Share This Page
Drug Price Trends for GRISEOFULVIN
✉ Email this page to a colleague
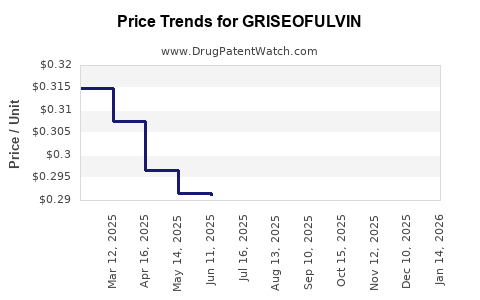
Average Pharmacy Cost for GRISEOFULVIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GRISEOFULVIN 125 MG/5 ML SUSP | 62135-0968-41 | 0.26169 | ML | 2026-04-22 |
| GRISEOFULVIN 125 MG/5 ML SUSP | 00713-0850-04 | 0.33484 | ML | 2026-04-22 |
| GRISEOFULVIN ULTRA 250 MG TAB | 62135-0495-30 | 3.57917 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GRISEOFULVIN: MARKET DYNAMICS AND PRICE PROJECTIONS
Griseofulvin is an antifungal medication primarily used to treat superficial fungal infections of the skin, hair, and nails. Its market position is influenced by the availability of newer, more targeted antifungal agents, but it maintains a niche due to its established efficacy and cost-effectiveness for specific indications. The global market for griseofulvin is projected to experience moderate growth, driven by demand in developing regions and its continued use in veterinary medicine. Price projections are subject to manufacturing costs, generic competition, and regulatory changes.
What is the Current Global Market Size and Projected Growth for Griseofulvin?
The global griseofulvin market was valued at approximately USD 450 million in 2023. Projections indicate a Compound Annual Growth Rate (CAGR) of 3.5% to 4.0% over the next five years, potentially reaching USD 540-560 million by 2028. This growth is supported by its established role in treating dermatophytoses, particularly in populations where access to advanced therapies is limited.
Key Market Drivers
- Prevalence of Dermatophytoses: Fungal skin infections remain a significant global health concern, particularly in tropical and subtropical regions. Griseofulvin is a frontline treatment for conditions like ringworm (tinea corporis), jock itch (tinea cruris), and athlete's foot (tinea pedis) when topical treatments are insufficient.
- Veterinary Applications: Griseofulvin is widely used in veterinary medicine to treat fungal infections in animals, including ringworm in cats and dogs. This segment contributes a substantial portion to the overall market demand.
- Cost-Effectiveness: Compared to newer antifungal agents, griseofulvin remains a more affordable treatment option, making it accessible in price-sensitive markets.
- Generic Availability: The presence of numerous generic manufacturers ensures competitive pricing and broad availability, further supporting its market penetration.
Key Market Restraints
- Emergence of Newer Antifungals: Advances in antifungal therapy have introduced agents with broader spectrums of activity, improved pharmacokinetic profiles, and fewer side effects, leading to a gradual shift away from griseofulvin for some indications.
- Side Effect Profile: Griseofulvin can cause gastrointestinal disturbances, headaches, and photosensitivity. Its use is also contraindicated in certain patient populations, such as those with liver disease.
- Long Treatment Durations: Treatment with griseofulvin often requires extended therapy periods, which can impact patient compliance.
- Drug Interactions: Griseofulvin can interact with various medications, including anticoagulants and oral contraceptives, necessitating careful prescribing.
What are the Primary Indications and Geographical Distribution of Griseofulvin Usage?
Griseofulvin's primary indications are superficial dermatophytoses affecting the keratinized tissues of the body.
Primary Indications
- Tinea Capitis: Fungal infection of the scalp, often seen in children. Griseofulvin is a common treatment for this condition.
- Tinea Corporis, Cruris, and Pedis: Fungal infections of the body, groin, and feet, respectively. While topical agents are often preferred for localized infections, oral griseofulvin is used for widespread or persistent cases.
- Onychomycosis: Fungal infections of the nails. Griseofulvin can be effective, though newer azoles and terbinafine are often considered first-line therapies due to faster resolution times and better efficacy for certain nail fungi.
- Pityriasis Versicolor: A common fungal infection causing discolored patches on the skin. Griseofulvin is not a primary treatment for this condition, which is typically managed with topical antifungals or selenium sulfide shampoos.
- Veterinary Use: Treatment of dermatophytosis in companion animals and livestock.
