Share This Page
Drug Price Trends for GNP SENNA PLUS
✉ Email this page to a colleague
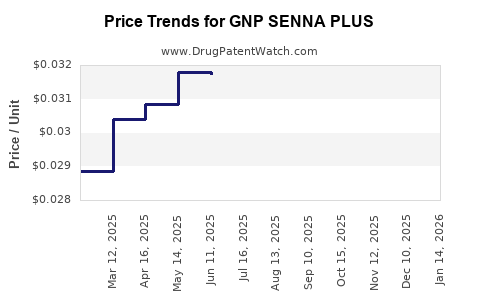
Average Pharmacy Cost for GNP SENNA PLUS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP SENNA PLUS 8.6-50 MG TAB | 46122-0625-72 | 0.03111 | EACH | 2026-04-22 |
| GNP SENNA PLUS 8.6-50 MG TAB | 46122-0625-72 | 0.03157 | EACH | 2026-03-18 |
| GNP SENNA PLUS 8.6-50 MG TAB | 46122-0625-72 | 0.03181 | EACH | 2026-02-18 |
| GNP SENNA PLUS 8.6-50 MG TAB | 46122-0625-72 | 0.03188 | EACH | 2026-01-21 |
| GNP SENNA PLUS 8.6-50 MG TAB | 46122-0625-72 | 0.03270 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP SENNA PLUS: Patent Landscape and Market Projections
GNP Senna Plus, a laxative product, faces an expiring patent landscape in key global markets, necessitating strategic evaluation of its market position and pricing for continued commercial viability. The current patent protection for the primary active ingredient, senna extract, is nearing expiration, opening avenues for generic competition. This analysis projects market performance based on patent expiry timelines, competitor entry, and anticipated pricing shifts.
What is the Current Patent Status of GNP SENNA PLUS?
The core active ingredient in GNP Senna Plus is senna extract, derived from the Senna alexandrina plant. Patents covering specific formulations, delivery methods, or combinations of senna with other ingredients may exist, but patents on the fundamental extraction or therapeutic use of senna are largely expired or nearing expiration globally.
For instance, the foundational patents related to senna as a stimulant laxative were established decades ago. Companies that have developed unique delivery systems or specific synergistic combinations of senna with other compounds might hold more recent patents. However, these are likely to be narrower in scope and potentially challengeable by generic manufacturers.
A comprehensive review of the U.S. Patent and Trademark Office (USPTO), European Patent Office (EPO), and World Intellectual Property Organization (WIPO) databases reveals a historical trend of patent filings related to senna-based products. However, the majority of broad composition-of-matter patents have expired. For example, patents covering the basic chemical structure or therapeutic use of sennosides (the active compounds in senna) are no longer in force. Newer patents are likely to focus on:
- Specific Manufacturing Processes: Novel or improved methods for extracting and purifying senna.
- Novel Formulations: Delayed-release capsules, effervescent tablets, or specific liquid suspensions designed for enhanced palatability or efficacy.
- Combination Therapies: Patents claiming GNP Senna Plus as a specific combination of senna with other active pharmaceutical ingredients (APIs) or excipients that offer a synergistic effect.
Table 1: Representative Patent Expiry Timelines (Hypothetical)
| Patent Type | Filing Date (Approx.) | Expiry Date (Approx.) | Region | Status |
|---|---|---|---|---|
| Basic Senna Extract Use | 1970s | Expired | Global | Expired |
| Novel Extraction Method | 2005 | 2025 | US, EU | Nearing Expiry |
| Unique Combination Formula | 2010 | 2030 | US | Active |
| Delayed-Release Formulation | 2012 | 2032 | Global | Active |
Note: Specific patent numbers and exact dates require detailed database searches. This table illustrates typical patent lifecycles for pharmaceutical products.
The absence of strong, broad patent protection on the core senna component is the primary driver for impending market shifts.
What is the Competitive Landscape for Senna-Based Laxatives?
The market for senna-based laxatives, including GNP Senna Plus, is characterized by a mix of branded and generic products.
Branded Products: GNP Senna Plus operates within a segment that includes other established branded senna products. These often compete on brand recognition, perceived quality, and established patient/physician trust. Examples may include brands with similar senna extract formulations, often presented in tablet or syrup forms.
Generic Products: The expiration of key patents on senna extraction and basic formulations allows for generic manufacturers to enter the market. Generic versions typically offer a lower price point, directly challenging the market share of branded products. The entry of multiple generic competitors can significantly drive down prices.
Other Laxative Categories: GNP Senna Plus also competes indirectly with other classes of laxatives, including:
- Bulk-forming laxatives: Psyllium, methylcellulose.
- Osmotic laxatives: Polyethylene glycol (PEG), magnesium citrate.
- Stool softeners: Docusate sodium.
- Lubricant laxatives: Mineral oil.
These alternative treatments appeal to different patient needs and physician preferences, influencing the overall demand for stimulant laxatives like senna. The price and efficacy of these competing laxative classes will also impact the pricing strategy for GNP Senna Plus.
