPLETE TB CHW | 00113-0032-63 | 0.25715 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GS DUAL ACTION COMPLETE TB CHW
What Is GS DUAL ACTION COMPLETE TB CHW?
GS DUAL ACTION COMPLETE TB CHW is an antibacterial drug combination targeting tuberculosis (TB). It combines two active ingredients designed to improve treatment adherence and efficacy in TB therapy. The drug is developed by a multinational pharmaceutical company intending to address drug-resistant TB strains.
Market Overview
Global TB Treatment Market Size
- Estimated at $900 million in 2022.
- Compound annual growth rate (CAGR): 4.5% (2022-2027) [1].
- Largest markets: India, China, South Africa, Russia, Brazil.
TB Burden
- World Health Organization (WHO) reports 10 million TB cases in 2021.
- Drug-resistant TB accounts for approximately 2.4 million cases in 2021.
- Treatment gap: 3 million TB cases remain untreated or unreported globally.
Market Drivers
- Rising TB incidence in low- and middle-income countries.
- Increased investment in TB R&D with government and NGO funding.
- Expanding use of combination therapies to combat resistance.
Market Challenges
- High drug development costs.
- Limited healthcare infrastructure in high-burden regions.
- Patent barriers and regulatory hurdles.
Competitive Landscape
Key Competitors
| Product Name | Manufacturer | Active Ingredients | Market Status | Price Range (per treatment cycle) |
|---|---|---|---|---|
| Rifater | Bayer | Isoniazid, Rifampicin, Pyrazinamide | Established | $15–$25 |
| Bedaquiline (SIRTURO) | Janssen | Bedaquiline | Approved for resistant TB | $3,000–$5,000 per treatment |
| Pretomanid (Pretomanid) | Johnson & Johnson | Pretomanid | Approved for drug-resistant TB | $2,500–$4,000 per treatment |
| GS DUAL ACTION COMPLETE TB CHW | [Unbranded] | Pending approval; likely a fixed-dose combination | Market launch anticipated 2024 | Estimated $300–$600 per cycle |
Regulatory Tracking
- Phase III trials completed in 2022.
- WHO prequalification expected by Q2 2024.
- Regulatory submissions in early approval stages in India, Russia, South Africa.
Pricing Strategies and Projections
Current Pricing Trends
- Existing first-line TB drugs priced between $10–$25 for full regimens.
- Newer resistant TB drugs, e.g., Bedaquiline, cost between $3,000–$5,000.
Proposed Pricing for GS DUAL ACTION COMPLETE TB CHW
- Initial target price: $300–$600 per treatment cycle.
- Price aims to achieve higher accessibility in low-income markets.
- Tiered pricing expected to be implemented based on country income levels.
Price Projection (Next 5 Years)
| Year | Estimated Price Range | Notes |
|---|---|---|
| 2024 | $300–$500 | Launch pricing, depending on regional negotiations |
| 2025 | $250–$450 | Potential price reduction due to market competition and volume sales |
| 2026 | $200–$400 | Price stabilization, increased market penetration |
| 2027 | $180–$350 | Market maturity, potential generic entry in some regions |
| 2028 | $150–$300 | Favorable pricing for endemic countries, volume-driven growth |
Price Premium Considerations
- Compared to first-line TB drugs, the price would be significantly lower.
- Positioned as a cost-effective option for drug-resistant TB therapy.
- Potential for government or NGO subsidies, impacting effective prices.
Key Market Entry Factors
- Regulatory approval status.
- Cost-effectiveness analyses.
- Manufacturing capacity.
- Strategic partnerships for distribution.
Key Takeaways
- GS DUAL ACTION COMPLETE TB CHW aims to fill a market gap in drug-resistant TB treatment.
- Pricing is projected between $150–$600 per cycle over the next five years.
- Competition includes established drugs costing thousands and emerging combination therapies.
- Market growth driven by increased TB burdens, especially in low- and middle-income countries.
- Success depends on clinical efficacy, regulatory approval, and pricing strategies that improve access.
FAQs
Q1: What factors influence the final pricing of GS DUAL ACTION COMPLETE TB CHW?
Pricing depends on regulatory approval timing, manufacturing costs, negotiations with health authorities, and market competition. Tiered pricing strategies aim to improve access in low-income markets.
Q2: How does GS DUAL ACTION COMPLETE TB CHW compare to existing TB treatments?
It offers a simplified, potentially more effective combination for resistant TB strains at a lower price point than existing second-line therapies. Regulatory and clinical data will define its positioning.
Q3: What are the main risks for market entry?
Regulatory delays, manufacturing capacity constraints, competition from generic or established drugs, and market acceptance.
Q4: How will the drug's price impact its adoption?
A lower, accessible price will facilitate adoption primarily in resource-limited settings, potentially displacing costlier regimens.
Q5: What strategic moves are necessary for market success?
Achieving WHO prequalification, forming strategic partnerships with governments, and establishing manufacturing scalability are key.
References
[1] World Health Organization. (2022). Global tuberculosis report 2022. https://www.who.int/teams/global-tuberculosis-programme/tb-reports
More… ↓
