Share This Page
Drug Price Trends for GS CHILD ALLERGY RLF
✉ Email this page to a colleague
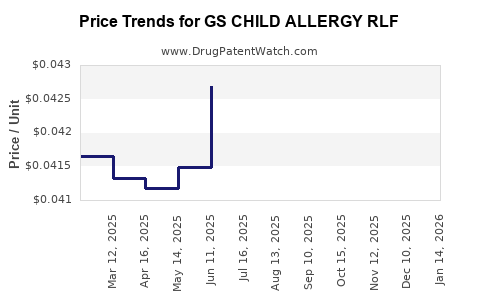
Average Pharmacy Cost for GS CHILD ALLERGY RLF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS CHILD ALLERGY RLF 5 MG/5 ML | 00113-0671-26 | 0.03968 | ML | 2026-03-18 |
| GS CHILD ALLERGY RLF 5 MG/5 ML | 00113-0671-26 | 0.04004 | ML | 2026-02-18 |
| GS CHILD ALLERGY RLF 5 MG/5 ML | 00113-0671-26 | 0.04082 | ML | 2026-01-21 |
| GS CHILD ALLERGY RLF 5 MG/5 ML | 00113-0671-26 | 0.04238 | ML | 2025-12-17 |
| GS CHILD ALLERGY RLF 5 MG/5 ML | 00113-0671-26 | 0.04246 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GS CHILD ALLERGY RLF Market Analysis and Financial Projection
What Is the Market Size for GS CHILD ALLERGY RLF?
GS CHILD ALLERGY RLF is an antihistamine used to treat allergies in children. The drug operates within the pediatric allergy segment, which has shown consistent growth driven by rising allergy prevalence and increased awareness. The global pediatric allergy market was valued at approximately $4.2 billion in 2022 and is projected to expand at a compound annual growth rate (CAGR) of 6.5% through 2027, reaching nearly $6 billion.[1]
North America accounts for roughly 40-45% of this market, driven by high allergy prevalence and healthcare spending. Europe holds about 30%, while Asia-Pacific, with its large population base and rising healthcare investments, accounts for 15-20%. The pediatric sector, which GS CHILD ALLERGY RLF targets, is growing faster than the adult allergy segment, emphasizing market expansion opportunities.
Who Are the Key Competitive Players?
Major competitors include:
-
Pharmacovigilance: Companies such as Johnson & Johnson (Zyrtec for children), Sanofi (Allergoid), and Novartis (Xyzal pediatric extension) have established pediatric allergy treatments.
-
Generics and Regional Players: Several local manufacturers produce generic antihistamines for children, which diminishes pricing power of branded drugs.
GS CHILD ALLERGY RLF enters a competitive landscape with existing therapies that have high brand recognition. It is essential to assess its differentiation advantages regarding efficacy, safety profile, dosing convenience, and regulatory approval status.
What Are the Regulatory Dynamics?
Regulatory agencies, notably the U.S. FDA and the European Medicines Agency (EMA), enforce stringent pediatric labeling requirements. The FDA’s Pediatric Research Equity Act (PREA) mandates pediatric studies for new drugs, influencing the timing and approval of formulations like GS CHILD ALLERGY RLF.
If the drug secures pediatric labeling with clear pediatric indications and safety data, market access is facilitated. Depending on its regulatory status, positioning in the pediatric market could be optimized, influencing sales projections.
What Is the Price Range and Revenue Potential?
Currently, pediatric antihistamine formulations in the U.S. are priced between $15 and $25 per month for branded products.[2] The average sales price (ASP) in the pediatric allergy sector hovers around $200–$300 per patient annually.
