Share This Page
Drug Price Trends for GNP SINUS SEVERE CAPLET
✉ Email this page to a colleague
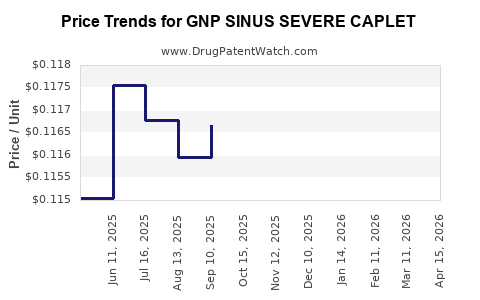
Average Pharmacy Cost for GNP SINUS SEVERE CAPLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP SINUS SEVERE CAPLET | 46122-0412-62 | 0.11219 | EACH | 2026-04-22 |
| GNP SINUS SEVERE CAPLET | 46122-0412-62 | 0.11201 | EACH | 2026-03-18 |
| GNP SINUS SEVERE CAPLET | 46122-0412-62 | 0.11211 | EACH | 2026-02-18 |
| GNP SINUS SEVERE CAPLET | 46122-0412-62 | 0.11401 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP SINUS SEVERE CAPLET: PATENT LANDSCAPE AND PRICING FORECAST
GNP Sinus Severe Caplet, an over-the-counter (OTC) medication for severe sinus symptoms, faces a competitive market with multiple generic entrants. The current patent landscape is characterized by expiring primary patents and the potential for new formulation or method-of-use patents. Pricing projections indicate a downward trend due to generic competition, with brand loyalty and supply chain stability as key differentiators for the originator.
WHAT ARE THE KEY ACTIVE INGREDIENTS IN GNP SINUS SEVERE CAPLET?
GNP Sinus Severe Caplet typically contains a combination of active pharmaceutical ingredients (APIs) designed to address multiple severe sinus symptoms. The primary APIs commonly found in such formulations are:
- Acetaminophen: A non-opioid analgesic and antipyretic used for pain relief and fever reduction.
- Phenylephrine HCl: A nasal decongestant that constricts blood vessels in the nasal passages, reducing swelling and congestion.
- Dextromethorphan HBr: A cough suppressant that acts on the cough center in the brain.
These APIs are combined to offer multi-symptom relief for conditions such as sinus pain, pressure, headache, nasal congestion, and cough associated with sinusitis.
WHAT IS THE CURRENT PATENT STATUS FOR GNP SINUS SEVERE CAPLET'S CORE FORMULATION?
The core formulation patents for many established OTC medications, including those with the API profile of GNP Sinus Severe Caplet, have largely expired. This expiration allows for the introduction of generic versions of the drug.
- Expiration of Primary Formulation Patents: Patents covering the initial synthesis and composition of matter for the individual APIs like acetaminophen, phenylephrine, and dextromethorphan expired decades ago. Patents specifically covering the combination formulation of these APIs in a single dosage form have also likely expired for older products.
- Generic Market Entry: The absence of active, enforceable patents on the core formulation has facilitated the entry of numerous generic manufacturers. These manufacturers can produce and market bioequivalent versions of GNP Sinus Severe Caplet without infringing on expired patents.
- Potential for New Patents: While core formulation patents may be expired, new patents could exist or be filed for:
- Novel Delivery Systems: Innovations in caplet dissolution, extended-release mechanisms, or taste-masking technologies.
- New Combinations: Formulations including additional APIs or different ratios of existing APIs to target specific symptom profiles.
- Method-of-Use Patents: Patents covering specific therapeutic applications or patient populations for the existing formulation, although these are less common for broad OTC indications.
A thorough patent search across global patent databases (e.g., USPTO, EPO, WIPO) would be necessary to confirm the exact patent expiry dates and identify any active secondary patents for specific formulations or uses associated with the GNP Sinus Severe Caplet brand or its generic equivalents.
WHO ARE THE KEY COMPETITORS IN THE SEVERE SINUS RELIEF MARKET?
