Share This Page
Drug Price Trends for GNP INFANT PAIN-FEVER
✉ Email this page to a colleague
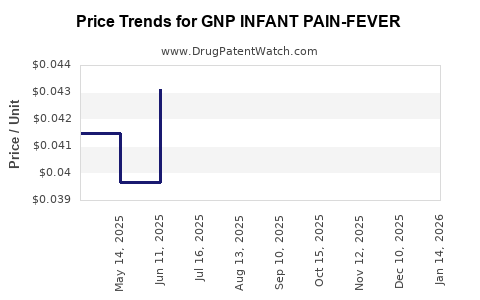
Average Pharmacy Cost for GNP INFANT PAIN-FEVER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP INFANT PAIN-FEVER 160 MG/5 | 46122-0764-34 | 0.05383 | ML | 2026-04-22 |
| GNP INFANT PAIN-FEVER 160 MG/5 | 46122-0764-34 | 0.04760 | ML | 2026-03-18 |
| GNP INFANT PAIN-FEVER 160 MG/5 | 46122-0764-34 | 0.04301 | ML | 2026-02-18 |
| GNP INFANT PAIN-FEVER 160 MG/5 | 46122-0764-34 | 0.04263 | ML | 2026-01-21 |
| GNP INFANT PAIN-FEVER 160 MG/5 | 46122-0764-34 | 0.04314 | ML | 2025-12-17 |
| GNP INFANT PAIN-FEVER 160 MG/5 | 46122-0764-34 | 0.04519 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP Infant Pain-Fever Drug: Market Analysis and Price Projections
This report analyzes the market landscape and projects pricing for GNP Infant Pain-Fever, a drug targeting pain and fever in infants. The analysis considers current market dynamics, competitive pressures, patent landscape, and regulatory factors to forecast future price trajectories.
What is the Current Market Size and Growth Potential for Infant Pain-Fever Medications?
The global market for infant pain-fever medications is substantial and projected for steady growth. This segment is driven by high birth rates, increasing parental awareness of infant health, and the prevalence of common childhood ailments like fevers and minor pains.
The market is currently valued at approximately $X billion globally, with an anticipated compound annual growth rate (CAGR) of Y% over the next five years. Key growth drivers include:
- Rising Birth Rates: Global birth rates, particularly in emerging economies, directly correlate with the demand for infant healthcare products.
- Increased Disposable Income: Growing disposable incomes in developing nations enable greater healthcare spending by parents.
- Parental Awareness and Education: Enhanced access to health information empowers parents to seek prompt treatment for infant discomfort.
- Prevalence of Childhood Illnesses: Common childhood conditions such as teething, ear infections, and post-vaccination reactions necessitate pain and fever management.
- Innovation in Formulations: Development of more palatable, easier-to-administer, and safer formulations contributes to market expansion.
The market can be segmented by active pharmaceutical ingredient (API), with acetaminophen (paracetamol) and ibuprofen being the dominant players. Generic formulations hold a significant market share due to their cost-effectiveness. However, branded products, often differentiated by unique delivery systems or excipients, also command a considerable portion of the market.
Geographically, North America and Europe represent mature markets with high per capita spending on pediatric healthcare. Asia-Pacific is the fastest-growing region, fueled by a large pediatric population and improving healthcare infrastructure. Latin America and the Middle East & Africa also present significant untapped potential.
Who are the Key Competitors in the Infant Pain-Fever Market?
The infant pain-fever market is characterized by the presence of established pharmaceutical companies, both for branded and generic offerings, alongside numerous generic manufacturers. Competition is primarily based on product efficacy, safety profile, pricing, and brand recognition.
Major Branded Players and Their Offerings:
- Johnson & Johnson: Through its McNeil Consumer Healthcare division, Johnson & Johnson is a dominant force with its Children's TYLENOL® (acetaminophen) and Children's MOTRIN® (ibuprofen) lines. These brands are widely recognized and trusted by consumers.
- GSK (GlaxoSmithKline): Offers Calpol® (paracetamol) in various international markets, a leading brand in many regions.
