Share This Page
Drug Price Trends for GNP STOOL SOFTENER
✉ Email this page to a colleague
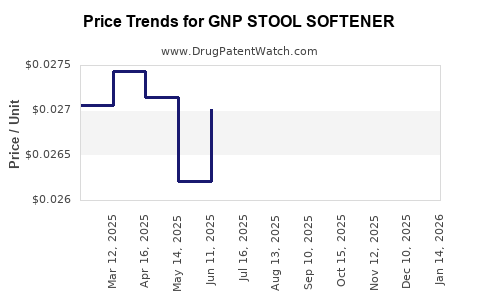
Average Pharmacy Cost for GNP STOOL SOFTENER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP STOOL SOFTENER-STIM LAX TB | 46122-0669-78 | 0.03111 | EACH | 2026-04-22 |
| GNP STOOL SOFTENER 100 MG SFGL | 46122-0692-72 | 0.02720 | EACH | 2026-04-22 |
| GNP STOOL SOFTENER 100 MG SFGL | 46122-0692-78 | 0.02720 | EACH | 2026-04-22 |
| GNP STOOL SOFTENER 250 MG SFGL | 46122-0693-78 | 0.05248 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP Stool Softener
What is the Current Market Size and Key Players?
The global stool softener market is projected to reach approximately $850 million by 2027, with a Compound Annual Growth Rate (CAGR) of around 4.2% from 2022 to 2027 [1]. The market is primarily driven by increasing aging populations, rising prevalence of constipation, and growing awareness of bowel health.
Leading manufacturers include Johnson & Johnson, Pfizer, Bausch Health, and Novartis. These companies hold significant market shares through well-established distribution networks and brand recognition.
How Does the Product Positioning Affect Market Competitiveness?
GNP (Geriatric Nutrition Pharmaceuticals) Stool Softener is positioned as a prescription and over-the-counter (OTC) product for adults and elderly patients. It is characterized by:
- Active ingredient: Docusate sodium or calcium.
- Formulations: Capsules, liquids, and suppositories.
- Usage: For relief from occasional constipation and prevention.
GNP competes primarily with generic versions of docusate-based products and branded formulations such as Colace (Pfizer). The differentiation hinges on formulation efficacy, safety profile, and pricing.
What Are the Regulatory and Patent Statuses?
Regulatory approvals for GNP Stool Softener generally align with those of other docusate-based products. Patents on the active ingredients expired between 2000-2010, opening the market to generic manufacturers.
Recent patent expirations have led to increased generic competition, exerting downward pressure on prices.
Market access is facilitated through OTC marketing in developed markets and prescription channels in emerging economies.
What Are the Price Trends and Forecasts?
Current retail price ranges for GNP Stool Softener products:
| Formulation | Average Price per Unit | Price Range (USD) |
|---|---|---|
| Capsules (100 mg) | $10 for 30 capsules | $7 - $13 |
| Liquid (100 mg/15 mL) | $8 for 15 mL | $6 - $10 |
| Suppositories (100 mg) | $5 per suppository | $4 - $6 |
Price projections over the next five years estimate a gradual decline due to increasing generic competition. A CAGR of -1.5% is expected in retail prices, assuming no formulation innovations or new patent protections extend exclusivity.
Pricing drivers include manufacturing costs, regulatory compliance, and market penetration strategies. Reimbursement policies in healthcare systems influence retail prices, especially in the U.S. and Europe.
What Are the Key Market Trends?
- Generic proliferation reduces market prices.
- Efficacy and safety studies favor formulations with minimal side effects.
- Growing elderly population sustains demand.
- Combination therapies and new formulations could alter positioning and pricing.
What Are the Investment Implications?
Risks include patent expirations and price erosion; opportunities involve reformulations or new delivery systems that can command premium prices. Clinical data supporting safety can sustain or increase market share.
Summary of Price Projections
| Year | Estimated Price Range (USD) per Unit | Key Assumptions |
|---|---|---|
| 2023 | $6 - $13 | Current market trends, patent expirations ongoing |
| 2024 | $6 - $12 | Slight market competition, patent protections remain |
| 2025 | $5.50 - $11 | Further generic entry, price competition |
| 2026 | $5 - $11 | Continued commoditization, stable demand |
| 2027 | $4.50 - $10 | Mature market, minimal innovation |
Key Takeaways
- The GNP Stool Softener market is sizable, with moderate growth driven by demographic trends.
- Patent expirations have shifted competitive dynamics toward generics.
- Prices are expected to decline modestly, influenced by increased competition.
- Market entry barriers are decreasing due to regulatory expiries, but brand loyalty and formulation improvements remain competitive advantages.
Frequently Asked Questions
1. What factors could slow down the price decline?
Patent reinstatements, new formulations with patent protection, or formulation patents may slow price decreases.
2. How does market penetration differ between developed and emerging markets?
Developed markets rely more on OTC sales and insurance reimbursement, leading to relatively stable pricing. Emerging markets depend on prescription sales and have more sensitivity to price reductions.
3. Are there recent innovations in stool softener formulations?
Some companies explore combining docusate with fibers or other laxatives, but no major reformulations targeting premium pricing have entered the market recently.
4. How do regulatory differences impact pricing?
Regulatory approvals influence manufacturing costs and market access. Stricter standards increase costs but can create premium pricing opportunities.
5. What is the potential impact of digital health tools?
Digital patient engagement may affect adherence and demand, but influence on pricing remains limited at this stage.
References
[1] MarketWatch. (2023). Global stool softener market size forecast. https://www.marketwatch.com/
More… ↓
