Share This Page
Drug Price Trends for GONAL-F RFF REDI-JECT
✉ Email this page to a colleague
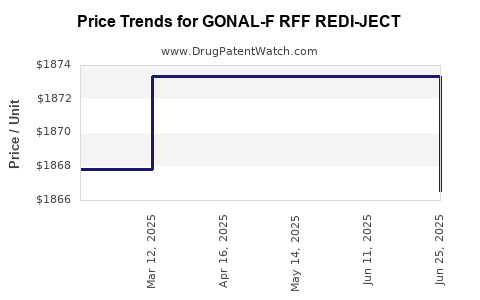
Average Pharmacy Cost for GONAL-F RFF REDI-JECT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GONAL-F RFF REDI-JECT 900 UNIT PEN | 44087-1117-01 | 1873.34445 | ML | 2025-06-18 |
| GONAL-F RFF REDI-JECT 900 UNIT PEN | 44087-1117-01 | 1873.34445 | ML | 2025-05-21 |
| GONAL-F RFF REDI-JECT 900 UNIT PEN | 44087-1117-01 | 1873.34445 | ML | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GONAL-F RFF REDI-JECT
Summary
GONAL-F RFF REDI-JECT, a recombinant human follicle-stimulating hormone (FSH) formulated for subcutaneous injection, targets infertility treatments. Its market relies on fertility clinics, endocrinologists, and hospitals. Commercially, it faces competition from biosimilars and branded alternatives. Pricing varies based on regional regulatory landscape, reimbursement policies, and competitive dynamics. Current price projections suggest stabilization within a range influenced by market penetration and regulatory adjustments over the next five years.
What Is the Market Size for GONAL-F RFF REDI-JECT?
Market size estimates come from the combined valuation of global infertility treatment segments, which include medications, assisted reproductive technology (ART) procedures, and monitoring. The fertility drugs segment was valued at approximately $650 million in 2022, with the reproductive gonadotropins segment representing a significant share.
- Key regions: North America (40%), Europe (30%), Asia-Pacific (20%), Rest of World (10%) [1].
- Growth rate: The global fertility drugs market CAGR is projected at 8.5% through 2030, driven by aging populations, increasing infertility awareness, and adoption of ART procedures [2].
Market drivers include:
- Higher infertility prevalence.
- Technological advancements in ART.
- Increasing reimbursement coverage in developed markets.
Market constraints include:
- High drug costs influencing patient access.
- Competition from biosimilars.
- Regulatory and patent expiration fears.
What Are the Key Competitors?
GONAL-F RFF REDI-JECT’s primary competitors include:
- Ovaleap (follitropin alfa): Biosimilar, marketed since 2013.
- Puregon (follitropin beta): Marketed globally with availability in multiple forms.
- Follistim AQ (follitropin beta): Widely used, available in multiple formulations.
- Fostimon (menotropins): Derived from urine, still in use but declining.
Biosimilar entry has compressed prices, especially in Europe, leading to aggressive price competition.
What Are the Price Dynamics and Trends?
Current pricing in major markets:
| Market | Price per injection (USD) | Note |
|---|---|---|
| US | $95 - $115 | Retail, insurance influences |
| EU | €40 - €70 ($45 - $78) | Varies by country, biosimilar competition |
| Asia-Pacific | $30 - $60 | Lower due to price sensitivity |
Pricing influences include:
- Patent cliffs: GONAL-F’s patent expired in Europe in 2018, enabling biosimilars.
- Market penetration: Higher in regions with reimbursement support.
- Regulatory policies: In some regions, price negotiations and reference pricing limit increases.
Projected price trends (2023-2028):
- Price stabilization in the US due to high brand loyalty.
- European prices may decline 10-15% driven by biosimilar adoption.
- Asia-Pacific prices expected to decrease further owing to market competition and cost pressures.
What Are the Regulatory and Reimbursement Factors?
- United States: FDA approval of biosimilars in 2015 has led to increased competition; reimbursement policies favor cost-effective options.
- Europe: EMA approval of biosimilars since 2007; price cuts mandated by national agencies. GONAL-F is subject to price negotiations; biosimilar uptake continues.
- Asia-Pacific: Governments promote affordability; reimbursement policies vary, but price reductions are common.
Pricing is also affected by manufacturer negotiations with payers and healthcare providers, with some markets implementing reference pricing systems.
What Are the Future Price Projections?
Based on current trends and competitive pressures:
- North America: Prices are likely to plateau at approximately $90-$110 per injection over five years.
- Europe: Prices could decline by 15%-20%, reaching €35-€55 per injection.
- Asia-Pacific: Prices might decrease further by 20-25%, settling around $25-$45.
Impact factors include new biosimilar approvals, policy shifts, and market share reallocation from branded to biosimilar products.
Conclusions
GONAL-F RFF REDI-JECT positions itself within a highly competitive, price-sensitive market. The patent expiration and biosimilar proliferation exert downward pressure on prices globally. Market growth will continue driven by increasing infertility rates and ART adoption, but pricing strategies will adapt to regulatory environments and payer pressures.
Key Takeaways
- The global fertility drugs market is expanding at approximately 8.5% CAGR, with gonadotropins being a major segment.
- Pricing varies regionally, with North America maintaining higher prices; Europe and Asia see significant declines due to biosimilars.
- Price projections suggest stabilization in North America, further reductions in Europe and Asia over the next five years.
- Biosimilar competition is the most significant factor influencing price trends.
- Regulatory policies will continue to shape pricing strategies and market access.
FAQs
1. How does biosimilar entry affect GONAL-F RFF REDI-JECT pricing?
Biosimilar entry creates price competition, prompting original products to lower prices or improve value propositions, leading to overall market price declines.
2. What is the potential for future price increases in the US?
Limited potential exists due to market saturation and insurance price controls; prices are likely to remain stable or decline gradually.
3. Are there regional differences in reimbursement policies affecting pricing?
Yes. Countries with centralized healthcare systems or aggressive negotiation mechanisms tend to have lower drug prices compared to markets with less regulation.
4. How does patent expiration influence future pricing?
Patent expiration allows biosimilars, which are typically priced 20-40% lower, to enter the market, exerting downward pressure on the original biologic.
5. What role do clinical guidelines play in pricing?
Guidelines that recommend cost-effective treatments influence payer coverage decisions, indirectly affecting drug pricing strategies.
References
[1] MarketsandMarkets. Fertility Drugs Market, 2022.
[2] Grand View Research. Fertility Drugs Market Size, 2022-2030.
More… ↓