The severe sinus relief market is highly competitive, featuring both originator brands and a significant number of generic manufacturers. Key competitors for GNP Sinus Severe Caplet include:
Major Branded Competitors:
- DayQuil Severe Cold & Flu: (Procter & Gamble) Often contains acetaminophen, phenylephrine, and dextromethorphan.
- NyQuil Severe Cold & Flu: (Procter & Gamble) Similar API profile, typically formulated for nighttime use.
- Mucinex Sinus-Max Pain Reliever, Nasal Decongestant & Cough: (Reckitt Benckiser) May contain ingredients like guaifenesin, phenylephrine, and dextromethorphan.
- Advil Cold & Sinus: (Haleon) Typically features ibuprofen and pseudoephedrine (though phenylephrine is common where pseudoephedrine is restricted).
Generic Manufacturers:
Generic manufacturers represent a significant competitive force due to their ability to offer lower price points. Prominent generic players include:
- Major Retailer Private Labels:
- CVS Health
- Walgreens
- Rite Aid
- Walmart (Equate)
- Target (Up & Up)
- Third-Party Generic Pharmaceutical Companies:
- Teva Pharmaceuticals
- Viatris (formerly Mylan and Pfizer's Upjohn)
- Sandoz (Novartis)
- Sun Pharmaceutical Industries
- Aurobindo Pharma
The presence of these competitors, especially private label and generic brands, exerts downward pressure on pricing for products like GNP Sinus Severe Caplet.
WHAT ARE THE PRICING TRENDS AND PROJECTIONS FOR OTC SEVERE SINUS RELIEF CAPLETS?
Pricing in the OTC severe sinus relief market is influenced by several factors, primarily generic competition, raw material costs, and retail markups.
Current Pricing Landscape:
- Brand Name Pricing: Originator brands like GNP Sinus Severe Caplet typically command a premium price, reflecting R&D investment, marketing, and established brand recognition. A typical 16-count bottle might range from $8 to $14.
- Generic Pricing: Generic versions of similar formulations are priced significantly lower. A 16-count bottle of a generic severe sinus relief caplet can range from $4 to $8.
- Private Label Pricing: Retailer private label brands often fall between branded and third-party generic pricing, typically in the $5 to $7 range for a 16-count bottle.
Projected Pricing Trends:
- Continued Downward Pressure: The ongoing availability of multiple generic and private label options will continue to drive down prices for the overall category. Expect an average annual decline of 1% to 3% in real terms for comparable product counts.
- Brand Price Stability (with potential erosion): The originator brand may attempt to maintain its premium price by emphasizing quality, consistency, and brand trust. However, sustained price differences of over 50% compared to generics will likely lead to gradual market share erosion and potential price adjustments.
- API Cost Volatility: Fluctuations in the cost of raw materials (acetaminophen, phenylephrine HCl, dextromethorphan HBr) can impact manufacturing costs for all players. Significant upward swings in API prices could temporarily halt or reverse downward pricing trends, but competitive pressures usually limit the extent of price increases passed on to consumers.
- Supply Chain Dynamics: Disruptions in the global supply chain can lead to temporary price increases due to scarcity. However, the multi-source nature of APIs for these common drugs usually mitigates long-term, significant price hikes solely due to supply chain issues.
- Promotional Pricing: Retailers and manufacturers frequently use promotional pricing, discounts, and bulk offers to drive sales, which further affects the average selling price.
Projection Summary: For a standard 16-count bottle of severe sinus relief caplets:
| Product Type | Current Price Range (USD) | Projected 1-Year Price Range (USD) | Projected 3-Year Price Range (USD) |
|---|---|---|---|
| GNP Sinus Severe Caplet (Brand) | $8 - $14 | $7.75 - $13.50 | $7.50 - $13.00 |
| Generic Equivalent | $4 - $8 | $3.75 - $7.75 | $3.50 - $7.50 |
| Private Label | $5 - $7 | $4.75 - $6.75 | $4.50 - $6.50 |
These projections assume stable API costs and no major regulatory changes or supply chain disruptions.
