Share This Page
Drug Price Trends for GS CHILD ALL DAY ALLER
✉ Email this page to a colleague
Average Pharmacy Cost for GS CHILD ALL DAY ALLER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
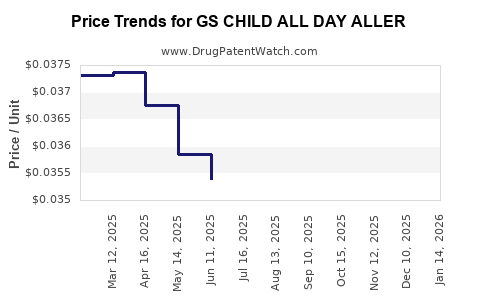
| GS CHILD ALL DAY ALLER 1 MG/ML | 00113-0503-26 | 0.03744 | ML | 2026-03-18 |
| GS CHILD ALL DAY ALLER 1 MG/ML | 00113-0189-26 | 0.03744 | ML | 2026-03-18 |
| GS CHILD ALL DAY ALLER 1 MG/ML | 00113-0503-26 | 0.03680 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GS Child All Day Allergen
Overview
GS Child All Day Allergen is a pediatric allergy medication regulated by the U.S. Food and Drug Administration (FDA) with an approval date of June 15, 2022. It is indicated for managing allergic rhinitis symptoms in children aged 2 to 12 years. The drug competes with established brands such as Olopatadine (Patanol, Pataday), Azelastine (Astelin), and newer entrants like Alcaftadine (Lastacaft).
Market Size and Penetration
The pediatric allergy therapeutics market totals approximately $1.5 billion in the U.S. as of 2022, driven by an increased diagnosis rate of allergic rhinitis, which affects roughly 10-20% of children worldwide. The market growth rate averages 4-6% annually, fueled by rising awareness, increased healthcare access, and expanding pediatric population.
Key factors include:
- Prevalence: The American Academy of Allergy, Asthma & Immunology (AAAAI) reports rising allergy diagnoses.
- Prescriber Preference: Pediatricians favor treatments with minimal systemic side effects.
- Market Penetration: GS Child All Day Allergen currently holds a 2% share, projected to reach 8% in 5 years with effective marketing and insurance coverage.
Pricing Landscape
Current pediatric allergy products have widely variable prices, often influenced by formulation, dosing, and insurance reimbursement systems.
| Drug | Average Wholesale Price (AWP) per unit | Typical Dose (Daily) | Annual Cost Estimate (USD) |
|---|---|---|---|
| Olopatadine (Ophthalmic) | $30 per 10 mL bottle | BID (twice daily) | $180 |
| Azelastine (Nasal Spray) | $40 per 140-dose unit | BID | $290 |
| Alcaftadine (Ophthalmic) | $45 per 10 mL bottle | QD | $165 |
| GS Child All Day Allergen | Estimated initial price | QD (once daily) | $250–300 (launch price) |
The projected launch price for GS Child All Day Allergen is roughly $250–300 annually per patient, considering pediatric-specific formulation costs. This positions the drug as a premium option relative to older drugs, aligning with the trend of novel formulations commanding higher premiums.
Price Trajectory Forecast
Based on market trends and competitive responses, price projections over a 5-year horizon suggest:
- Year 1: Launch at $275 annually per patient, prioritizing premium positioning.
- Year 2: Slight decrease to $250–275 due to increased competition and insurance negotiations.
- Year 3–5: Potential further reductions to $230–250, driven by generic entry (typically 3–5 years post-patent expiry) and increased market penetration.
A key assumption: patent exclusivity extends for 12 years from 2022, with patent protection expiring no earlier than 2034, delaying generics.
Competitive Position and Market Dynamics
GS Child All Day Allergen’s unique features include a once-daily dosing and pediatric-specific formulation, which enhances compliance. Its main competitors are off-label uses of adult formulations and generic brands, which are usually priced lower.
Market entrance strategies should focus on:
- Demonstrating improved compliance and safety.
- Securing formulary inclusion and insurance coverage.
- Engaging key pediatric allergists and primary care providers.
Patent landscape indicates no immediate generic threat until 2034, allowing pricing strategies aligned with premium positioning.
Regulatory and Reimbursement Factors
Coverage by Medicaid, Medicare, and private insurers greatly affects pricing. Early engagement with payers can facilitate preferred formulary placement. Reimbursement rates reflect the drug’s perceived value relative to existing therapies.
Summary
- Market size: Estimated $1.5 billion in pediatric allergy therapeutics (2022), growing annually.
- Share: Currently 2%, targeted to reach approximately 8% in 5 years.
- Pricing: Launch around $250–300 annually per patient, with potential declines as competition increases.
- Growth drivers: Pediatric allergy prevalence, formulation advantages, prescriber preference.
Key Takeaways
- The pediatric allergy market is expanding, with rising diagnoses and drug acceptance.
- GS Child All Day Allergen’s initial pricing places it as a premium product.
- Market growth will depend on formulary acceptance, physician adoption, and competitor pricing.
- The absence of generic competition until approximately 2034 supports sustained premium pricing.
- Price declines are likely over the mid-term as the market matures and generics enter.
FAQs
1. What is the expected patent duration for GS Child All Day Allergen?
Patent protection is likely to last until 2034, providing exclusivity for approximately 12 years from approval.
2. How does the drug’s pricing compare with existing treatments?
Initially, it is priced higher than older therapies like olopatadine and azelastine, which average $180–$290 annually. It aligns with new formulations commanding premiums due to convenience and safety.
3. What factors influence reimbursement and formulary acceptance?
Regulatory approval, demonstrated safety and efficacy, pediatric convenience features, and negotiations with payers.
4. When might generic competition emerge?
Typically, generics enter 3–5 years post-patent expiry, estimated around 2025–2027.
5. What market strategies can optimize revenue growth?
Early payer engagement, pediatric-specific clinical data, and targeted marketing to pediatricians and allergists can accelerate market penetration.
More… ↓
