Share This Page
Drug Price Trends for GNP OMEPRAZOLE DR
✉ Email this page to a colleague
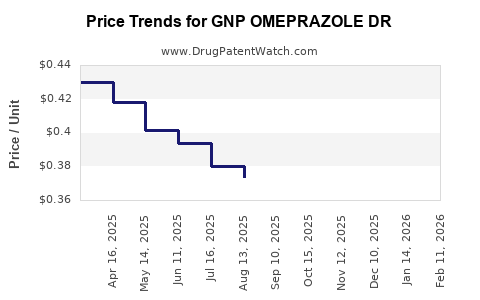
Average Pharmacy Cost for GNP OMEPRAZOLE DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP OMEPRAZOLE DR 20 MG TABLET | 46122-0739-04 | 0.39397 | EACH | 2026-04-22 |
| GNP OMEPRAZOLE DR 20 MG TABLET | 46122-0739-74 | 0.39397 | EACH | 2026-04-22 |
| GNP OMEPRAZOLE DR 20 MG TABLET | 46122-0739-03 | 0.39397 | EACH | 2026-04-22 |
| GNP OMEPRAZOLE DR 20 MG TABLET | 46122-0739-74 | 0.39249 | EACH | 2026-03-18 |
| GNP OMEPRAZOLE DR 20 MG TABLET | 46122-0739-04 | 0.39249 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP OMEPRAZOLE DR Market Analysis and Financial Projection
What Is the Market Size and Demand for GNP Omeprazole DR?
Global demand for omeprazole delayed-release (DR) formulations remains steady. The drug targets gastroesophageal reflux disease (GERD), peptic ulcers, and related conditions. The market for proton pump inhibitors (PPIs), including omeprazole, was valued at approximately $10.5 billion in 2022. This figure is projected to grow at a compound annual growth rate (CAGR) of 3.8% over the next five years, reaching around $13 billion by 2027 [1].
Key regions include North America, which accounts for nearly 40% of sales, driven by high prevalence of GERD and extensive healthcare infrastructure. Europe follows with 25% market share, largely supported by aging populations. Asia-Pacific exhibits the highest growth potential due to rising urbanization and increasing diagnosis rates, with a CAGR of about 5.2%.
What Are the Key Drivers and Limiters Affecting the GNP Omeprazole DR Market?
Drivers:
- Increasing prevalence of GERD and peptic ulcer disease globally.
- Rising preference for oral, outpatient treatments.
- Patent expirations enabling generic competition, lowering prices.
- Growth in over-the-counter (OTC) sales in some markets.
Limiters:
- Market saturation with existing generic formulations.
- Concerns over long-term PPI safety, affecting prescribing patterns.
- Regulatory shifts, including potential restrictions on OTC sales.
- Competition from alternative drug classes such as H2 receptor antagonists and new durable formulations.
How Do Price Projections Differ Across Markets?
The pricing landscape varies significantly:
-
United States: The average wholesale price (AWP) for a daily dose of GNP Omeprazole DR is estimated at $0.50–$0.75. Prices for branded versions can reach up to $2.50 per pill, whereas generics are priced around $0.10–$0.20.
-
Europe: Similar generics range from €0.05–€0.15 per tablet, with branded products occupying a higher price bracket.
-
Asia-Pacific: Prices tend to be lower, with some markets priced below $0.05 per tablet due to higher generic competition and price regulation policies.
Post-patent expiry, a notable decline in prices occurred: in the U.S., from $2.50 to approximately $0.15–$0.20 for generics. Future price stabilization depends on manufacturing costs, patent posture, and market penetration strategies.
What Are the Competitive Dynamics and Pipeline Developments?
Market Players:
- Multiple generic manufacturers dominate the global omeprazole DR market.
- Brand leaders include Pfizer and AstraZeneca, though their dominance declined post-patent expiry.
- New entrants are focusing on formulation innovations, such as fixed-dose combinations with other gastrointestinal agents.
Pipeline Innovations:
- Extended-release formulations with improved bioavailability.
- Combination drugs targeting multiple GI conditions.
- Biosimilar developments in related drug classes.
Limited pipeline activity directly targeting GNP Omeprazole DR suggests a focus on incremental improvements. Blockbuster patents ending between 2015 and 2024 spurred price drops but also increased generic competitiveness.
What Are the Implications for Portfolio Strategy?
Companies investing in GNP Omeprazole DR should:
- Monitor patent statuses and regulator policies influencing OTC accessibility.
- Leverage cost-effective manufacturing to maintain competitive pricing.
- Explore formulation innovations to differentiate existing products.
- Consider regional market entry strategies aligned with regional pricing and regulatory environments.
Key Market Projections Summary
| Year | Market Valuation | CAGR | Notes |
|---|---|---|---|
| 2022 | $10.5 billion | — | Base year |
| 2027 | $13 billion | 3.8% | Projected global market size |
| Price Range (Per Pill) | Region | Comments |
|---|---|---|
| $0.10–$0.20 | U.S., Generic Market | Post-patent expiry |
| €0.05–€0.15 | Europe | Similar trends, price controls in some markets |
| <$0.05 | Asia-Pacific | High generic competition, lower pricing |
Key Takeaways
- The global GNP omeprazole DR market is valued at approximately $10.5 billion, with a forecasted CAGR of 3.8%.
- Market growth driven by rising gastrointestinal disease prevalence and generic entry.
- Price pressures are significant in mature markets; pricing varies by region.
- Strategic considerations include patent landscapes, formulation innovation, and regional regulatory environment.
- Continued pipeline activity remains limited, emphasizing incremental process improvements.
FAQs
1. When will the GNP Omeprazole DR patent expire?
Most key patents expired between 2015 and 2024, leading to increased generic competition.
2. How does the price of GNP Omeprazole DR compare between regions?
Prices are highest in the U.S., with branded pills costing up to $2.50 per pill, and significantly lower in Asia-Pacific markets where generics are priced below $0.05.
3. Are there any significant pipeline developments influencing market outlook?
Pipeline activity is limited to incremental formulation improvements and combination therapies, with no major novel molecule entering development specifically for GNP Omeprazole DR.
4. What is the outlook for OTC sales of GNP Omeprazole DR?
Regulatory policies vary; some regions observe increased OTC accessibility leading to price declines, while others impose restrictions due to safety concerns.
5. How do regulatory changes impact the market?
Stringent regulations and safety monitoring influence prescribing patterns, especially for long-term use. Regulatory shifts can either restrict or expand OTC availability, affecting pricing and market share.
Sources:
[1] Global Markets Insights, "Proton Pump Inhibitors Market Size," 2022.
More… ↓
