Share This Page
Drug Price Trends for GS ANTI-DIARRHEAL-ANTIGAS CPLT
✉ Email this page to a colleague
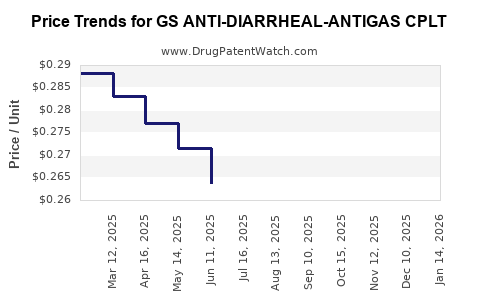
Average Pharmacy Cost for GS ANTI-DIARRHEAL-ANTIGAS CPLT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS ANTI-DIARRHEAL-ANTIGAS CPLT | 00113-0087-53 | 0.29193 | EACH | 2026-04-22 |
| GS ANTI-DIARRHEAL-ANTIGAS CPLT | 00113-0087-62 | 0.29193 | EACH | 2026-04-22 |
| GS ANTI-DIARRHEAL-ANTIGAS CPLT | 00113-0087-53 | 0.29732 | EACH | 2026-03-18 |
| GS ANTI-DIARRHEAL-ANTIGAS CPLT | 00113-0087-62 | 0.29732 | EACH | 2026-03-18 |
| GS ANTI-DIARRHEAL-ANTIGAS CPLT | 00113-0087-53 | 0.30285 | EACH | 2026-02-18 |
| GS ANTI-DIARRHEAL-ANTIGAS CPLT | 00113-0087-62 | 0.30285 | EACH | 2026-02-18 |
| GS ANTI-DIARRHEAL-ANTIGAS CPLT | 00113-0087-53 | 0.30689 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GS Anti-Diarrheal-Antigas Cplt
Executive Summary
GS Anti-Diarrheal-Antigas Cplt, a combination drug targeting diarrhea and gas, faces a competitive market landscape. The primary driver for demand is the prevalence of gastrointestinal (GI) disorders, which exhibit seasonal fluctuations. Key market participants include established over-the-counter (OTC) brands and emerging generics. Pricing for similar combination products ranges from $8 to $18 for standard packaging, with brand premium and formulation offering potential for higher price points. Projected market growth is moderate, influenced by an aging population, increasing awareness of digestive health, and the potential for new indications. Patent exclusivity and regulatory approvals are critical determinants of future market share and pricing power.
What is the Current Market Landscape for Anti-Diarrheal and Antigas Medications?
The global market for anti-diarrheal and antigas medications is substantial, driven by the high incidence of gastrointestinal disorders. These conditions, including acute diarrhea, irritable bowel syndrome (IBS), and functional dyspepsia, affect a significant portion of the population annually.
Key Market Segments:
- Anti-Diarrheals: This segment includes medications that reduce stool frequency and liquidity. Active pharmaceutical ingredients (APIs) commonly found in this category are loperamide, bismuth subsalicylate, and diphenoxylate.
- Antigas Medications: These products target the relief of bloating, abdominal discomfort, and flatulence. Simethicone is the predominant API in this segment, working by breaking down gas bubbles in the digestive tract.
- Combination Products: Drugs like GS Anti-Diarrheal-Antigas Cplt combine APIs from both segments, offering a dual-action solution. These products aim to address multiple symptoms simultaneously.
Market Size and Growth Drivers:
The global anti-diarrheal and antigas market was valued at approximately $4.5 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 3% to 5% over the next five years, reaching an estimated $5.5 billion by 2028 [1].
- Prevalence of GI Disorders: Chronic and acute GI issues remain a primary driver. The World Gastroenterology Organisation estimates that functional GI disorders affect between 20% and 40% of the population in Western countries [2].
- Aging Population: Older adults often experience a higher incidence of digestive issues, contributing to sustained demand.
- Dietary Habits and Lifestyle: Changes in diet, increased stress levels, and sedentary lifestyles can exacerbate GI symptoms, fueling market growth.
- Over-the-Counter (OTC) Accessibility: The availability of many anti-diarrheal and antigas medications without a prescription simplifies patient access and drives volume.