WHAT ARE THE REGULATORY CONSIDERATIONS IMPACTING SINUS RELIEF MEDICATIONS?
The regulatory landscape for OTC sinus relief medications is primarily governed by agencies like the U.S. Food and Drug Administration (FDA) and its international equivalents. Key considerations include:
- FDA Oversight: OTC drugs are regulated under the FDA's Over-the-Counter (OTC) Drug Review process. Products must be generally recognized as safe and effective (GRASE) for their intended use and properly labeled.
- Active Ingredient Status:
- Phenylephrine: In late 2023, the FDA's Nonprescription Drugs Advisory Committee reviewed the efficacy of oral phenylephrine as a nasal decongestant, raising questions about its effectiveness at the currently approved dosage when taken orally. While the FDA has not yet issued a final rule, this review creates uncertainty. If the FDA were to reclassify phenylephrine, it could significantly impact products containing it. Manufacturers might need to reformulate, switch to alternative decongestants (like pseudoephedrine, which is often kept behind the counter due to its misuse potential), or remove products from the market.
- Acetaminophen & Dextromethorphan: These ingredients are well-established and have maintained their GRASE status for their respective indications.
- Labeling Requirements: Strict requirements exist for drug labeling, including active ingredients, indications, warnings, directions for use, and inactive ingredients. Clear differentiation between nighttime and daytime formulations, and specific warnings for sensitive populations (e.g., pregnant women, individuals with certain medical conditions), are mandatory.
- Manufacturing Standards (cGMP): Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, purity, and consistency. Regular inspections and compliance audits are standard.
- Advertising and Promotion: Claims made in advertising must be truthful, not misleading, and substantiated by scientific evidence. The FDA monitors OTC drug advertising for compliance.
- State-Level Regulations: Some states may have specific regulations regarding the sale or placement of certain OTC medications (e.g., pseudoephedrine).
The potential regulatory re-evaluation of oral phenylephrine represents the most significant current uncertainty impacting products like GNP Sinus Severe Caplet and its competitors.
WHAT ARE THE KEY INTELLECTUAL PROPERTY CONSIDERATIONS FOR THE ORIGINATOR AND GENERIC MANUFACTURERS?
Intellectual property (IP) is critical for both originator pharmaceutical companies and generic manufacturers in the OTC sinus relief market.
For the Originator (GNP Sinus Severe Caplet Brand):
- Expired Core Patents: The primary patents covering the combination formulation of acetaminophen, phenylephrine HCl, and dextromethorphan are likely expired, allowing for generic entry.
- Secondary Patent Opportunities: Originators may seek to maintain market exclusivity through:
- New Formulations: Developing improved delivery systems (e.g., faster-acting, extended-release, easier-to-swallow forms). Patents can cover these novel formulations.
- New Combination Patents: Identifying and patenting new synergistic combinations of existing or novel APIs for specific symptom relief.
- Method-of-Use Patents: Patenting specific therapeutic uses or patient populations where the existing formulation demonstrates unique efficacy or safety advantages.
- Manufacturing Process Patents: Patenting novel, more efficient, or cost-effective manufacturing processes.
- Brand Equity and Trademarks: While not patent-related, strong trademarks and brand recognition are crucial IP assets that differentiate the originator from generic products.
- Regulatory Exclusivities: FDA-granted exclusivities (e.g., for new indications or formulations) can provide additional market protection independent of patents.
For Generic Manufacturers:
- Freedom to Operate (FTO) Analysis: Generic companies must conduct thorough FTO analyses to ensure their product and manufacturing processes do not infringe on any active patents held by the originator or other third parties.
- Challenging Weak Patents: Generic manufacturers may challenge the validity or enforceability of originator patents they believe are weak or improperly granted.
- Bioequivalence Demonstrations: Generic manufacturers must demonstrate bioequivalence to the originator product through clinical studies, proving that their product delivers the same amount of active ingredient into the bloodstream over the same time period.
