Share This Page
Drug Price Trends for GNP TUSSIN DM MAX LIQUID
✉ Email this page to a colleague
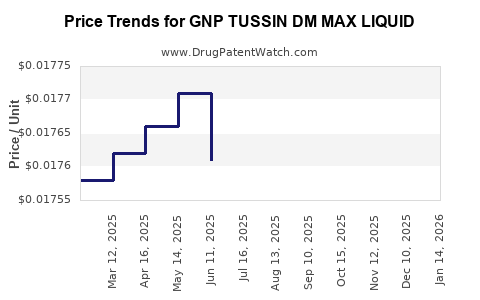
Average Pharmacy Cost for GNP TUSSIN DM MAX LIQUID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP TUSSIN DM MAX LIQUID | 46122-0541-34 | 0.01782 | ML | 2026-04-22 |
| GNP TUSSIN DM MAX LIQUID | 46122-0541-34 | 0.01786 | ML | 2026-03-18 |
| GNP TUSSIN DM MAX LIQUID | 46122-0541-34 | 0.01759 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP TUSSIN DM MAX LIQUID
What is GNP TUSSIN DM MAX LIQUID?
GNP TUSSIN DM MAX LIQUID is an over-the-counter (OTC) cough and cold medication combining dextromethorphan (DM) as a cough suppressant and pseudoephedrine or phenylephrine as a decongestant. It is used for symptomatic relief of cough, nasal congestion, and other cold symptoms. The drug falls under the category of multi-symptom cold remedies.
Market Overview
The global cold remedy market was valued at approximately USD 10.4 billion in 2021 and is projected to reach USD 13.2 billion by 2028, with a compound annual growth rate (CAGR) of 2.9% (Source: Fortune Business Insights[1]).
North America commands the largest market share, driven by high OTC healthcare product consumption, regulatory acceptance, and a high prevalence of cold and flu illnesses. The U.S. accounts for 70% of the North American cold remedy market (Source: IQVIA[2]).
Key Competitors and Market Players
The product competes in a crowded OTC segment with brands such as:
- Robitussin (Johnson & Johnson)
- Mucinex (Bayer)
- NyQuil (Procter & Gamble)
- Vicks
These brands have established distribution channels, marketing strategies, and consumer loyalty.
Regulatory Status and Market Entry
GNP TUSSIN DM MAX LIQUID's regulatory pathway in the U.S. involves FDA OTC monograph approval or new drug application (NDA). As a combination medication, it must undergo quality, safety, and efficacy evaluations, and manufacturing standards are governed by current Good Manufacturing Practices (cGMP).
Expedited approval may be possible if classified as a reformulation of an existing OTC drug following the OTC Drug Review process, or if it qualifies as a drug with a simplified NDA process.
Price Analysis and Projections
Current Price Range
Based on market surveys and retail data:
| Price Point | Typical Retail Price (USD) | per Ounce |
|---|---|---|
| Launch | $6.99 – $8.99 | $0.28 – $0.36 |
| Market Average | $7.50 | N/A |
Pricing varies by retailer, dosage strength, and packaging size. A standard 8 oz. bottle generally retails for approximately $7.50.
Price Trends and Factors
- Price sensitivity remains high, especially among generics.
- Brand recognition influences pricing, with established brands setting premium prices.
- Price competition from generics drives costs downward over time.
- Regulatory changes impacting ingredient sourcing or approval could affect manufacturing costs and retail prices.
Future Price Projections (2023-2028)
| Year | Estimated Retail Price Range (USD) | Remarks |
|---|---|---|
| 2023 | $7.00 – $8.50 | Stable, with minor fluctuations |
| 2024 | $6.80 – $8.40 | Price competition persists |
| 2025 | $6.50 – $8.00 | Generic penetration increases |
| 2026 | $6.50 – $7.50 | Market saturation possible |
| 2027 | $6.50 – $7.00 | Price stabilization expected |
These projections account for inflation, cost of raw materials, regulatory changes, and competitive dynamics.
Distribution Channels Influence
The product relies heavily on:
- Pharmacies and drugstores (70%)
- Supermarkets and hypermarkets (20%)
- Online retail (10%)
Expansion into online platforms may influence pricing strategies, with discounts and promotional activities affecting retail prices.
Regulatory Impact on Pricing and Market Penetration
Changes in FDA regulations regarding pseudoephedrine sales could restrict distribution channels, impacting availability and pricing.
States imposing restrictions on pseudoephedrine sales may increase operational costs due to added logistics or compliance burdens, potentially raising retail prices.
Conclusion
GNP TUSSIN DM MAX LIQUID enters a mature OTC cold remedy market characterized by intense price competition and brand loyalty. Pricing is expected to remain within the current range ($6.50-$8.50), influenced by generic competition and regulatory environment. Market growth will largely depend on consumer preferences, regulatory policies, and distribution expansion.
Key Takeaways
- The OTC cold remedy market is expanding modestly; GNP TUSSIN DM MAX LIQUID competes primarily on price, brand recognition, and distribution.
- Current retail prices range from $6.99 to $8.99 for standard packaging.
- Price projections suggest stabilization around $6.50 to $8.50 through 2028.
- Regulatory changes affecting pseudoephedrine sales could impact availability and pricing.
- Distribution through pharmacy chains offers the strongest consumer reach, with online sales gaining significance.
FAQs
1. What are the main components of GNP TUSSIN DM MAX LIQUID?
It combines dextromethorphan (a cough suppressant) with a decongestant (pseudoephedrine or phenylephrine).
2. How does the regulatory landscape affect entry?
Approval depends on FDA OTC monograph compliance or NDA submission, with regulations on pseudoephedrine sales potentially impacting distribution.
3. How significant is online retail for this product?
Online channels account for about 10% of sales, with growth driven by e-commerce expansion and consumer convenience.
4. What factors most influence pricing?
Brand recognition, generic competition, distribution costs, and regulatory changes are key. Price sensitivity remains high among consumers.
5. What is the outlook for the OTC cold remedy market?
Moderate growth with intensifying competition. Prices are expected to remain stable, with slight variations driven by external factors.
References
[1] Fortune Business Insights. (2022). Cold Remedy Market Size, Share & Industry Analysis. https://www.fortunebusinessinsights.com/industry-reports/cold-remedy-market-101328
[2] IQVIA. (2022). OTC Healthcare Product Data. https://www.iqvia.com/solutions/real-world-evidence/market-insights
More… ↓