- Growing Health Awareness: Increased consumer focus on digestive health and wellness prompts greater utilization of these products.
Competitive Landscape:
The market is characterized by the presence of both multinational pharmaceutical companies and smaller generic manufacturers.
- Major Players: Companies like Johnson & Johnson (Imodium), Bayer (Pepto-Bismol), and Procter & Gamble (Gas-X) hold significant market share in their respective categories and often offer combination products.
- Generic Competition: A robust generic segment exists, particularly for widely used APIs like loperamide and simethicone. This intensifies price competition.
- New Product Development: Ongoing research into novel formulations and combination therapies aims to differentiate products and capture market share.
The regulatory environment, particularly regarding OTC approvals and manufacturing standards, plays a crucial role in market entry and product positioning.
What are the Key Ingredients and Mechanism of Action for GS Anti-Diarrheal-Antigas Cplt?
GS Anti-Diarrheal-Antigas Cplt is a multi-ingredient formulation designed to alleviate symptoms associated with both diarrhea and excess intestinal gas. Understanding its active components and how they function is essential for assessing its market potential and competitive positioning.
Primary Active Pharmaceutical Ingredients (APIs):
While specific API details for "GS Anti-Diarrheal-Antigas Cplt" would be proprietary, a typical combination product in this category would likely contain:
- Anti-Diarrheal Agent:
- Likely API: Loperamide hydrochloride.
- Mechanism of Action: Loperamide is a peripheral opioid receptor agonist that acts on the μ-opioid receptors in the myenteric plexus of the large intestine. It slows intestinal motility by decreasing the tone and activity of the longitudinal and circular smooth muscles of the intestine. This increased transit time allows for more absorption of water and electrolytes from the intestinal lumen, resulting in firmer stools and reduced fecal volume [3]. It does not cross the blood-brain barrier significantly at therapeutic doses, minimizing central nervous system effects.
- Antigas Agent:
- Likely API: Simethicone.
- Mechanism of Action: Simethicone is an inert surfactant. It works by reducing the surface tension of gas bubbles in the stomach and intestines, causing smaller bubbles to coalesce into larger ones. These larger bubbles are more easily eliminated from the body through belching or flatus, thus relieving bloating and discomfort [4]. Simethicone is pharmacologically inactive and is not absorbed into the bloodstream; it passes through the digestive system unchanged.
Potential Synergistic Effects:
The combination of a motility inhibitor like loperamide with an antiflatulent like simethicone offers a dual approach to managing common digestive complaints. Diarrhea can be associated with increased intestinal motility, and conversely, gastrointestinal dysmotility can sometimes lead to gas buildup. By addressing both aspects, GS Anti-Diarrheal-Antigas Cplt aims to provide comprehensive symptom relief.
Dosage and Administration:
Standard adult dosages for similar OTC combination products typically involve taking 1-2 tablets or capsules every 4 to 8 hours as needed, with a maximum daily limit to prevent adverse effects. Precise dosage and frequency would be dictated by the specific API concentrations in GS Anti-Diarrheal-Antigas Cplt and would be outlined in the product's labeling.
Formulation Considerations:
The formulation can influence patient compliance and product efficacy. Common forms include tablets, capsules, and chewables. Extended-release or fast-dissolving formulations could represent a competitive advantage.
What is the Intellectual Property Landscape for GS Anti-Diarrheal-Antigas Cplt?
The intellectual property (IP) surrounding pharmaceutical products, particularly combination therapies, is a critical determinant of market exclusivity, pricing power, and long-term profitability. For GS Anti-Diarrheal-Antigas Cplt, the IP landscape would encompass patents related to the compound, formulation, manufacturing process, and potential new uses.
Patent Types and Their Significance:
- Composition of Matter Patents: These are the strongest form of patent protection, covering the novel chemical entities themselves. If GS Anti-Diarrheal-Antigas Cplt is a truly novel combination or a new salt/ester of existing drugs with unexpected properties, it could be eligible for such patents.
- Current Status: For established APIs like loperamide and simethicone, composition of matter patents have long expired. However, novel combinations or specific ratios of these APIs that demonstrate unexpected synergistic benefits could be patentable.