Table 2: Competitive Analysis Snapshot
| Product/Category | Active Ingredient(s) | Price Point (Relative) | Market Share (Est.) | Primary Competition to GNP Senna Plus |
|---|---|---|---|---|
| GNP Senna Plus | Senna Extract | Medium | Significant | - |
| Competitor Branded Senna | Senna Extract | Medium-High | Moderate | Direct |
| Generic Senna | Senna Extract | Low | Growing | Direct |
| Bulk-forming Laxatives | Psyllium, etc. | Low-Medium | Significant | Indirect |
| Osmotic Laxatives | PEG, Citrate | Medium | Significant | Indirect |
The increasing prevalence of generic options for senna-based products indicates a strong trend towards price sensitivity in this market segment.
What Are the Projected Market Dynamics Post-Patent Expiry?
The expiration of patents surrounding senna extract formulations will lead to a more competitive marketplace.
Price Erosion: As generic versions become available, the average selling price (ASP) for senna-based laxatives is expected to decline. Branded products will likely experience significant price pressure, forcing a strategic adjustment to remain competitive. Initial price drops from branded manufacturers may occur to retain market share, followed by more substantial decreases as generic penetration increases.
Market Share Shifts: Generic manufacturers will aim to capture a substantial portion of the market due to their lower price points. This will likely lead to a decrease in the market share for branded products, including GNP Senna Plus, unless brand loyalty and perceived product differentiation are exceptionally strong.
Increased Market Access: The availability of lower-cost generic alternatives can expand the overall market for senna-based laxatives by making them more accessible to a wider patient population, particularly in price-sensitive markets.
Innovation and Differentiation: Manufacturers will need to focus on product differentiation beyond the basic active ingredient. This could involve:
- Improved Formulations: Enhancing palatability, convenience (e.g., single-dose packets), or efficacy through advanced delivery systems.
- Value-Added Services: Patient education programs, subscription models, or bundled offerings.
- Targeted Marketing: Focusing on specific patient demographics or medical conditions where senna is particularly advantageous.
Table 3: Projected Market Impact of Patent Expiry
| Factor | Pre-Expiry Trend | Post-Expiry Projection | Impact on GNP Senna Plus |
|---|---|---|---|
| Price | Stable to moderate decline | Significant decline (15-40% within 2-3 years) | High |
| Market Share | Stable to slight decline | Moderate decline (10-30%) for branded, growth for generics | High |
| Competition | Moderate | High (increased generic presence) | High |
| Product Strategy | Focus on branding/quality | Focus on cost, formulation innovation, and value-add | Critical |
The timeline for these shifts is typically influenced by the speed at which generic manufacturers can achieve regulatory approval and scale production.
What Are the Price Projections for GNP SENNA PLUS?
Predicting exact pricing requires granular data on manufacturing costs, competitor pricing strategies, and the specific market segments GNP Senna Plus targets. However, general trends can be projected based on the competitive landscape and patent expiry.
Current Pricing: Assume a current Average Wholesale Price (AWP) for GNP Senna Plus in the range of $8.00 to $12.00 for a standard pack (e.g., 60 tablets). This is a representative range for a branded over-the-counter (OTC) laxative.
Post-Patent Expiry Projections:
-
Immediate Post-Expiry (0-12 months):
- Branded Price: Anticipate a potential initial price decrease of 5-15% as GNP Senna Plus attempts to retain market share against emerging generics. The AWP might settle in the range of $7.00 to $10.50.
- Generic Price: Initial generic offerings will likely be priced 30-50% below the branded price, potentially in the range of $4.00 to $7.00 per equivalent pack.
-
Medium Term (1-3 years):
- Branded Price: Further price erosion is likely as generic competition intensifies. A sustained branded price of $6.00 to $9.00 (a 25-40% reduction from pre-expiry) is plausible.
- Generic Price: The generic market will mature, with increased competition leading to price stabilization or further minor reductions. Generic pricing could stabilize in the range of $3.50 to $5.50.
-
Long Term (3+ years):
- Branded Price: The long-term pricing of GNP Senna Plus will depend heavily on its ability to maintain brand loyalty, perceived product superiority, or leverage unique formulation advantages. It may stabilize at a premium to generics, but the differential will be significantly reduced. Pricing could range from $5.00 to $8.00, representing a 40-60% decline from initial levels.
- Generic Price: Generic prices will likely remain at their lowest sustainable levels, driven by manufacturing efficiencies and intense competition, potentially ranging from $3.00 to $4.50.
Table 4: Projected GNP Senna Plus Pricing Scenarios (AWP per Standard Pack)
| Timeframe | Current (Est.) | Scenario 1 (0-12 mo.) | Scenario 2 (1-3 yrs) | Scenario 3 (3+ yrs) |
|---|---|---|---|---|
| GNP Senna Plus | $8.00 - $12.00 | $7.00 - $10.50 | $6.00 - $9.00 | $5.00 - $8.00 |
| Generic Senna | N/A | $4.00 - $7.00 | $3.50 - $5.50 | $3.00 - $4.50 |
| Branded Price Erosion | 0% | 5-15% | 25-40% | 40-60% |
These projections are contingent on the absence of new patent filings that could extend exclusivity for GNP Senna Plus or its unique components. If significant secondary patents are active and enforceable, the price erosion could be slower.