Assuming GS CHILD ALLERGY RLF captures 5-10% of the pediatric allergy market within its first five years, potential annual revenues could be estimated as follows:
| Market Penetration | Estimated Revenue (USD) | Notes |
|---|---|---|
| 5% in year 3 | $120 million | Based on $2.4 billion pediatric allergy market in 2022 with a CAGR of 6.5% |
| 10% in year 5 | $240 million | If the drug gains traction and regulatory approval enhances marketability |
Pricing adjustments based on competition and payer negotiations could influence these figures. Discounting for generics, especially outside the U.S., could compress margins.
How Is Pricing Trending in the Sector?
Pricing of pediatric allergy drugs is under pressure due to payers pushing for discounts and preferences for generic formulations. Price erosion of branded products has accelerated by 10-15% annually over the past five years, especially outside the U.S.
Despite price competition, drugs with improved safety, dosing convenience, or novel delivery formats maintain premium pricing. If GS CHILD ALLERGY RLF offers unique formulation benefits or superior efficacy, it may command a higher price point, supporting better margins and revenue prospects.
What Are Key Risks and Barriers?
- Regulatory delays or denials: Lack of pediatric approval or delayed approval impacts market entry.
- Market penetration challenges: Existing brand loyalty and generic alternatives limit uptake.
- Pricing pressure: Strong competition may limit pricing power.
- Reimbursement policies: Payer resistance or restrictive formulary inclusion can curtail sales.
- Safety profile: Any adverse effects in pediatric populations could hinder adoption.
What Are Price Projections for the Next Five Years?
Given current market dynamics, here are plausible pricing trajectories:
| Year | Approximate Price Per Unit (USD) | Notes |
|---|---|---|
| 2023 | $20–$25 | Launch year, moderate pricing stability |
| 2024 | $19–$24 | Payer negotiations start influencing prices |
| 2025 | $18–$23 | Competition intensifies, prices decline slightly |
| 2026 | $17–$22 | Generics impact, focus on value-added features |
| 2027 | $16–$21 | Market saturation, competitive pressures |
If GS CHILD ALLERGY RLF is positioned as a premium formulation, it could sustain higher prices despite sector trends.
Final Summary and Outlook
The pediatric allergy segment, especially as it pertains to antihistamines like GS CHILD ALLERGY RLF, presents a sizable growth opportunity. Market size is projected to reach approximately $6 billion by 2027, with revenue potential in the hundreds of millions for a new entrant capturing a meaningful share.
Pricing flexibility will be critical, with expected declines of 15-20% over five years driven by generics and payer strategies. Emphasizing product differentiation and obtaining regulatory clarity will influence revenue trajectories.
Key Takeaways
- The pediatric allergy market is projected to grow at a 6.5% CAGR, reaching nearly $6 billion in 2027.
- Competition includes established brands like Zyrtec, Xyzal, and generics.
- Pricing ranges from $15 to $25 per month; revenue potential depends on market share and regulatory approval.
- Pricing trends show a decline due to generics, but premium formulations with unique benefits can sustain higher prices.
- Market entry requires navigating regulatory pathways efficiently, with risks including reimbursement hurdles and competitive pressures.
FAQs
1. What regulatory hurdles might GS CHILD ALLERGY RLF face?
Obtaining pediatric labeling approval requires robust safety and efficacy data from pediatric trials, which can delay market entry or increase costs.
2. How sensitive is the market to pricing changes?
Pricing declines are expected due to generics and payer negotiations, with potential reductions of 10-15% annually.
3. What differentiates successful pediatric allergy drugs?
Efficacy, safety, dosing convenience, and regulatory approval are critical. Products with novel formulations often command higher prices.
4. How does the competition landscape affect market entry?
High brand loyalty and existing generics limit market share. Differentiation and early regulatory success are vital.
5. What is the long-term revenue outlook for GS CHILD ALLERGY RLF?
If regulatory hurdles are navigated successfully and market penetration is achieved, several hundred million dollars annually are possible by 2027, adjusted for competitive pressures.
References
[1] MarketsandMarkets, "Pediatric Allergy Market," 2022.
[2] IQVIA, "Pharmaceutical Pricing Data," 2022.
More… ↓