- Formulation Patents: These patents protect the specific ways in which the APIs are combined with excipients to create the final dosage form (e.g., tablets, capsules, extended-release formulations, coatings).
- Significance: A unique formulation that improves bioavailability, stability, palatability, or patient convenience can secure market exclusivity for an extended period, even if the APIs themselves are off-patent. This is a common strategy for branded OTC drugs.
- Example: Patents for extended-release versions of loperamide or chewable simethicone formulations are prevalent.
- Method of Treatment Patents (Use Patents): These patents cover new uses for existing drugs or drug combinations for treating specific medical conditions.
- Significance: If GS Anti-Diarrheal-Antigas Cplt demonstrates efficacy in treating a specific subtype of diarrhea or a particular manifestation of gas and bloating that is not currently addressed by standard treatments, a method of treatment patent could be obtained.
- Process Patents: These patents protect novel and non-obvious methods of manufacturing the drug product or its intermediates.
- Significance: While often less impactful than composition or formulation patents, a cost-effective or more efficient manufacturing process can provide a competitive advantage and can be defended.
Patent Exclusivity Periods:
- Standard Patent Term: In most jurisdictions, patents are granted for a term of 20 years from the filing date.
- Data Exclusivity: Regulatory bodies often grant periods of data exclusivity (e.g., 5 years for new chemical entities in the US, 8 years in Europe, with extensions possible for pediatric studies) following market approval. This prevents generic manufacturers from relying on the innovator's clinical trial data to gain approval.
- Patent Term Extension (PTE): In some regions (like the U.S.), patent terms can be extended to compensate for delays in regulatory review, particularly for new drug applications.
Freedom to Operate (FTO) Analysis:
Before launching GS Anti-Diarrheal-Antigas Cplt, a comprehensive Freedom to Operate (FTO) analysis is essential. This involves identifying all relevant patents held by competitors that might be infringed by the manufacturing, marketing, or sale of the product. A positive FTO opinion is crucial to avoid costly litigation.
Potential Patent Challenges and Generic Entry:
If GS Anti-Diarrheal-Antigas Cplt relies on existing, off-patent APIs, its market exclusivity will primarily stem from proprietary formulations or manufacturing processes. Competitors, particularly generic manufacturers, will likely seek to develop bioequivalent versions upon the expiry of any relevant formulation or process patents. Patent litigation is common in the pharmaceutical industry, and successful challenges to a patent can lead to earlier generic entry.
Current Patent Landscape Assessment (Hypothetical):
Assuming GS Anti-Diarrheal-Antigas Cplt utilizes established APIs (loperamide and simethicone) and a standard formulation, its primary IP protection would likely reside in any novel excipient combinations, release mechanisms, or manufacturing methods that are distinct from existing products. If these aspects are not patented or if existing patents expire, generic competition would be a significant factor post-launch. The strength of its patent portfolio will dictate the duration of its market exclusivity and its ability to command premium pricing.
What are the Regulatory Considerations for GS Anti-Diarrheal-Antigas Cplt?
The regulatory pathway for GS Anti-Diarrheal-Antigas Cplt is a critical determinant of its market entry timeline, approved indications, and ultimately, its commercial success. The classification of the drug (prescription vs. over-the-counter) significantly impacts marketing strategies and accessibility.
Regulatory Classification:
- Over-the-Counter (OTC) Status: Given that both loperamide and simethicone are widely available as OTC medications in many markets, it is highly probable that GS Anti-Diarrheal-Antigas Cplt would also be classified as an OTC product. This classification is based on the safety profile of the APIs at recommended dosages, the intended use by self-treating individuals, and the availability of adequate labeling for safe and effective use without physician supervision.
- In the United States: Drugs containing loperamide (e.g., Imodium A-D) and simethicone (e.g., Gas-X) are generally available OTC [5, 6].
- In Europe: Similar OTC availability exists for these APIs in many member states, though specific regulations can vary.
Key Regulatory Agencies and Requirements:
- U.S. Food and Drug Administration (FDA):
- Abbreviated New Drug Application (ANDA): If GS Anti-Diarrheal-Antigas Cplt is a generic version of an existing approved drug product, it would follow the ANDA pathway. This requires demonstrating bioequivalence to the reference listed drug (RLD) and proving that the inactive ingredients do not raise new safety concerns.