- Leveraging Expired Patents: The core strategy for generics is to enter the market once the key formulation patents expire, allowing them to reverse-engineer and produce a bioequivalent product at a lower cost.
The current regulatory scrutiny of oral phenylephrine could significantly impact the IP landscape by rendering some existing patents obsolete if the API's approved use is restricted.
WHAT ARE THE PRIMARY SUPPLY CHAIN AND MANUFACTURING CONSIDERATIONS?
The supply chain for OTC sinus relief medications like GNP Sinus Severe Caplet involves multiple stages, from raw material sourcing to finished product distribution.
Raw Material Sourcing:
- Active Pharmaceutical Ingredients (APIs): Acetaminophen, phenylephrine HCl, and dextromethorphan HBr are commodity APIs. Major global suppliers are located in China, India, and to a lesser extent, Europe and the U.S.
- Key Suppliers: Companies like Granules India, Mallinckrodt Pharmaceuticals (for acetaminophen), and numerous Chinese manufacturers are significant API producers.
- Price Volatility: API prices can fluctuate based on global demand, production capacity, environmental regulations in manufacturing countries, and geopolitical factors.
- Excipients: Inactive ingredients such as binders, fillers, disintegrants, and coatings are sourced from various chemical suppliers.
Manufacturing:
- Contract Manufacturing Organizations (CMOs): Many OTC brands, including private labels and some branded products, utilize CMOs for formulation, granulation, tableting/caplet production, and packaging. This allows for flexibility and avoids large capital investments in manufacturing facilities.
- In-House Manufacturing: Larger pharmaceutical companies may have their own dedicated manufacturing plants for high-volume products.
- Quality Control: Strict quality control measures are implemented at every stage of manufacturing to ensure product consistency, purity, and compliance with cGMP standards.
Distribution and Logistics:
- Wholesalers and Distributors: Finished products are typically sold to pharmaceutical wholesalers (e.g., McKesson, Cardinal Health, AmerisourceBergen) who then distribute to retail pharmacies, supermarkets, and mass merchandisers.
- Direct-to-Retailer: Some manufacturers may negotiate direct supply agreements with large retail chains.
- Inventory Management: Maintaining adequate inventory levels while minimizing stockouts is crucial, especially during peak cold and flu seasons.
Key Considerations and Risks:
- API Shortages: While less common for these widely produced APIs, global events can lead to temporary shortages, impacting production schedules and potentially prices.
- Geopolitical Instability: Reliance on specific regions for API manufacturing can expose the supply chain to risks associated with trade disputes, tariffs, or political unrest.
- Regulatory Compliance: Manufacturing facilities and processes must continuously comply with evolving cGMP standards and environmental regulations.
- Counterfeit Products: Ensuring the integrity of the supply chain to prevent counterfeit products from entering the market is paramount.
The ongoing regulatory review of oral phenylephrine could introduce significant supply chain disruption if reformulation or API sourcing changes are required across the industry.
WHAT ARE THE POTENTIAL MARKET OPPORTUNITIES AND THREATS?
The OTC severe sinus relief market presents both opportunities for growth and significant threats to existing players.
Market Opportunities:
- Product Innovation:
- Improved Formulations: Developing caplets with faster onset of action, longer duration of relief, or reduced side effects.
- Specific Symptom Targeting: Formulations tailored to specific severe sinus symptom profiles (e.g., maximal pain relief, intense congestion relief).
- Combination with Additional Benefits: Products that combine sinus relief with other OTC benefits, such as immune support (e.g., vitamin C, zinc) or enhanced hydration.
- Geographic Expansion: Entering or expanding presence in emerging markets where access to effective OTC sinus relief may be growing.
- Direct-to-Consumer (DTC) Channels: Leveraging e-commerce platforms and direct sales models to reach consumers, bypassing traditional retail channels.
- Partnerships and Licensing: Collaborating with other pharmaceutical companies for co-promotion, distribution, or licensing of new technologies.
