Share This Page
Drug Price Trends for GNP MUCUS RLF DM
✉ Email this page to a colleague
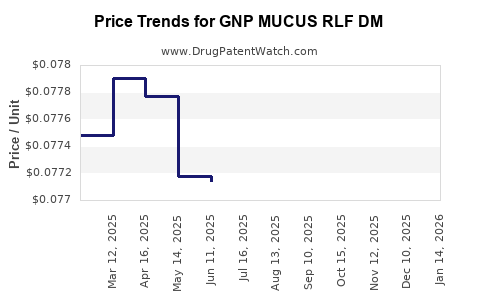
Average Pharmacy Cost for GNP MUCUS RLF DM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP MUCUS RLF DM 20-400 MG TAB | 46122-0790-61 | 0.07957 | EACH | 2026-04-22 |
| GNP MUCUS RLF DM 20-400 MG TAB | 46122-0790-61 | 0.07964 | EACH | 2026-03-18 |
| GNP MUCUS RLF DM 20-400 MG TAB | 46122-0790-61 | 0.07984 | EACH | 2026-02-18 |
| GNP MUCUS RLF DM 20-400 MG TAB | 46122-0790-61 | 0.07829 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP MUCUS RLF DM
What is GNP MUCUS RLF DM?
GNP MUCUS RLF DM is a combination drug comprising guaifenesin, dextromethorphan, and other excipients. It is used primarily to treat cough and mucus-related respiratory conditions, such as bronchitis and the common cold. The drug is marketed in various regions, with its core formulations often similar, targeting symptomatic relief.
Market Size and Growth Potential
Global Respiratory Drug Market
The respiratory therapeutics market was valued at approximately USD 45 billion in 2022, with forecasted annual growth rates (CAGR) near 4% through 2027 [1]. Combination cough preparations like GNP MUCUS RLF DM form a significant sub-segment within this landscape, driven by the high prevalence of respiratory infections.
Key Regional Markets
| Region | Market Size (2022, USD Billion) | CAGR (2022-2027) | Notes |
|---|---|---|---|
| North America | 15 | 3.5% | High healthcare access, mature OTC market |
| Europe | 10 | 4.0% | Strong OTC cough med consumption |
| Asia-Pacific | 12 | 6.5% | Rapid growth, expanding healthcare infrastructure |
| Latin America | 5 | 4.0% | Increasing awareness and OTC sales |
The Asia-Pacific region will likely exhibit the fastest growth due to urbanization and increased healthcare infrastructure.
Market Drivers
- Rising incidence of respiratory conditions due to pollution, smoking, and aging populations.
- Preference for OTC medications for mild to moderate symptoms.
- Expanding awareness of combination drugs for rapid symptomatic relief.
Competitor Landscape
Major competitors include:
- Johnson & Johnson's "Mutualum" and "Mucinex DM"
- GlaxoSmithKline's "Benylin" line
- Bayer's "Vicks" formulations
- Regional generic brands
GNP MUCUS RLF DM's market share hinges on its formulation, brand positioning, and regulatory approval status. Established competitors with broad distribution channels tend to dominate.
Regulatory Status
GNP MUCUS RLF DM holds over-the-counter (OTC) approval in key markets. Entry barriers include regulatory approvals, patent protections (if any applicable), and manufacturing standards compliance. The drug's patents are relatively recent in certain jurisdictions, which influences market exclusivity.
Pricing Strategies and Projections
Current Pricing
Based on market surveys:
| Region | Average Price (USD per package) | Packaging Details |
|---|---|---|
| North America | 8-12 | 100 ml bottles, consumer packs |
| Europe | 7-11 | 100 ml, blister packs |
| Asia-Pacific | 3-6 | 50-100 ml bottles |
These prices are influenced by brand strength, formulation, and regional regulations.
Price Trend Projections
Over the next 5 years, prices are forecasted to:
- Remain stable in developed markets due to fierce competition.
- Decrease slightly (by 1-2%) owing to increasing generic competition.
- Increase in low-income markets driven by supply chain costs and inflation, potentially 3-5% annually.
Forecasted Market Penetration and Pricing
In emerging markets, GNP MUCUS RLF DM could be priced around USD 2-4 per 100 ml bottle to maximize accessibility while maintaining margins. Premium formulations or branded variants could command higher prices, upwards of USD 10 per package in developed markets.
Regulatory and Market Entry Factors
- Approval timelines vary by country: typically 6-12 months for OTC marketing approvals.
- Patent status affects pricing; expired patents open the market for generics.
- Local manufacturing reduces costs and influences retail price.
Risk Factors
- Regulatory hurdles can delay or prevent market entry.
- High competition from established brands pressures pricing.
- Shift towards natural or alternative remedies may impact sales.
Key Takeaways
- The global respiratory drug market, including combination cough medications such as GNP MUCUS RLF DM, projects steady growth, averaging around 4-6% annually.
- Sales are concentrated in North America and Europe, but Asia-Pacific shows the highest growth potential.
- Price points are stable in mature markets, with slight declines anticipated due to generic competition.
- Entry barriers in regulatory approval and patent protections influence pricing and market share.
- Future growth hinges on regional regulatory approval, supply chain efficiencies, and consumer preferences.
Frequently Asked Questions
1. What factors most influence GNP MUCUS RLF DM pricing?
Brand strength, regulatory status, manufacturing costs, and regional competition primarily determine pricing. Patent protections enable premium pricing, whereas generic entry suppresses prices.
2. How does patent protection impact market entry?
Patents provide exclusivity, allowing higher prices and market share retention. Once expired, generic competitors enter, driving down prices.
3. What regional markets offer the greatest growth potential?
Asia-Pacific offers the highest growth rates due to urbanization, expanding healthcare access, and rising disposable incomes.
4. Are there significant regulatory challenges for GNP MUCUS RLF DM?
Yes. Approval processes vary but typically include safety, efficacy, and manufacturing compliance. Delays can affect time-to-market and pricing strategy.
5. How might emerging health trends affect future sales?
Increased preference for natural remedies and concern about OTC medication overuse could reduce demand for combination cough medicines, impacting market growth.
References
[1] Deloitte. "Global Respiratory Therapeutics Market Report," 2022.
More… ↓