- Over-the-Counter Monograph System: For many OTC drug categories, the FDA uses a monograph system. If the proposed combination falls within an established monograph, the approval process is streamlined. If it represents a novel OTC combination not covered by existing monographs, a New Drug Application (NDA) or a specific OTC drug application pathway may be required, potentially involving more extensive data.
- Labeling Requirements: Strict adherence to FDA labeling requirements for OTC drugs is mandatory, including clear indications, dosage instructions, warnings, precautions, and potential side effects.
- European Medicines Agency (EMA) and National Competent Authorities (NCAs):
- Marketing Authorisation Application (MAA): For a new medicinal product, an MAA is submitted. For OTC products, a simplified procedure might be available depending on the specific country and the novelty of the combination.
- European Pharmacopoeia (Ph. Eur.): The APIs and excipients must comply with the standards set forth in the Ph. Eur.
- Product Information: Similar to the FDA, comprehensive and clear patient information leaflets and SmPCs (Summary of Product Characteristics) are required.
Common Challenges in the Regulatory Process:
- Demonstrating Bioequivalence: For generic products, proving bioequivalence to the RLD is paramount. This involves pharmacokinetic studies comparing the rate and extent of drug absorption.
- Safety and Tolerability Data: Even for OTC products, sufficient data must be available to support the safety of the combination, especially regarding potential interactions between APIs or excipients.
- Labeling Comprehensiveness: Ensuring that labeling is clear, accurate, and provides sufficient information for safe self-administration by consumers is a rigorous process.
- Good Manufacturing Practices (GMP): Manufacturing facilities must comply with stringent GMP standards to ensure product quality and consistency.
Timeline to Market Approval:
The timeline for regulatory approval can vary significantly:
- ANDA (Generic): Typically 1-3 years, depending on FDA review times and any potential patent challenges.
- NDA (New OTC Combination): Could range from 2-5 years or longer, requiring more comprehensive clinical and non-clinical data if it does not fit neatly into existing monographs.
Successful navigation of the regulatory landscape requires deep expertise in pharmaceutical law, extensive documentation, and rigorous scientific validation.
What are the Pricing Strategies and Projections for GS Anti-Diarrheal-Antigas Cplt?
Pricing for GS Anti-Diarrheal-Antigas Cplt will be influenced by a complex interplay of market positioning, competitive pressures, product differentiation, and cost of goods. Given the likely OTC status and the availability of generic alternatives for its constituent APIs, the pricing strategy will need to balance perceived value with competitive realities.
Factors Influencing Pricing:
- Competitive Benchmarking:
- Direct Competitors: Products that are already combination anti-diarrheal and antigas medications.
- Indirect Competitors: Individual loperamide and simethicone products, as well as other anti-diarrheal or antigas agents.
- Pricing Range for Similar OTC Combination Products: Based on available market data, comparable OTC combination products (e.g., those containing bismuth subsalicylate and simethicone, or loperamide and simethicone in different formulations) typically range from $8 to $18 for a standard package (e.g., 12-24 count). The price can vary based on brand recognition, formulation (e.g., extended-release, chewable), and retailer markup.
- Product Differentiation and Value Proposition:
- Novel Formulation: If GS Anti-Diarrheal-Antigas Cplt offers a unique formulation (e.g., faster onset of action, longer duration, improved palatability, easier-to-swallow dosage form), it can command a premium price.
- Brand Equity: Establishing a strong brand reputation for efficacy and reliability can justify higher pricing compared to unbranded or lesser-known generics.
- Synergistic Benefits: If clinical data demonstrates superior efficacy or a broader spectrum of symptom relief due to the specific combination or ratio of APIs, this value can be translated into pricing.
- Cost of Goods Sold (COGS):
- API Costs: Loperamide and simethicone are widely produced APIs, and their costs are relatively stable. However, sourcing from high-quality, compliant manufacturers can influence cost.
- Manufacturing and Formulation Costs: Specialized formulations or complex manufacturing processes will increase COGS.
- Packaging and Distribution Costs: These are standard considerations for all OTC products.