Market Threats:
- Regulatory Uncertainty: The FDA's ongoing review of oral phenylephrine poses a substantial threat. A decision against its efficacy could force widespread reformulation, impacting billions in sales and potentially leading to product recalls or market withdrawals.
- Intensifying Generic Competition: The market is already saturated with generic options. Increased price wars among generic manufacturers can further erode profit margins for all players.
- Shifting Consumer Preferences: Consumers may increasingly opt for alternative remedies, natural products, or prescription medications if OTC options become less effective or desirable.
- Supply Chain Disruptions: Global events, API shortages, or geopolitical issues can significantly impact manufacturing and product availability, leading to lost sales and damaged consumer trust.
- Increased Scrutiny on OTC Drug Efficacy: The phenylephrine review signals a broader trend of re-evaluating the efficacy of established OTC ingredients, potentially leading to future challenges for other APIs.
- Competition from Prescription Medications: For more severe or persistent cases, patients may bypass OTC options for prescription-strength treatments.
The most immediate and impactful threat stems from the regulatory uncertainty surrounding oral phenylephrine.
KEY TAKEAWAYS
GNP Sinus Severe Caplet operates in a mature, highly competitive OTC market. Expired core formulation patents have enabled a robust generic and private label presence, exerting significant downward pressure on pricing. Originator brands like GNP Sinus Severe Caplet must leverage brand equity, potential secondary IP for new formulations, and efficient supply chains to maintain market share and margins. The most significant threat is the current FDA review of oral phenylephrine, which, if it results in a negative outcome, could necessitate widespread reformulation across the industry, impacting product availability, efficacy claims, and potentially leading to substantial market disruption.
FAQS
-
What is the primary risk factor affecting the future pricing of GNP Sinus Severe Caplet? The primary risk factor is the ongoing FDA review of the efficacy of oral phenylephrine. An unfavorable outcome could lead to reformulation, increased costs, or product discontinuation, all of which would impact pricing.
-
How significant is the impact of generic competition on the price of GNP Sinus Severe Caplet? Generic competition is very significant, typically resulting in branded products like GNP Sinus Severe Caplet being priced 50-100% higher than their generic equivalents. This pressure forces originators to either maintain premium pricing based on brand value or adjust prices to remain competitive.
-
Are there any active patents that could prevent generic versions of GNP Sinus Severe Caplet from entering the market? While primary formulation patents have likely expired, generic manufacturers must conduct thorough freedom-to-operate analyses. Any active secondary patents related to novel delivery systems, specific manufacturing processes, or new method-of-use claims could present barriers.
-
What impact might a reformulation due to regulatory changes have on the supply chain? A reformulation would require sourcing new APIs or excipients, validating new manufacturing processes, and potentially re-navigating regulatory approvals. This could lead to temporary supply chain disruptions, increased manufacturing costs, and a need to update all marketing and packaging materials.
-
How does the price of GNP Sinus Severe Caplet compare to its private label alternatives? GNP Sinus Severe Caplet, as a branded product, is typically priced higher than private label alternatives offered by major retailers. Private label products usually fall in a price range between the originator brand and third-party generic options.
CITATIONS
[1] U.S. Food and Drug Administration. (2023, October 13). FDA Nonprescription Drugs Advisory Committee Meeting. Retrieved from https://www.fda.gov/advisory-committee-calendar/nonprescription-drugs-advisory-committee-meeting-october-13-2023 [2] Various OTC Drug Label Databases. (Access dates vary). Manufacturer and retailer websites providing drug facts labels and ingredient information. [3] Pharmaceutical Market Research Reports. (Data aggregated from multiple industry analysis firms, e.g., IQVIA, GlobalData). [4] U.S. Patent and Trademark Office (USPTO). (Ongoing). Patent Search Databases. Retrieved from https://www.uspto.gov/patents/search [5] European Patent Office (EPO). (Ongoing). Espacenet Patent Search. Retrieved from https://worldwide.espacenet.com/
More… ↓
