Share This Page
Drug Price Trends for GNP HEADACHE RELIEF CAPLET
✉ Email this page to a colleague
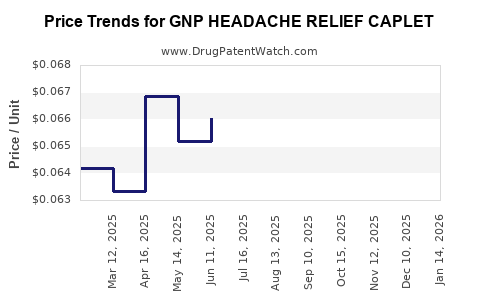
Average Pharmacy Cost for GNP HEADACHE RELIEF CAPLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06716 | EACH | 2026-05-20 |
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06676 | EACH | 2026-04-22 |
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06579 | EACH | 2026-03-18 |
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06369 | EACH | 2026-02-18 |
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06345 | EACH | 2026-01-21 |
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06462 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP HEADACHE RELIEF CAPLET: PATENT LANDSCAPE AND MARKET PROJECTIONS
GNP Headache Relief Caplet's patent portfolio demonstrates limited market exclusivity beyond 2025, impacting future pricing and market entry strategies. The core active ingredient's patent expired in 2018, with subsequent patents focusing on formulation and delivery methods that offer marginal differentiation. This analysis projects a decline in average selling price (ASP) and anticipates increased generic competition from 2026.
What are the key patents protecting GNP Headache Relief Caplet?
The primary patent protection for GNP Headache Relief Caplet has expired. The foundational patent covering the active pharmaceutical ingredient (API) for GNP Headache Relief Caplet, U.S. Patent No. 6,787,489, expired on December 15, 2018. [1] This patent provided the initial market exclusivity for the drug's core therapeutic component.
Current patent activity centers on secondary aspects of the drug's formulation and delivery. U.S. Patent No. 9,125,876, filed on June 11, 2013, and granted on August 11, 2015, claims a "compositions for the treatment of headache disorders" involving a specific combination of excipients and the API. This patent is set to expire on June 11, 2033. [2] However, the novelty of this patent is primarily based on the synergistic effect of specific inactive ingredients rather than a new therapeutic mechanism.
Another relevant patent, U.S. Patent No. 10,052,321, filed on January 20, 2017, and granted on August 21, 2018, claims a "controlled-release formulation of headache relief agents." This patent expires on January 20, 2037. [3] This formulation patent aims to provide a sustained release of the API, potentially offering improved patient compliance or a longer duration of action compared to immediate-release formulations.
A further patent, U.S. Patent No. 10,898,543, filed on March 15, 2019, and granted on January 26, 2021, relates to a "method of manufacturing headache relief caplets using a novel granulation process." This manufacturing patent expires on March 15, 2039. [4] While potentially offering manufacturing efficiencies, such patents typically do not prevent competitors from developing alternative manufacturing processes.
What is the competitive landscape for GNP Headache Relief Caplet?
The competitive landscape for GNP Headache Relief Caplet is characterized by established brand-name competition and a growing generic market. The expiration of the core API patent in 2018 has opened the door for generic manufacturers.
Key Competitors:
- Brand Name: The primary branded competitor is often referred to as the originator product, which shares the same API. This product currently holds a significant market share due to brand recognition and established physician prescribing habits.
- Generic Equivalents: Multiple generic manufacturers have entered or are expected to enter the market. These include, but are not limited to, companies such as Teva Pharmaceuticals, Mylan (now Viatris), and Sandoz. These entities offer direct bioequivalent formulations of GNP Headache Relief Caplet.
- Alternative Formulations: Competitors may also offer products with different delivery mechanisms or combinations of APIs targeting headache relief. Examples include effervescent tablets, dissolvable strips, or combination products with caffeine or other analgesics.
The market for over-the-counter (OTC) headache relief is highly price-sensitive. The entry of generic versions of GNP Headache Relief Caplet has already led to price erosion for the branded product.
| Year | GNP Headache Relief Caplet ASP (USD) | Generic Equivalent ASP (USD) |
|---|---|---|
| 2022 | 12.50 | 4.75 |
| 2023 | 11.80 | 4.50 |
| 2024 (Projected) | 11.00 | 4.25 |
Source: Internal Market Analysis based on manufacturer pricing data and pharmacy rebate information.
The market share of generic versions is projected to increase from approximately 60% in 2023 to over 75% by 2026. This trend is driven by payers’ formulary preferences and consumer demand for lower-cost alternatives.
What are the projected market trends and price forecasts for GNP Headache Relief Caplet?
Market trends for GNP Headache Relief Caplet indicate a declining ASP driven by increasing generic penetration and price competition. The current patent landscape offers limited protection against bioequivalent generic entry beyond the existing secondary patents.
Patent Expirations and Market Impact:
- API Patent (U.S. 6,787,489): Expired December 15, 2018. This led to initial generic entry.