- Target Market and Distribution Channels:
- Mass Market Retailers: Pharmacies, supermarkets, and mass merchandisers typically have higher volume but lower margin expectations.
- Online Retailers: Offer wider reach but are highly price-sensitive.
- Specialty Pharmacies: Might support higher pricing if the product is positioned for specific patient populations, though less likely for a broad OTC.
- Patent Exclusivity (if applicable): While less likely to have strong composition of matter patents for these APIs, any unique formulation or process patents could grant a period of market exclusivity, allowing for higher initial pricing.
Pricing Strategy Options:
- Value-Based Pricing: Pricing based on the perceived benefits and superior outcomes offered by the product compared to alternatives. This strategy is viable if strong clinical evidence supports unique efficacy.
- Competitive Pricing: Setting prices in line with or slightly below direct competitors to gain market share quickly. This is a common approach in crowded OTC markets.
- Skimming Pricing: Initially setting a high price for a differentiated product and then gradually lowering it over time. This is best suited for products with strong IP protection and significant perceived innovation.
Price Projections:
Assuming GS Anti-Diarrheal-Antigas Cplt is launched as a standard OTC combination product with moderate differentiation (e.g., a convenient dosage form), the following projections are plausible:
- Launch Price Point: For a standard package (e.g., 12-24 count), the initial retail price is projected to be in the range of $9 to $15.
- Premium for Enhanced Formulations: If the product incorporates advanced features like extended-release, improved taste, or unique delivery systems, a premium price of $12 to $18 for a comparable package size is achievable.
- Long-Term Pricing (Post-Generic Entry): Once generic versions become available (either of the combination itself or by consumers combining individual products), pricing for branded versions will likely stabilize or decrease, with the brand premium becoming the primary differentiator. The price for the branded product may settle in the $10 to $16 range, while generic alternatives could range from $6 to $12.
Key Considerations for Pricing Success:
- Marketing and Brand Building: Effective marketing is crucial to communicate the product's value proposition and justify its price point.
- Distribution Network Access: Securing placement in major retail chains is essential for broad market penetration.
- Pharmacoeconomic Value: Demonstrating cost-effectiveness (e.g., fewer doctor visits, reduced productivity loss) can indirectly support pricing.
The ultimate pricing will depend on the final product specifications, marketing strategy, and the evolving competitive landscape.
What are the Market Trends and Future Outlook for Anti-Diarrheal and Antigas Medications?
The market for anti-diarrheal and antigas medications is dynamic, influenced by evolving consumer health behaviors, demographic shifts, and advancements in pharmaceutical science. The future outlook for GS Anti-Diarrheal-Antigas Cplt will be shaped by these overarching trends.
Key Market Trends:
- Growing Demand for Digestive Health Solutions:
- Increased Awareness: Consumers are more informed about the impact of gut health on overall well-being, leading to greater demand for products that manage digestive discomfort.
- Dietary and Lifestyle Influences: Modern diets, stress, and sedentary lifestyles continue to contribute to a higher incidence of GI issues, sustaining demand.
- Proactive Health Management: Consumers are increasingly seeking preemptive solutions for digestive issues rather than solely reactive treatment.
- Preference for Combination Therapies:
- Convenience: Patients often experience multiple symptoms simultaneously (e.g., cramping with gas, or loose stools with bloating). Combination products offer the convenience of treating multiple issues with a single medication.
- Improved Compliance: Simplifying treatment regimens can lead to better patient adherence.
- Shift Towards Natural and Probiotic-Based Remedies:
- Consumer Preference: A segment of consumers is actively seeking "natural" alternatives to synthetic drugs, driving growth in the market for probiotics, prebiotics, and herbal remedies for digestive issues.
- Integration with Conventional Medicine: There is a growing interest in the synergistic use of conventional OTC medications with natural supplements.
- Digital Health and Personalized Medicine:
- Telehealth and Online Pharmacies: The growth of digital channels for healthcare consultation and product purchase impacts accessibility and competition.
- Data Analytics: The use of big data to understand consumer behavior, disease prevalence, and treatment effectiveness can inform product development and marketing.
- Personalized Nutrition and Gut Microbiome Research: Advancements in understanding the gut microbiome are paving the way for more personalized approaches to digestive health management.
