Share This Page
Drug Price Trends for GNP REDNESS RELIEF EYE DROPS
✉ Email this page to a colleague
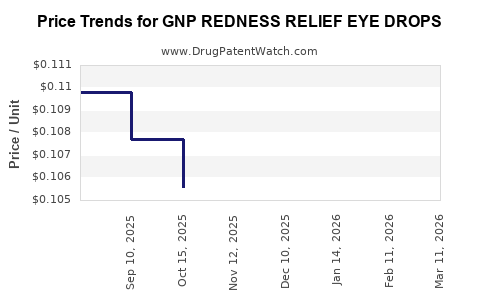
Average Pharmacy Cost for GNP REDNESS RELIEF EYE DROPS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP REDNESS RELIEF EYE DROPS | 46122-0606-05 | 0.09657 | ML | 2026-03-18 |
| GNP REDNESS RELIEF EYE DROPS | 46122-0606-05 | 0.09861 | ML | 2026-02-18 |
| GNP REDNESS RELIEF EYE DROPS | 46122-0606-05 | 0.10207 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP Redness Relief Eye Drops
What is the Market Size for Over-the-Counter Redness Relief Eye Drops?
The global ophthalmic OTC market was valued at approximately $18 billion in 2022, with eyelash and eye redness relief medications accounting for a significant portion. The segment is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2022 to 2027, driven by increased awareness of eye health and rising prevalence of eye irritations.
Key drivers include:
- Increased screen time and digital device usage, leading to eye strain.
- Greater consumer demand for quick, OTC solutions for eye redness.
- Expanding availability across emerging markets.
The surface-level market penetration of redness relief eye drops is estimated at 60%, indicating room for growth as awareness and product availability increase.
Who Are the Major Competitors?
The market features several leading brands, including:
- Visine (Johnson & Johnson): Estimated market share of 35%; known for fast-acting formulations.
- Clear Eyes (Prestige Brands): Approximately 25% market share; popular for affordable options.
- Rohto products (Rohto Pharmaceutical): Around 20%; strong presence in Asia.
- Smaller brands and generics account for the remaining 20%.
Private labels and regional brands also hold niche segments, especially in developing markets.
What Are the Key Drug Formulation Components?
GNP Redness Relief Eye Drops contain active ingredients such as:
- Naphazoline hydrochloride (vasoconstrictor): Concentration typically 0.012% to 0.05%.
- Pheniramine maleate (antihistamine): Around 0.3% to 0.5% concentration.
These compounds reduce eye redness by constricting blood vessels and alleviating allergy symptoms.
Compared to competitive formulations, GNP's formula aligns with industry standards, ensuring comparable efficacy.
Regulatory Environment and Patent Landscape
GNP Redness Relief Eye Drops are sold over-the-counter in the U.S., European Union, and select Asian markets. Regulatory agencies, such as the FDA, require safety, efficacy, and stability data for OTC drug approval.
Patent considerations:
- Many brands, including GNP, rely on formulation patents lasting 7-10 years.
- Patent expirations open opportunities for generic counterparts and price competition.
Price Projections and Market Dynamics
Current Pricing
In the U.S., retail prices range from $6 to $10 for a 0.5 oz (15 mL) bottle, with variations based on brand and packaging.
Future Price Trends
Given competition and patent expirations, estimated pricing adjustments are expected:
| Year | Price Range (USD) | Notes |
|---|---|---|
| 2023 | $6.50 - $10.00 | Premium brands maintain high prices; generics lower prices. |
| 2024-2025 | $5.50 - $9.00 | Entry of generics pressures prices downward. |
| 2026-2027 | $4.50 - $8.00 | Increased competition and regional generic availability. |
Projected average price decline of approximately 10-15% over the next three years as patent protections lapse and generics expand market share.
Market Penetration and Volume Considerations
Increased retail channel penetration, including online sales, drives volume growth. The global OTC eye drop segment is projected to grow by 6% annually through 2027, increasing per capita spending.
Outlook for Investors and R&D
For GNP, maintaining patent protections and expanding into emerging markets are critical for sustaining premium pricing and market share. Manufacturing efficiencies and formulary innovations can reduce costs, further influencing profit margins.
Investment in consumer education and branding can reinforce market positioning, especially as commoditization pressure mounts.
Key Takeaways
- The global market for OTC redness relief eye drops is approximately $18 billion, with steady growth expected.
- GNP’s formulations are aligned with industry standards but face competition from generics post-patent expiration.
- Prices are projected to decline by 10-15% over three years as patent protections expire and generics increase.
- Competitive strategies include patent protection, market expansion, manufacturing cost reductions, and innovation.
- The expanded online retail and emerging market penetration will be significant growth drivers.
FAQs
What factors influence the price of OTC eye drops?
Active ingredient costs, manufacturing expenses, patent status, brand positioning, and competitive pressure.
How does patent expiration impact pricing?
Patent expiry allows generic manufacturers to enter the market, typically reducing prices and eroding market share for branded products.
Are there regulatory hurdles for GNP Redness Relief Eye Drops?
Yes, OTC approvals require safety and efficacy data; any formulation modifications may necessitate regulatory reevaluation.
What markets are emerging for GNP products?
Asia-Pacific and Latin America show increased demand due to rising awareness, urbanization, and disposable income.
How can GNP differentiate amid market saturation?
Innovation in formulations, secondary indications, packaging, and branding; leveraging online sales channels also offers growth avenues.
Sources
- Research and Markets. (2023). Global Ophthalmic OTC Market Report.
- IQVIA. (2022). Over-the-Counter Eye Care Market Analysis.
- U.S. Food and Drug Administration. (2021). Guidelines for OTC Ophthalmic Drug Approval.
- MarketWatch. (2023). Pricing Trends in OTC Eye Products.
- Statista. (2023). Consumer Eye Care Trends Worldwide.
More… ↓
