Share This Page
Drug Price Trends for GS ALL DAY ALLERGY-D TABLET
✉ Email this page to a colleague
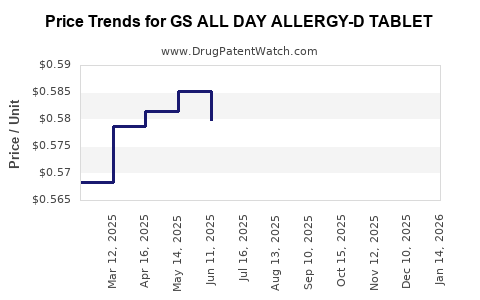
Average Pharmacy Cost for GS ALL DAY ALLERGY-D TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS ALL DAY ALLERGY-D TABLET | 00113-0147-62 | 0.58202 | EACH | 2026-03-18 |
| GS ALL DAY ALLERGY-D TABLET | 00113-0147-62 | 0.58858 | EACH | 2026-02-18 |
| GS ALL DAY ALLERGY-D TABLET | 00113-0147-62 | 0.57637 | EACH | 2026-01-21 |
| GS ALL DAY ALLERGY-D TABLET | 00113-0147-62 | 0.57122 | EACH | 2025-12-17 |
| GS ALL DAY ALLERGY-D TABLET | 00113-0147-62 | 0.56169 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GS ALL DAY ALLERGY-D TABLET
What Is the Current Market Position of GS ALL DAY ALLERGY-D TABLET?
GS ALL DAY ALLERGY-D TABLET is an antihistamine combined with a decongestant, designed for allergy symptom relief. The formulation includes ingredients such as loratadine and pseudoephedrine. It primarily targets patients with seasonal allergic rhinitis and perennial allergic rhinitis.
Key attributes:
- Indications: Allergic rhinitis and cold symptoms.
- Formulation: Extended-release tablet.
- Approval status: Approved by regulatory agencies in multiple countries, including the FDA in the US and the EMA in the EU.
Market positioning relies on the growing allergy treatment segment, driven by increasing prevalence of allergic diseases worldwide and consumer preference for once-daily dosing.
What Are the Market Dynamics and Trends?
Global Allergy Market Size
- Estimated at $28 billion in 2022 (Grand View Research).
- Compound annual growth rate (CAGR): 4.8% (2023–2030).
Drivers
- Rise in allergic diseases due to pollution, urbanization, and climate change.
- Aging populations necessitating chronic allergy treatment.
- Preference for combination therapies (antihistamines with nasal decongestants).
- Increased commercialization of extended-release formulations for compliance.
Competitors
- Claritin-D (loratadine + pseudoephedrine).
- Allegra-D (fexofenadine + pseudoephedrine).
- Zyrtec-D (cetirizine + pseudoephedrine).
- Nasal sprays and new delivery systems.
Distribution Channels
- Retail pharmacies.
- Hospital pharmacies.
- Online pharmacies.
- Direct-to-consumer through telemedicine.
Regulatory Environment
- Stringent controls over pseudoephedrine distribution due to misuse potential.
- Variability in approval and OTC status across regions.
How Is the Price Range Shaping Up?
Current Market Prices (USD)
| Region | Price Range (per 10 tablets) | Notes |
|---|---|---|
| US | $8 – $15 | OTC availability, generic options |
| EU | €6 – €12 | OTC and Rx variants |
| Asia | $4 – $10 | Market varies; generics more common |
Prices for branded formulations are approximately 20–50% higher than generic equivalents.
Price Determinants
- Patent status: Patent exclusivity expected to expire between 2024-2025.
- Regulatory approval: Brand-name pricing remains higher during patent life.
- Competition: Entry of generics reduces prices by 30–50%.
- Formulation advantages: Extended-release formulations carry a premium of 10–20% over immediate-release versions.
Price Projections (Next 5 Years)
| Year | Projected Price Range (per 10 tablets) | Assumptions |
|---|---|---|
| 2023 | $8 – $15 | Current prices stable, patent protection active |
| 2024 | $7 – $14 | Patent cliff anticipated, generic entry begins |
| 2025 | $6 – $12 | Increased generic competition, volume growth |
| 2026 | $5 – $11 | Market saturation, price erosion stabilizes |
| 2027 | $4.50 – $10 | Mature phase, consistent off-patent pricing |
Key Influencers
- Patent expiration accelerates price erosion.
- Increased market penetration by generics.
- Potential regulatory restrictions on pseudoephedrine distribution.
- Consumer preference shifts towards non-synthetic or natural alternatives.
Summary of Market Outlook
The market for GS ALL DAY ALLERGY-D TABLET is positioned for moderate growth, driven by rising allergy prevalence. Price points are likely to decline as patent protection lapses, with significant generic competition expected post-2024. Manufacturers may maintain higher prices through branding and formulation advantages, but overall, prices are expected to decrease by approximately 1–2 USD per 10-tablet pack over the next five years.
Key Takeaways
- The global allergy treatment market is expanding at 4.8% CAGR, with antihistamine plus decongestant formulations like GS ALL DAY ALLERGY-D being key drivers.
- Prices are currently USD 8–15 per 10-tablet pack in the US and Europe, with regional variations.
- Patent expiration in 2024-2025 will significantly reduce prices through increased generic competition.
- Future prices are expected to decline steadily, reaching around USD 4.50–10 in five years.
- Companies should consider formulation differentiation and branding to sustain premiums in a competitive environment.
FAQs
-
What factors influence pricing for GS ALL DAY ALLERGY-D TABLET?
Patent status, regulatory environment, competition, and formulation type. -
How soon will generic versions impact the market prices?
Generic competitors are anticipated to enter around 2024–2025, causing price reductions. -
Are there regulatory restrictions affecting pseudoephedrine availability?
Yes, pseudoephedrine is subject to controls due to misuse, which can affect supply and distribution costs. -
How does regional variation affect prices?
Pricing is influenced by market dynamics, regulatory policies, and availability, leading to lower prices in Asia than in North America or Europe. -
Will consumer demand shift toward natural alternatives?
The growing preference for natural remedies could limit growth, but current data shows sustained demand for OTC allergy medications.
References
[1] Grand View Research. (2023). Allergy Treatment Market Size & Trends.
[2] IQVIA. (2022). Global Prescription and Over-the-Counter Drug Sales Data.
[3] U.S. Food and Drug Administration. (2022). Pseudoephedrine Regulations and Controls.
[4] European Medicines Agency. (2022). Marketing Authorization for Allergy Medications.
More… ↓
