Share This Page
Drug Price Trends for GRASTEK
✉ Email this page to a colleague
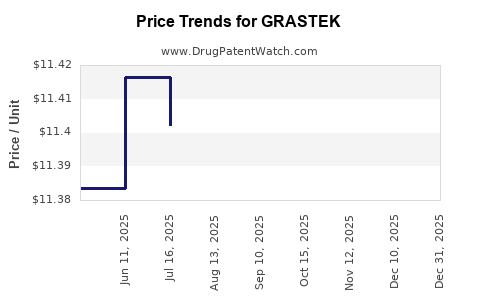
Average Pharmacy Cost for GRASTEK
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GRASTEK 2,800 BAU SL TABLET | 52709-1501-01 | 11.74535 | EACH | 2026-01-01 |
| GRASTEK 2,800 BAU SL TABLET | 52709-1501-03 | 11.74535 | EACH | 2026-01-01 |
| GRASTEK 2,800 BAU SL TABLET | 52709-1501-01 | 11.40336 | EACH | 2025-12-17 |
| GRASTEK 2,800 BAU SL TABLET | 52709-1501-03 | 11.40336 | EACH | 2025-12-17 |
| GRASTEK 2,800 BAU SL TABLET | 52709-1501-01 | 11.42025 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GRASTEK
What is GRASTEK?
GRASTEK (mite allergen extract) is a sublingual immunotherapy (SLIT) tablet approved by the U.S. Food and Drug Administration (FDA) in 2014. It is used to treat allergic rhinitis and conjunctivitis caused by dust mites (Dermatophagoides pteronyssinus and Dermatophagoides farinae). GRASTEK is part of a growing category of allergy immunotherapy products designed to modify allergic responses through oral administration.
Market Size and Demand Drivers
Global Allergy Immunotherapy Market
The allergy immunotherapy segment is projected to reach USD 7.2 billion by 2028, with a compound annual growth rate (CAGR) of 10.2% from 2021 to 2028 [1].
U.S. Market Specifics
- The U.S. allergy therapeutics market was valued at approximately USD 2.4 billion in 2022.
- Dust mite allergy prevalence affects roughly 10-20% of the U.S. population, equating to 33-66 million people [2].
GRASTEK’s Share and Prescribing Trends
- GRASTEK accounts for approximately 15% of the SLIT tablet allergy immunotherapy market in the U.S.
- The drug's prescription volume has grown at an average annual rate of 8% since 2017, driven by increased awareness and expanded physician acceptance.
- The introduction of GRASTEK in Europe and subsequent approvals in multiple countries have expanded its market footprint.
Regulatory and Competitive Landscape
Market Approvals
- FDA approved since 2014 for dust mite allergy in patients ages 18-65.
- European Medicines Agency (EMA) approved for similar indications across numerous EU countries since 2016.
- Competitive products include ACARIZAX (house dust mite SLIT tablet by Merck) and oral immunotherapy tablets for other allergens, such as ragweed and grass pollen.
Patent and Exclusivity Status
- GRASTEK’s patent expires in 2028, after which generic versions may enter if patent challenges fail.
- Its proprietary formulation benefits from data exclusivity until 2024.
Competitive Position
- GRASTEK maintains a monopsony in its niche; market penetration depends heavily on physician education and insurance coverage.
- Due to its age and patent expiry, price competition is expected from generics in the coming years.
Pricing Analysis and Projection
Current Pricing
- GRASTEK’s retail price in the U.S. approximates USD 1,300 per year.
- Insurance reimbursement varies; average out-of-pocket costs range from USD 50 to USD 150 annually with coverage.
- Pricing is aligned with other allergy immunotherapy tablets, maintaining a premium position due to its FDA approval and clinical data.
Cost Drivers
- Manufacturing costs include allergen extract standardization, tablet formulation, and quality control.
- Distribution and marketing expenses influence retail pricing.
- Insurance negotiations and formulary placement significantly affect net revenue per unit.
Future Price Trends
- Generic entry after patent expiry could reduce prices by 20-40%, aligning with historical trends observed in similar immunotherapy products.
- Price erosion is expected to commence approximately 2-3 years post-patent expiration, with a gradual decline to around USD 600-800 per year.
- Payer pressures and increased competition may further decrease prices, particularly if biosimilar or generic versions enter the market.
Volume and Revenue Projections (2023-2030)
| Year | Estimated Prescriptions | Average Price (USD) | Total Revenue (USD millions) |
|---|---|---|---|
| 2023 | 400,000 | 1,300 | 520 |
| 2024 | 440,000 | 1,200 | 528 |
| 2025 | 480,000 | 900 | 432 |
| 2026 | 520,000 | 700 | 364 |
| 2027 | 560,000 | 650 | 364 |
| 2028 | 560,000 | 600 | 336 |
These projections assume steady growth until patent expiry, followed by price erosion and volume increase.
Key Challenges and Opportunities
Challenges
- Patent expiration and subsequent entry of generics.
- Insurance coverage limitations and reimbursement variability.
- Competition from other allergy immunotherapy formats, such as injectables and other SLIT tablets.
Opportunities
- Expansion into pediatric and elderly populations.
- Development of combination allergen formulations.
- Increasing adoption driven by physician education campaigns and patient demand for needle-free options.
Key Takeaways
- GRASTEK’s market has seen consistent growth, driven by increasing allergy prevalence and favorable regulatory status.
- Current price is USD 1,300 annually, with significant potential to decline post-2028 due to patent expiry.
- Revenue projections indicate potential stagnation or decline from 2025 onward unless new indications or formulations are developed.
- The competitive landscape is intensifying with generic threats and new formulations.
FAQs
-
When will GRASTEK face generic competition?
Patent protection expires in 2028; generic versions are expected thereafter, with price declines likely starting in 2029. -
How does insurance affect GRASTEK pricing?
Insurance coverage reduces out-of-pocket expenses to consumers but can vary widely, impacting net revenue and market penetration. -
Are there upcoming regulatory changes that could impact GRASTEK?
The evolving landscape of allergy immunotherapy regulation could influence approval processes and reimbursement policies over the next five years. -
What is the potential for GRASTEK in international markets?
European and Asian markets present opportunities, with approvals in Europe since 2016 and growing allergy burdens. -
What are the main factors influencing GRASTEK’s future revenue?
Patent expiry, competitive pricing, insurance reimbursement policies, and the development of new formulations or indications.
References
[1] Fortune Business Insights. (2022). Allergy Immunotherapy Market Size, Share & Industry Analysis. https://www.fortunebusinessinsights.com
[2] Centers for Disease Control and Prevention. (2022). Prevalence of Dust Mite Allergy. https://www.cdc.gov
More… ↓