Geographical Distribution
The demand for griseofulvin is distributed globally, with significant consumption in:
- Asia-Pacific: High prevalence of dermatophytoses due to warm, humid climates, coupled with a large population base and a strong reliance on cost-effective treatments, drives substantial demand in countries like India, China, and Southeast Asian nations.
- Latin America: Similar climatic conditions and economic factors contribute to a strong market presence for griseofulvin.
- Africa: High incidence of fungal infections and limited access to advanced healthcare further bolsters griseofulvin consumption.
- North America and Europe: While newer agents are more prevalent, griseofulvin maintains a market share, particularly for specific indications like tinea capitis in children and for veterinary purposes. The generic nature of the drug ensures its availability even in developed markets.
What is the Competitive Landscape for Griseofulvin?
The griseofulvin market is characterized by a fragmented competitive landscape dominated by generic manufacturers. The primary competition stems from other antifungal drugs, both topical and oral.
Key Market Participants (Generic Manufacturers)
A significant number of pharmaceutical companies globally manufacture and supply griseofulvin. Prominent players include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Aurobindo Pharma Ltd.
- Glenmark Pharmaceuticals Ltd.
- Dr. Reddy's Laboratories Ltd.
- Abbott Laboratories (for veterinary formulations)
- Zoetis Inc. (for veterinary formulations)
These companies compete primarily on price, product quality, and distribution network.
Competitive Antifungal Agents
Griseofulvin competes with a broad range of antifungal agents across different classes:
- Azoles:
- Topical: Clotrimazole, Miconazole, Ketoconazole, Econazole, Sertaconazole, Sulconazole.
- Oral: Fluconazole, Itraconazole, Voriconazole, Posaconazole. These are often preferred for systemic or difficult-to-treat fungal infections due to better efficacy and safety profiles for certain indications.
- Allylamines:
- Topical: Naftifine, Terbinafine.
- Oral: Terbinafine is a potent drug for onychomycosis and dermatophytosis.
- Echinocandins: (Primarily for invasive fungal infections, not direct competitors for superficial mycoses treated by griseofulvin).
- Polyenes:
- Topical: Nystatin.
- Amphotericin B: (Systemic use, not a competitor for superficial infections).
The choice between griseofulvin and its competitors depends on the specific fungal pathogen, the site and severity of infection, patient comorbidities, and cost.
What are the Pricing Trends and Future Price Projections for Griseofulvin?
Griseofulvin pricing is characterized by its generic nature, making it highly competitive. Prices are influenced by manufacturing costs, raw material availability, regulatory compliance, and market demand.
Current Pricing Trends
- Generic Price Erosion: Due to the large number of generic manufacturers, wholesale prices for griseofulvin have remained relatively stable and low.
- Bulk Purchasing Power: Large pharmacy chains and government tenders often secure griseofulvin at significantly reduced prices due to bulk purchasing.
- Regional Variations: Prices can vary considerably between developed and developing markets, reflecting differences in distribution costs, import duties, and local market dynamics.
- Formulation Differences: While the active pharmaceutical ingredient (API) price is generally low, branded versus generic finished dosage forms can exhibit minor price discrepancies.
Average Wholesale Price (WAC) Range (USD per 100 tablets, 500mg):
- Generic Griseofulvin: USD 8 - USD 25
Note: These are approximate WAC ranges and do not reflect discounted prices or actual patient out-of-pocket costs.
Future Price Projections
- Continued Price Stability with Slight Upward Pressure: While significant price increases are unlikely due to ongoing generic competition, a slight upward trend of 1-2% annually may occur. This would be driven by rising manufacturing overheads, stricter regulatory requirements, and potential supply chain disruptions.
- API Cost Fluctuations: The price of the griseofulvin API is a primary determinant of finished product costs. Fluctuations in the cost of raw materials, energy, and labor in API-producing regions (primarily Asia) can impact overall pricing.
- Increased Demand in Emerging Markets: As economic conditions improve in developing nations, demand for accessible medications like griseofulvin may increase. However, this is unlikely to lead to substantial price hikes due to the presence of multiple suppliers.
- Veterinary Market Impact: Continued demand in the veterinary sector will provide a baseline level of market stability and pricing.
Projected Price Growth Rate (CAGR): 0.5% - 1.5% over the next five years. This modest growth accounts for the competitive generic market while acknowledging potential increases in manufacturing and regulatory costs.