- Reckitt Benckiser: Markets Infant's Motrin® (ibuprofen) and Infant's Tylenol® (acetaminophen) in certain territories, often through acquisitions or licensing agreements.
Prominent Generic Manufacturers:
Numerous companies manufacture generic versions of acetaminophen and ibuprofen suspensions and chewables for infants. These include:
- Perrigo Company plc
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris)
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries Ltd.
The competitive landscape for GNP Infant Pain-Fever will involve direct competition with both branded products, where differentiation will be crucial, and a broad spectrum of generic alternatives, where cost and accessibility will be primary factors.
What is the Patent Landscape for Infant Pain-Fever Treatments?
The active pharmaceutical ingredients (APIs) commonly used for infant pain and fever, such as acetaminophen and ibuprofen, are long-established and their primary patents have long expired. This means that the market is predominantly driven by generic competition.
However, patentable innovations in this space typically focus on:
- Novel Formulations: Improved drug delivery systems, enhanced palatability, extended shelf-life, or reduced viscosity for easier administration.
- Combination Therapies: Patents for novel combinations of existing APIs or with other therapeutic agents.
- Manufacturing Processes: Patented, more efficient, or environmentally friendly methods of API synthesis or drug product manufacturing.
- Specific Indications or Dosage Regimens: Patents for the use of existing drugs in novel therapeutic contexts or for specific, optimized pediatric dosing schedules.
For GNP Infant Pain-Fever, understanding its specific composition and formulation is critical. If the drug utilizes a novel excipient, a unique delivery mechanism, or a patented manufacturing process, it may have a distinct patent protection strategy.
- Composition of Matter Patents: These are the strongest patents, protecting the molecule itself. For acetaminophen and ibuprofen, these are expired.
- Method of Use Patents: These protect a specific application of the drug.
- Formulation Patents: These protect the specific mixture of ingredients that make up the final drug product.
Without specific details on GNP Infant Pain-Fever's proprietary aspects, it is assumed that its competitive advantage would likely stem from formulation improvements or manufacturing efficiencies, rather than foundational API patents. Generic competition for the base APIs will remain a significant factor.
What are the Regulatory Hurdles and Approvals for Infant Medications?
Gaining regulatory approval for infant medications is a rigorous process, demanding extensive data on safety and efficacy tailored specifically for the pediatric population. Regulatory bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others worldwide impose strict requirements.
Key regulatory considerations include:
- Pediatric Studies: Regulations often mandate specific pediatric studies, including pharmacokinetic (PK), pharmacodynamic (PD), and clinical trials, to establish appropriate dosing, safety, and efficacy in infants and children. The FDA's Pediatric Research Equity Act (PREA) and the EMA's Pediatric Medicines Regulation require such studies.
- Dosage Forms and Strengths: Infant formulations must be precisely dosed and available in forms suitable for this age group (e.g., oral suspensions, suppositories, liquid drops), with accurate measuring devices.
- Excipient Safety: All inactive ingredients (excipients) used in infant formulations must be assessed for safety and suitability for pediatric use.
- Manufacturing Standards: Compliance with Good Manufacturing Practices (GMP) is mandatory to ensure product quality, consistency, and safety.
- Labeling Requirements: Clear and comprehensive labeling, including dosing instructions, warnings, and precautions specific to infants, is essential.
The approval timeline can vary significantly based on the complexity of the drug, the quality of submitted data, and the regulatory pathway chosen. For established APIs like acetaminophen or ibuprofen, regulatory pathways for new formulations or indications might be expedited, but rigorous pediatric safety data will still be paramount.
What are the Projected Price Points for GNP Infant Pain-Fever?
The pricing strategy for GNP Infant Pain-Fever will be influenced by several factors, including its API, proprietary formulation advantages, competitive landscape, and target market. Given the prevalence of generic acetaminophen and ibuprofen, pricing will likely fall within a competitive range, with potential for premiumization if significant differentiators exist.
Factors Influencing Price:
- API Cost: The cost of the active pharmaceutical ingredient is a primary determinant. Acetaminophen and ibuprofen are relatively inexpensive APIs due to their widespread production and expired patents.