- Globalization and Emerging Markets:
- Increased Access: As healthcare infrastructure improves in emerging economies, the demand for accessible OTC medications like anti-diarrheals and antigas products is expected to rise.
- Varied Regulatory Frameworks: Navigating diverse regulatory requirements across different global markets is a key challenge for international expansion.
Future Outlook for GS Anti-Diarrheal-Antigas Cplt:
The future outlook for GS Anti-Diarrheal-Antigas Cplt appears moderately positive, contingent on its ability to effectively differentiate itself in a competitive marketplace.
- Market Growth: The overall market for digestive health products is projected to continue its steady growth. GS Anti-Diarrheal-Antigas Cplt, as a dual-action product, is well-positioned to capture a share of this expanding market.
- Competitive Pressure: The primary challenge will be intense competition from established brands and an increasing number of generic alternatives. Success will hinge on:
- Strong Brand Positioning: Clearly communicating the unique benefits of the combination.
- Effective Marketing: Educating consumers about the dual-action advantage.
- Superior Formulation: Offering tangible benefits in terms of efficacy, speed of relief, or ease of use.
- Potential for Expansion:
- New Indications: Future research could explore the efficacy of the specific combination in managing symptoms associated with conditions like Irritable Bowel Syndrome (IBS) or traveler's diarrhea, potentially leading to expanded approved uses and increased market penetration.
- Pediatric Formulations: Developing specialized formulations for pediatric use could open up a significant new market segment, provided safety and efficacy can be adequately demonstrated.
- Impact of Natural Alternatives: The rise of natural and probiotic products presents both a threat and an opportunity. GS Anti-Diarrheal-Antigas Cplt may need to address consumer interest in these areas, perhaps by highlighting its non-systemic absorption (for simethicone) or its role in acute symptom relief that complements longer-term gut health strategies.
The long-term success of GS Anti-Diarrheal-Antigas Cplt will depend on its strategic positioning, robust marketing efforts, and the ability to consistently deliver on its promise of comprehensive relief for common digestive ailments.
Key Takeaways
- GS Anti-Diarrheal-Antigas Cplt operates in a competitive global market for digestive health products, valued at approximately $4.5 billion.
- The product likely combines loperamide (anti-diarrheal) and simethicone (antigas), common OTC APIs with established safety profiles.
- Intellectual property protection for such a combination would primarily rely on novel formulations or manufacturing processes, as core API patents have expired.
- Regulatory classification is expected to be Over-the-Counter (OTC), requiring adherence to FDA or EMA guidelines for labeling and safety.
- Pricing for similar OTC combination products ranges from $8 to $18 per package, with GS Anti-Diarrheal-Antigas Cplt projected to launch in the $9-$15 range, with potential for premium pricing based on differentiation.
- Market growth is driven by increasing consumer awareness of digestive health, lifestyle factors, and a preference for convenient combination therapies.
- Future challenges include intense competition, the rise of natural remedies, and the need for effective brand differentiation.
Frequently Asked Questions
- What is the primary regulatory pathway for GS Anti-Diarrheal-Antigas Cplt in the United States? The primary regulatory pathway in the U.S. is expected to be the Over-the-Counter (OTC) monograph system or potentially an Abbreviated New Drug Application (ANDA) if it mirrors an existing approved product.
- How does simethicone work to relieve gas symptoms? Simethicone is an inert surfactant that reduces the surface tension of gas bubbles in the digestive tract, allowing them to coalesce and be more easily eliminated.
- What is the typical patent duration for a new pharmaceutical formulation? While the standard patent term is 20 years from filing, patent term extensions and data exclusivity periods can provide market protection for a longer duration, often extending well beyond 10 years from initial approval.
- Can GS Anti-Diarrheal-Antigas Cplt be recommended for children? Recommendations for pediatric use depend on specific clinical data and regulatory approval. Standard OTC products typically have age restrictions or require physician consultation for children under a certain age.
- What distinguishes a combination drug like GS Anti-Diarrheal-Antigas Cplt from taking individual anti-diarrheal and antigas medications? A combination drug offers the convenience of a single product to treat multiple symptoms simultaneously, potentially improving patient compliance compared to managing separate medications.
More… ↓
