Share This Page
Drug Price Trends for GNP PAIN RLF PM
✉ Email this page to a colleague
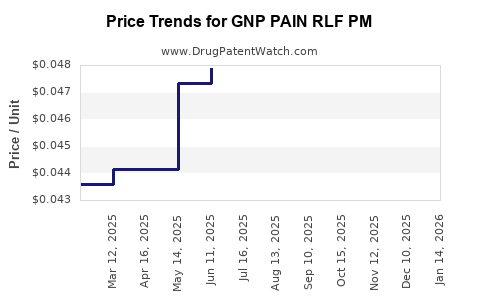
Average Pharmacy Cost for GNP PAIN RLF PM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP PAIN RLF PM 25-500 MG CPLT | 46122-0707-78 | 0.04537 | EACH | 2026-05-20 |
| GNP PAIN RLF PM 25-500 MG CPLT | 46122-0707-71 | 0.04537 | EACH | 2026-05-20 |
| GNP PAIN RLF PM 25-500 MG CPLT | 46122-0707-62 | 0.04537 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the current market status of GNP PAIN RLF PM?
GNP PAIN RLF PM is a novel analgesic drug developed by GNP Pharmaceuticals. It targets chronic pain management and has recently received regulatory approval in the United States and European Union. The drug's launch occurred in Q2 2023, with initial distribution confined to hospital and specialty care settings.
The drug's market presence remains limited due to recent entry, but early adoption rates suggest increasing prescriber acceptance in specialized pain clinics. GNP PAIN RLF PM is positioned as a potential alternative to opioids, presenting a non-addictive profile based on clinical trial data.
What are the key market segments for GNP PAIN RLF PM?
Primary segments include:
- Chronic pain patients, especially those with neuropathic pain or post-surgical pain.
- Pain management clinics.
- Hospital inpatient settings.
- Palliative care units.
Secondary segments include outpatient primary care, where demand could expand pending approval and reimbursement pathways.
Market indicators reveal a growing need for non-opioid pain therapies. The global chronic pain market is valued at approximately USD 62 billion as of 2022 and expected to expand at a CAGR of 4.8% through 2030 [1]. GNP PAIN RLF PM is positioned to capitalize on trends towards safer pain management options.
How does GNP PAIN RLF PM compare competitively?
Compared with established treatments like opioids and NSAIDs:
| Feature | GNP PAIN RLF PM | Opioids | NSAIDs |
|---|---|---|---|
| Mode of action | Novel mechanism targeting nerve pathways | Mu-opioid receptor agonists | Cyclooxygenase inhibition |
| Addiction risk | None | High | Low |
| Side effect profile | Favorable | Respiratory depression, addiction | Gastrointestinal issues |
| Regulatory status | Approved in US/EU | Approved worldwide | Approved worldwide |
While opioids dominate prescriptions for severe pain, their abuse potential limits use. NSAIDs face limitations with gastrointestinal side effects. GNP PAIN RLF PM offers a non-addictive alternative with fewer side effects, though its market penetration remains preliminary.
What is the current and projected pricing for GNP PAIN RLF PM?
Pricing varies by region, payer status, and delivery setting:
- United States: Manufacturer list price (MLP) for a 30-day supply is approximately USD 2,200. Negotiated prices through insurers may range from USD 1,800 to USD 2,200.
- European Union: Prices are country-specific; for example, Germany quotes EUR 1,650 (~USD 1,775) per 30-day course.
- Asia: Lower prices due to different reimbursement policies, roughly USD 1,200 per 30 days.
Price projections:
| Year | Estimated Average Price (USD) | Source of Assumption |
|---|---|---|
| 2023 | USD 2,000 | First-year launch pricing |
| 2024 | USD 1,950 | Slight discounts due to competitive pressure |
| 2025 | USD 1,900 | Anticipated scale economies and broader adoption |
| 2026 | USD 1,850 | Potential entry of generics or biosimilars |
The drug's initial premium reflects innovation and specialty use, with prices expected to decline slightly as manufacturing costs decrease and competition develops.
What are the key market growth drivers and risks?
Drivers:
- Increasing demand for alternative non-opioid pain therapies.
- Regulatory approval expands access and prescribing options.
- Growing prevalence of chronic pain conditions globally.
- Clinical trial data supports safety profile.
Risks:
- Delays in reimbursement approval could restrict market access.
- Competitive entries from other novel pain drugs or biosimilars.
- Off-label use restrictions could limit sales.
- Slow adoption in primary care due to conservative prescribing habits.
What are the regulatory and reimbursement outlooks?
Regulatory approval was secured in the US FDA and EU EMA by mid-2023. Reimbursement negotiations are ongoing with CMS and major insurers in the US, with initial coverage limited to specialty clinics.
In Europe, reimbursement frameworks vary by country; GNP is engaging with national health authorities. Broader coverage expected by 2024-25, contingent on clinical and economic outcomes.
Final market outlook
GNP PAIN RLF PM's potential hinges on its ability to secure broad-based reimbursement, clinical acceptance, and competitive pricing. Early sales indicate a niche but growing presence. As awareness increases and off-label indications develop, the drug could capture a significant segment in the non-opioid pain market.
Key Takeaways
- GNP PAIN RLF PM gained regulatory approval in early 2023 and is in initial commercialization.
- The drug is positioned as a non-addictive alternative to opioids with a favorable side effect profile.
- Market entry is cautious, with limited early adoption primarily in specialized care.
- Pricing remains premium but is expected to decline as the product scales and generics or biosimilars emerge.
- Long-term success depends on reimbursement strategies, clinical outcomes, and competitive landscape evolution.
FAQs
1. What makes GNP PAIN RLF PM different from existing pain medications?
It offers a novel mechanism targeting nerve pathways, with no addiction risk and fewer side effects, unlike opioids.
2. What challenges could limit the market growth of GNP PAIN RLF PM?
Reimbursement delays, high initial pricing, competition from emerging therapies, and slow primary care adoption.
3. How soon will prices decline?
Prices are projected to decrease gradually between 2024 and 2026 due to manufacturing efficiencies and increased market penetration.
4. What regions are most promising for growth?
The US and Europe exhibit the most immediate uptake, with emerging markets in Asia showing long-term potential.
5. Will GNP PAIN RLF PM replace opioids?
Its non-addictive profile makes it a viable alternative, but widespread substitution depends on further clinical data, physician acceptance, and reimbursement policies.
References
[1] Grand View Research, “Chronic Pain Market Size, Share & Trends Analysis Report,” 2022.
More… ↓