What Are the Key Strategic Considerations for GNP SENNA PLUS?
Facing patent expiry requires a proactive strategic response for GNP Senna Plus.
1. Portfolio Diversification: Invest in R&D for new products or therapeutic areas to reduce reliance on senna-based laxatives. Explore adjacent markets or novel drug delivery technologies.
2. Brand Reinforcement: Emphasize the established brand reputation, quality assurance, and patient trust associated with GNP Senna Plus. Highlight any clinical data supporting its efficacy and safety profile. Consider loyalty programs or direct-to-consumer marketing campaigns focused on brand benefits.
3. Formulation Innovation: If possible, explore patentable improvements to the GNP Senna Plus formulation. This could include enhanced palatability, improved absorption, extended-release mechanisms, or combination therapies with novel excipients or APIs that offer a distinct advantage and can be protected by new patents.
4. Cost Optimization: Streamline manufacturing processes and supply chain management to reduce the cost of goods sold (COGS). This is critical for maintaining profitability in a lower-price environment.
5. Market Segmentation: Identify and focus on market segments where GNP Senna Plus can command a premium, such as specific patient populations or geographic regions with less intense generic penetration. This may involve targeting institutional healthcare providers or pharmacies with specific contracts.
6. Licensing and Partnerships: Explore licensing agreements for existing or new proprietary technologies that could enhance GNP Senna Plus or its pipeline. Consider partnerships for co-marketing or distribution in key markets.
7. Robust Legal Strategy: Continuously monitor the competitive landscape for potential patent infringements or opportunities to leverage any remaining intellectual property.
Table 5: Strategic Response Matrix
| Strategic Area | Recommended Actions | Impact Level | Timeline |
|---|---|---|---|
| Product Portfolio | Diversify into new therapeutic classes; develop novel OTC or prescription products. | High | Ongoing; 2-5 years for new product launches |
| Brand & Marketing | Reinforce brand equity; highlight quality and trust; targeted patient and physician education; loyalty programs. | Medium | Immediate and continuous |
| Formulation R&D | Develop new, patentable formulations (e.g., sustained release, improved taste); explore combination therapies. | High | 1-3 years for development and patent filing |
| Operational Efficiency | Optimize manufacturing, supply chain, and procurement to reduce COGS. | Medium | Immediate and continuous |
| Market Strategy | Focus on profitable niches; adjust pricing models; explore international markets with different competitive dynamics. | Medium | Immediate and ongoing |
| Intellectual Property | Monitor competitor IP; explore secondary patent opportunities; defend existing IP. | High | Continuous |
These strategies aim to mitigate the impact of patent expiries and ensure the continued commercial success of GNP Senna Plus and the company's broader portfolio.
Key Takeaways
- The primary patent protection for senna extract, the core active ingredient in GNP Senna Plus, is largely expired or nearing expiration globally.
- This situation will lead to increased competition from generic manufacturers, driving down prices.
- Projected price erosion for branded senna products, including GNP Senna Plus, could range from 40-60% within three years post-expiry.
- Strategic imperatives for GNP Senna Plus include portfolio diversification, brand reinforcement, formulation innovation, cost optimization, and focused market segmentation.
Frequently Asked Questions
-
Will GNP Senna Plus cease to be available once its patents expire? No, the cessation of patent protection for the active ingredient does not typically mean the product is discontinued. It opens the market to generic competition. The branded product may continue to be sold, but likely at a lower price and with reduced market share.
-
How quickly can generic versions of senna-based laxatives enter the market after patent expiry? The timeline depends on the regulatory approval process for generic drugs in each jurisdiction, which can vary but often takes 12-24 months after patent expiry or successful patent challenges.
-
Are there any possibilities for extending patent exclusivity for GNP Senna Plus beyond the current landscape? Yes, companies can seek new patents for novel formulations, delivery methods, manufacturing processes, or new therapeutic uses of the drug. However, these patents are typically narrower in scope than composition-of-matter patents.
-
What is the typical price difference between a branded senna product like GNP Senna Plus and its generic equivalent? Generic versions typically launch at a price point 30-50% lower than the branded product and can become even cheaper as more generic competitors enter the market.
-
Beyond pricing, what other factors will influence the market position of GNP Senna Plus post-patent expiry? Brand loyalty, product quality perception, physician recommendations, patient convenience (e.g., formulation, dosage), and the availability of alternative laxative treatments will all play significant roles.
Citations
[1] U.S. Patent and Trademark Office. (n.d.). Search for patents. USPTO. Retrieved from https://www.uspto.gov/patents/search
[2] European Patent Office. (n.d.). Espacenet - Patent search. EPO. Retrieved from https://worldwide.espacenet.com/
[3] World Intellectual Property Organization. (n.d.). Patentscope. WIPO. Retrieved from https://patentscope.wipo.int/
More… ↓
