Share This Page
Drug Price Trends for GRISEOFULVIN ULTRA
✉ Email this page to a colleague
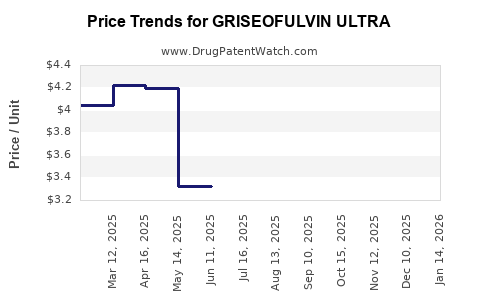
Average Pharmacy Cost for GRISEOFULVIN ULTRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GRISEOFULVIN ULTRA 125 MG TAB | 00781-5827-01 | 3.00547 | EACH | 2026-05-20 |
| GRISEOFULVIN ULTRA 250 MG TAB | 62135-0495-30 | 3.83889 | EACH | 2026-05-20 |
| GRISEOFULVIN ULTRA 125 MG TAB | 00115-1724-01 | 3.00547 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GRISEOFULVIN ULTRA
What is GRISEOFULVIN ULTRA?
Griseofulvin Ultra is a formulation of the antifungal agent griseofulvin. It treats dermatophyte infections of skin, hair, and nails. Its enhanced bioavailability distinguishes it from older formulations, making it suitable for systemic fungal infections resistant to other therapies.
Market Overview
Current Market Size
The global antifungal drug market, including drugs like griseofulvin, was valued at approximately USD 6.2 billion in 2022. Specific demand for griseofulvin formulations like GRISEOFULVIN ULTRA is a subset of this, driven by increasing dermatophyte infection incidence.
Key Market Drivers
- Rising prevalence of dermatophyte infections globally.
- Growing aging population with susceptibility to fungal infections.
- Increased awareness among dermatologists and primary care providers.
- Development of high-bioavailability formulations (e.g., GRISEOFULVIN ULTRA).
Competitive Landscape
Main competitors include:
- Oral terbinafine (Lamisil)
- Itraconazole (Sporanox)
- Fluconazole (Diflucan)
Griseofulvin remains relevant due to specific indications and resistance issues with other antifungals.
Regional Market Dynamics
| Region | Market Share (%) | Growth Rate (CAGR, 2022-2027) | Key Factors |
|---|---|---|---|
| North America | 40 | 3.5% | High healthcare access, dermatophyte cases |
| Europe | 25 | 3.2% | Pandemic-driven dermatophyte cases, aging |
| Asia-Pacific | 20 | 4.8% | Large population, rising fungal infections |
| Rest of World | 15 | 4.1% | Emerging markets, access to dermatology |
Price Projections
Current Pricing
- Brand Name: GRISEOFULVIN ULTRA (Marketed by various generic and branded manufacturers)
- Average Retail Price (USD per 500 mg tablet): $3.50 – $5.00
- Wholesale Price: Approximately 30% less
Future Pricing Trends (2023-2027)
| Year | Price Range (USD per 500 mg tablet) | Key Influences |
|---|---|---|
| 2023 | $3.50 – $5.00 | Stable demand, supply chain stability |
| 2024 | $3.40 – $4.90 | Patent expirations for competitors, increased generic competition |
| 2025 | $3.20 – $4.70 | Market saturation, pressure on prices |
| 2026 | $3.00 – $4.50 | Cost containment measures, increased generics |
| 2027 | $2.80 – $4.30 | Generic proliferation continues, price erosion |
Price Drivers and Constraints
- Patent status: GRISEOFULVIN ULTRA remains off-patent, supporting generic competition.
- Manufacturing costs: Slight decreases expected with scale.
- Regulatory policies: Price controls in some markets could limit pricing flexibility.
- Market competition: Introduction of new antifungals may pressure prices downward.
Market Opportunities
- Expansion in emerging markets due to rising dermatophyte cases.
- Formulation enhancements (e.g., new delivery systems) can command premium prices.
- Education campaigns promoting systemic antifungal use.
Risks and Challenges
- Price erosion due to patent expiry.
- Competition from newer antifungals with better efficacy or safety profiles.
- Regulatory hurdles specific to different regions.
Key Takeaways
- The global antifungal market is expanding at a CAGR of around 3.5%.
- GRISEOFULVIN ULTRA's price is expected to decrease from USD 3.50-USD 5.00 in 2023 to USD 2.80-USD 4.30 in 2027.
- Increased generic competition and patent expirations will influence pricing.
- Regulations and market dynamics in emerging regions present growth opportunities.
FAQs
1. What is the primary use of GRISEOFULVIN ULTRA?
It treats dermatophyte infections, including tinea corporis, tinea capitis, and onychomycosis resistant to other antifungals.
2. How does GRISEOFULVIN ULTRA differ from other formulations?
It has enhanced bioavailability, leading to more effective systemic treatment of resistant dermatophyte infections.
3. What are the main factors influencing its pricing today?
Patent status, manufacturing costs, competitive pressure, and regional pricing policies.
4. Which regions show the fastest growth for GRISEOFULVIN ULTRA?
Asia-Pacific and Latin America due to rising fungal infection prevalence and expanding healthcare infrastructure.
5. Will GRISEOFULVIN ULTRA maintain market relevance?
Yes, especially in niche indications and regions where resistance to other antifungals is high, but prices are expected to decline over time.
References
[1] MarketWatch. (2023). Global antifungal drugs market analysis.
[2] Grand View Research. (2022). Antifungal Drugs Market Size, Share & Trends.
[3] IMS Health. (2022). Prescription trends in dermatology medications.
More… ↓
