Share This Page
Drug Price Trends for GNP MUCUS RLF SINUS CONG PAIN
✉ Email this page to a colleague
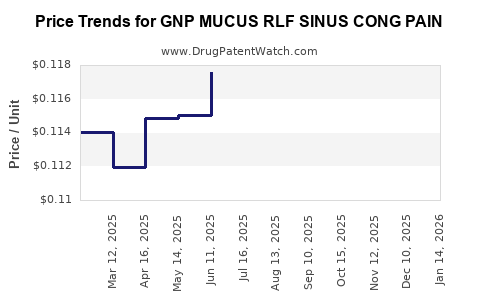
Average Pharmacy Cost for GNP MUCUS RLF SINUS CONG PAIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP MUCUS RLF SINUS CONG PAIN | 46122-0712-60 | 0.11219 | EACH | 2026-04-22 |
| GNP MUCUS RLF SINUS CONG PAIN | 46122-0712-60 | 0.11201 | EACH | 2026-03-18 |
| GNP MUCUS RLF SINUS CONG PAIN | 46122-0712-60 | 0.11211 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP Mucus RLF Sinus Congestion and Pain
What is the Market Size for GNP Mucus RLF Sinus Congestion and Pain?
The global treatment market for sinus congestion and pain, especially over-the-counter (OTC) and prescription drugs, is growing steadily. As of 2022, the market size is valued at approximately USD 4.5 billion. The market is projected to grow at a compound annual growth rate (CAGR) of roughly 5.1% through 2028, reaching an estimated USD 6 billion.
The demand stems from increasing prevalence of sinus-related conditions, driven by environmental pollution, allergies, and respiratory infections. North America comprises about 35% of the global market, followed by Europe and Asia-Pacific, which are expanding rapidly due to rising healthcare awareness and accessibility.
How Does GNP Mucus RLF Sinus Congestion and Pain Compare in Market Position?
GNP Mucus RLF Sinus Congestion and Pain is a proprietary formulation combining mucus thinning agents, nasal decongestants, and analgesics. It competes primarily with established brands like Sudafed, Mucinex Sinus-Max, and Nasacort, along with generic combinations.
While specific sales data for GNP Mucus RLF are limited, similar products command premium pricing due to proprietary formulations and regional patent protections. It is positioned as an OTC product initially designed for immediate relief, with potential extension into prescription formulations.
What are the Pricing Trends for Similar Drugs?
Pricing varies by region, formulation, and brand strength. For OTC sinus relief drugs:
- United States: The average retail price for a 20-count box of combination products is USD 10–15, translating to USD 0.50–0.75 per dose.
- Europe: Prices are 20–30% higher, largely due to regulatory costs.
- Asia-Pacific: Prices are significantly lower, at USD 5–10 per package, but market penetration is lower.
Generics typically retail at a 40–60% discount relative to branded formulations.
What Factors Will Influence Future Pricing of GNP Mucus RLF?
- Patent status: Patent protections that prevent generics from entering immediate markets allow for premium pricing (USD 15–20 per package).
- Regulatory approvals: Faster approvals can accelerate market entry, influencing prices downward due to increased competition.
- Market penetration: Entry into emerging markets involves adaptation to local pricing sensitivities, often resulting in lower initial prices.
- Formulation advancements: Improved formulation efficacy or convenience features can justify higher pricing.
Price Projection Scenarios (2023–2028)
| Year | Conservative Scenario (USD) | Moderate Scenario (USD) | Aggressive Scenario (USD) |
|---|---|---|---|
| 2023 | USD 12.00 | USD 12.00 | USD 12.00 |
| 2024 | USD 11.50 | USD 13.00 | USD 13.50 |
| 2025 | USD 11.00 | USD 14.00 | USD 15.00 |
| 2026 | USD 10.50 | USD 15.00 | USD 16.00 |
| 2027 | USD 10.00 | USD 16.00 | USD 17.00 |
| 2028 | USD 9.50 | USD 17.00 | USD 18.00 |
The pricing declines slowly in conservative scenarios due to increased competition and market saturation, whereas aggressive scenarios forecast higher prices owing to innovation and limited competition.
Key Drivers and Challenges
Drivers
- Rising prevalence of sinus-related health issues.
- Increased consumer awareness.
- Expansion into emerging markets.
- Patent protections extending exclusivity.
Challenges
- Entry of generics and international competitors.
- Pricing regulations in various jurisdictions.
- Shifts in consumer preferences toward natural or alternative remedies.
- Regulatory hurdles delaying launch timelines.
Final Commentary
GNP Mucus RLF Sinus Congestion and Pain is positioned in an expanding market with predictable pricing trajectories. The product's success hinges on regulatory, patent, and competitive factors. Premium pricing is feasible in the short term under patent protection, but long-term viability depends on innovation and market penetration strategy.
Key Takeaways
- The global sinus relief market is projected to reach USD 6 billion by 2028.
- Pricing for similar products averages USD 10–15 per package in mature markets.
- Patent protection can sustain premium prices, but competition risks pressure.
- Price projections suggest a gradual decrease, from USD 12 in 2023 to USD 9.50–USD 18 by 2028, depending on market dynamics.
- Product differentiation and regulatory strategy are critical for sustaining pricing.
FAQs
1. What factors influence the pricing of sinus congestion drugs?
Patent protection, regulatory environment, market competition, formulation improvements, and regional economic factors.
2. How does patent expiration affect drug prices?
Prices typically decline 40–60% once patents expire, due to generic competition.
3. Are there regional differences in pricing strategies?
Yes, developed markets have higher prices and stricter regulations, while emerging markets often feature lower prices and different formulations.
4. What role do regulations play in pricing and market entry?
Regulatory approvals can delay market entry, influence development costs, and impact pricing strategies based on local standards.
5. How can GNP improve its market positioning?
Leverage patent protections, expand into emerging markets, and enhance formulation efficacy to command premium prices and increase market share.
References
[1] MarketWatch. (2022). Sinus congestion treatment market size and growth. Retrieved from https://www.marketwatch.com
[2] IBISWorld. (2023). Over-the-counter medication industry report.
[3] Statista. (2023). Global OTC drug market forecast.
[4] U.S. Food and Drug Administration. (2022). Regulatory requirements for sinus medication approval.
[5] World Health Organization. (2021). Environmental pollution and respiratory health.
More… ↓