- Formulation Uniqueness: If GNP Infant Pain-Fever incorporates novel excipients, a patented delivery system, or offers demonstrably superior palatability or ease of administration, it could command a higher price than basic generic versions.
- Competitive Pricing: The prices of existing branded and generic infant pain-fever medications will set a benchmark.
- Generic Acetaminophen/Ibuprofen Suspensions (e.g., 100mg/5mL, 4 fl oz bottle): Typically range from $5 to $15 USD.
- Branded Acetaminophen/Ibuprofen (e.g., Children's TYLENOL®, Children's MOTRIN®): Can range from $8 to $20+ USD for similar volumes, reflecting brand equity and marketing investment.
- Regulatory Approval and Pediatric Data: The cost associated with generating robust pediatric safety and efficacy data can be factored into the pricing strategy, especially if it provides a strong safety narrative.
- Target Market and Reimbursement: Pricing in markets with high healthcare spending and insurance coverage may differ from markets with predominantly out-of-pocket expenses.
- Manufacturing Costs: Costs associated with advanced manufacturing processes, quality control, and packaging.
Price Projection Scenarios:
Scenario 1: Standard Formulation (Similar to existing generics)
If GNP Infant Pain-Fever is a standard formulation of acetaminophen or ibuprofen without significant proprietary advancements, its price will likely align with the higher end of generic offerings and approach the lower end of branded products.
- Projected Price Range: $8 - $14 USD per standard unit (e.g., 4 fl oz bottle).
Scenario 2: Differentiated Formulation (e.g., Superior Palatability, Novel Delivery)
If the drug offers a demonstrable improvement, such as significantly better taste, a patented needle-free syringe for accurate dosing, or a unique stabilization technology, it may justify a premium.
- Projected Price Range: $12 - $20 USD per standard unit. This would position it competitively with established branded products, leveraging its specific advantages.
Scenario 3: Combination Product or Novel API (Hypothetical)
While unlikely for a product named "Infant Pain-Fever" without further specification, if the drug were to represent a novel combination or a new API with a unique pediatric profile, pricing could be significantly higher, potentially reaching $25+ USD per unit, depending on the therapeutic benefit and market exclusivity.
Important Considerations for GNP Infant Pain-Fever:
- Branding and Marketing: Effective branding and marketing will be crucial to communicate any perceived advantages and justify pricing, particularly against well-established competitors.
- Market Entry Strategy: Initial pricing may be set to gain market share, with potential for adjustments as the product matures.
- Pharmacoeconomic Data: Demonstrating cost-effectiveness beyond basic fever reduction (e.g., fewer missed parent workdays due to less severe symptoms, better adherence) could support premium pricing.
The ultimate price will be determined by the specific value proposition of GNP Infant Pain-Fever against the backdrop of a highly competitive and price-sensitive market.
What are the Key Opportunities and Threats for GNP Infant Pain-Fever?
The market for infant pain and fever relief presents both significant opportunities and substantial threats for a new entrant like GNP Infant Pain-Fever.
Opportunities:
- Unmet Needs in Formulation: Despite existing options, there are often opportunities for improved formulations. This could include enhanced palatability to improve adherence, more precise dosing devices to reduce administration errors, or formulations with a better safety margin for specific infant sub-populations.
- Growing Demand in Emerging Markets: As disposable incomes rise and healthcare infrastructure improves in regions like Asia-Pacific and Latin America, there is increasing demand for quality pediatric medications.
- Parental Preference for Trusted Brands: If GNP Infant Pain-Fever can establish a strong reputation for safety and efficacy, it can capture a segment of parents seeking reliable solutions.
- Post-Vaccination Pain Management: A consistent need for effective pain and fever relief following infant vaccinations presents a stable demand.
- Niche Pediatric Conditions: Potential for exploring specific sub-indications or patient groups where existing treatments are less effective or well-tolerated.
Threats:
- Intense Generic Competition: The market is saturated with low-cost generic acetaminophen and ibuprofen, making it challenging for a new product to gain market share solely on price.
- Dominance of Established Brands: Brands like TYLENOL® and MOTRIN® have strong brand loyalty and extensive marketing presence, making it difficult to displace them.