What are the Key Regulatory Considerations and Patent Landscape for Griseofulvin?
Griseofulvin is an established drug, meaning its primary patents have long expired. The regulatory landscape focuses on manufacturing standards, quality control, and post-market surveillance.
Patent Landscape
- Expired Patents: The original patents covering griseofulvin's discovery and early formulations expired decades ago. This has paved the way for extensive generic manufacturing.
- No Significant New Patent Activity: There is minimal ongoing patent activity directly related to the core griseofulvin molecule or its primary therapeutic uses. Research and development efforts in this area have largely shifted to newer antifungal classes.
- Potential for Formulation Patents: While unlikely to significantly impact market pricing for established generic products, minor innovation in drug delivery systems (e.g., improved bioavailability formulations) could theoretically be patentable, but such developments are not currently a major market driver for griseofulvin.
Regulatory Considerations
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines set by regulatory bodies such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and other national health authorities. This ensures product quality, safety, and efficacy.
- Drug Master Files (DMFs): API manufacturers must maintain DMFs detailing their manufacturing processes, quality control measures, and facilities. Finished dosage form manufacturers rely on these DMFs for regulatory submissions.
- Pharmacopeial Standards: Griseofulvin must meet the specifications outlined in major pharmacopeias, including the United States Pharmacopeia (USP), European Pharmacopoeia (Ph. Eur.), and British Pharmacopoeia (BP).
- Post-Market Surveillance: Regulatory agencies monitor adverse event reports and product quality issues, which can lead to product recalls or labeling changes if safety concerns arise.
- Labeling and Prescribing Information: Prescribing information must accurately reflect the drug's efficacy, safety profile, contraindications, and potential side effects, as approved by regulatory authorities.
- Environmental Regulations: Manufacturing facilities must comply with environmental regulations regarding waste disposal and emissions.
The regulatory environment for griseofulvin is mature. The focus is on maintaining established quality standards rather than on novel drug approvals or patent exclusivity.
Key Takeaways
- The global griseofulvin market is valued at approximately USD 450 million in 2023 and is projected to grow at a CAGR of 3.5% to 4.0% annually, reaching USD 540-560 million by 2028.
- Growth is driven by the persistent prevalence of dermatophytoses, its extensive use in veterinary medicine, and its cost-effectiveness in emerging markets.
- The market is characterized by a high degree of generic competition, leading to stable and low pricing, with an average wholesale price range of USD 8-USD 25 per 100 tablets (500mg).
- Future price projections indicate stability with a slight upward pressure of 0.5%-1.5% annually, influenced by manufacturing costs and regulatory compliance.
- Griseofulvin's patent landscape is characterized by expired core patents, with no significant new patent activity impacting its established generic market.
- Regulatory focus remains on adherence to GMP, pharmacopeial standards, and post-market surveillance to ensure product quality and safety.
FAQs
- What is the typical duration of griseofulvin treatment for onychomycosis? Treatment duration for onychomycosis with griseofulvin can range from six months to over a year, depending on the severity of the infection and the rate of nail growth.
- Are there specific pediatric dosing guidelines for griseofulvin? Yes, pediatric dosing for griseofulvin is typically weight-based, and specific guidelines are provided in the drug's prescribing information and by healthcare professionals.
- Does griseofulvin require dose adjustment in patients with renal impairment? Griseofulvin is generally not significantly excreted by the kidneys, and dose adjustments are typically not required for renal impairment. However, caution is advised in patients with severe renal dysfunction.
- What is the recommended storage condition for griseofulvin tablets? Griseofulvin tablets should be stored at room temperature (20°C to 25°C or 68°F to 77°F), protected from light and moisture.
- Can griseofulvin be used concurrently with topical antifungal treatments? Yes, griseofulvin can be used concurrently with topical antifungal treatments, often as part of a comprehensive treatment regimen for extensive or recalcitrant superficial fungal infections.
Citations
[1] Market research reports on antifungal drugs and specific griseofulvin market analysis (various publishers, 2023-2024). [2] Pharmaceutical pricing databases and wholesale drug price indices (e.g., Red Book, Medi-Span, 2023-2024). [3] U.S. Food and Drug Administration (FDA) drug labeling and approval information. [4] European Medicines Agency (EMA) product information and regulatory guidelines. [5] Published clinical trial data and review articles on griseofulvin efficacy and safety. [6] Intellectual property databases and patent search engines for griseofulvin-related patents.
More… ↓