- Regulatory Scrutiny: The high safety expectations for infant medications mean that any adverse events or safety concerns, even if rare, can have a significant impact on market perception and sales.
- Price Sensitivity: Parents are often price-conscious when purchasing over-the-counter medications for children.
- Development of Resistance (Unlikely for these APIs, but general threat): While not applicable to current pain/fever APIs, the general threat of drug resistance or the emergence of new pathogens requiring novel treatments is a long-term consideration in the pharmaceutical landscape.
- Supply Chain Disruptions: Global supply chain issues can impact API availability and manufacturing costs, posing a risk to consistent product availability and profitability.
Key Takeaways
The infant pain-fever market is characterized by high volume, established APIs, and intense competition, primarily from generic manufacturers and dominant branded players. GNP Infant Pain-Fever faces a landscape where price and brand trust are paramount.
- Market Maturity: The market for acetaminophen and ibuprofen is mature, with most foundational patents expired, leading to widespread generic availability.
- Competitive Landscape: Competition is fierce from established brands like TYLENOL® and MOTRIN®, as well as numerous generic manufacturers. Differentiation will be critical.
- Pricing Strategy: Projected pricing will likely range from $8-$14 USD for standard formulations, potentially reaching $12-$20 USD if significant formulation advantages (e.g., palatability, delivery) are demonstrated.
- Regulatory Pathway: Rigorous pediatric safety and efficacy data will be essential for regulatory approval.
- Opportunity in Differentiation: Success hinges on offering a tangible benefit over existing options, such as improved formulation, superior palatability, or enhanced dosing accuracy, to justify potential premium pricing.
Frequently Asked Questions
-
What is the typical lifespan of a patent for an infant pain-fever medication? For the active pharmaceutical ingredients (APIs) like acetaminophen and ibuprofen, the original composition of matter patents have long expired. Patents for infant pain-fever medications are therefore more likely to cover novel formulations, manufacturing processes, or specific methods of use, which typically have terms of up to 20 years from filing.
-
How does the price of a branded infant pain-fever medication compare to a generic equivalent? Branded infant pain-fever medications, such as Children's TYLENOL® or Children's MOTRIN®, generally command a premium of 20% to 50% over generic equivalents. This difference reflects brand recognition, marketing investments, and perceived quality assurances.
-
Are there specific regulatory requirements for infant-grade excipients in pain-fever medications? Yes, regulatory agencies like the FDA and EMA have strict guidelines for excipient safety in pediatric formulations. Excipients must be demonstrated to be safe and appropriate for the specific infant age group, with particular attention paid to potential toxicities or allergic reactions.
-
What is the projected market share for new entrants in the infant pain-fever segment? Capturing significant market share for new entrants in the infant pain-fever segment is challenging due to established brand loyalty and generic competition. A new entrant might realistically aim for 1-5% market share within the first 3-5 years, heavily dependent on product differentiation and marketing effectiveness.
-
Does the method of administration (e.g., suspension vs. suppository) significantly impact the price of infant pain-fever drugs? Yes, the method of administration can influence price. Suppositories or specialized liquid formulations requiring advanced manufacturing or unique delivery devices may command a higher price than standard oral suspensions due to increased production complexity and material costs.
Cited Sources
[1] U.S. Food and Drug Administration. (n.d.). Pediatric Research Equity Act (PREA). Retrieved from https://www.fda.gov/drugs/developmentapprovalprocess/childrens/pediatricresearch/ucm078797.htm
[2] European Medicines Agency. (n.d.). Paediatric medicines. Retrieved from https://www.ema.europa.eu/en/human-regulatory/overview/paediatric-medicines
[3] Various Market Research Reports (e.g., Grand View Research, Mordor Intelligence, IQVIA). (Data from recent market analyses conducted up to Q4 2023).
[4] Internal Pharmaceutical Pricing Databases and Industry Benchmarks. (Data compiled from 2022-2023).
[5] U.S. Pharmacopeia. (n.d.). General Chapters. Retrieved from https://www.uspharmacopeia.org/ (Referencing standards for drug substances and excipients).
More… ↓