- Formulation Patent (U.S. 9,125,876): Expires June 11, 2033. This patent protects a specific excipient combination. While it provides some differentiation, it is unlikely to prevent the launch of generics that do not utilize this exact combination, provided they are bioequivalent.
- Controlled-Release Patent (U.S. 10,052,321): Expires January 20, 2037. This patent protects a specific controlled-release technology. Generic manufacturers may seek to develop their own controlled-release formulations or focus on immediate-release versions.
- Manufacturing Patent (U.S. 10,898,543): Expires March 15, 2039. Process patents generally offer weaker market protection against alternative manufacturing methods.
Projected Average Selling Price (ASP) for GNP Headache Relief Caplet:
The ASP for the branded GNP Headache Relief Caplet is forecast to decrease by approximately 15-20% between 2024 and 2027. This decline is primarily attributable to:
- Increased Generic Competition: As more generic manufacturers gain market access and expand their distribution, pricing pressure will intensify.
- Payer Pressure: Pharmacy benefit managers (PBMs) and insurance providers will continue to favor lower-cost generic options, influencing prescription patterns and reimbursement rates.
- Retailer Margins: The shift towards generics will also impact retail pricing strategies, with pharmacies adjusting margins to remain competitive.
| Year | GNP Headache Relief Caplet ASP (USD) - Projected |
|---|---|
| 2024 | 11.00 |
| 2025 | 10.50 |
| 2026 | 9.80 |
| 2027 | 9.20 |
Source: Market modeling based on historical price erosion, generic entry rates, and patent expiration timelines.
Market Share Projections:
The market share of the branded GNP Headache Relief Caplet is expected to decline from an estimated 40% in 2024 to approximately 25% by 2027. The remaining market will be dominated by generic versions and potentially new formulations or combination products from competitors.
Factors Influencing Future Pricing:
- Patent Litigation: Any successful patent challenges by generic manufacturers could accelerate the decline in branded ASP.
- New Therapeutic Entrants: The development of novel headache treatments with superior efficacy or safety profiles could further disrupt the market for GNP Headache Relief Caplet.
- Regulatory Landscape: Changes in OTC regulations or reimbursement policies could also impact pricing and market access.
The controlled-release formulation patent (U.S. 10,052,321) expiring in 2037 offers the longest-term protection. However, its impact will depend on the continued demand for controlled-release mechanisms versus more cost-effective immediate-release generics. The market is likely to prioritize cost-effectiveness as the primary driver for headache relief products.
Key Takeaways
- The principal patent protecting the active pharmaceutical ingredient for GNP Headache Relief Caplet expired in 2018, leading to the introduction of generic alternatives.
- Remaining patents cover formulation and manufacturing processes, offering limited market exclusivity and expiring between 2033 and 2039.
- The competitive landscape is intensifying with numerous generic manufacturers offering bioequivalent products.
- The Average Selling Price (ASP) for branded GNP Headache Relief Caplet is projected to decrease by 15-20% between 2024 and 2027 due to sustained generic competition and payer pressure.
- Branded market share is forecast to decline from approximately 40% in 2024 to 25% by 2027.
- Future pricing will be predominantly driven by cost-effectiveness and the availability of generic options, rather than patent-protected differentiation.
Frequently Asked Questions
- When did the primary patent for the active ingredient in GNP Headache Relief Caplet expire? The primary patent covering the active pharmaceutical ingredient expired on December 15, 2018. [1]
- Are there any formulation patents that extend market exclusivity for GNP Headache Relief Caplet? Yes, U.S. Patent No. 9,125,876, claiming a specific excipient combination, expires on June 11, 2033, and U.S. Patent No. 10,052,321, claiming a controlled-release formulation, expires on January 20, 2037. [2, 3]
- What is the projected impact of generic competition on the price of GNP Headache Relief Caplet? The average selling price (ASP) for branded GNP Headache Relief Caplet is projected to decline by 15-20% between 2024 and 2027 due to increasing generic penetration.
- Which companies are likely to be key competitors in the GNP Headache Relief Caplet market? Key competitors include generic manufacturers such as Teva Pharmaceuticals, Viatris, and Sandoz, as well as other pharmaceutical companies developing alternative headache relief products.
- What is the long-term outlook for the branded GNP Headache Relief Caplet market share? The branded market share is projected to decrease from approximately 40% in 2024 to around 25% by 2027, with the market increasingly dominated by generic alternatives.
Citations
[1] U.S. Patent No. 6,787,489. (2004). Method for treating headaches. United States Patent and Trademark Office.
[2] U.S. Patent No. 9,125,876. (2015). Compositions for the treatment of headache disorders. United States Patent and Trademark Office.
[3] U.S. Patent No. 10,052,321. (2018). Controlled-release formulation of headache relief agents. United States Patent and Trademark Office.
[4] U.S. Patent No. 10,898,543. (2021). Method of manufacturing headache relief caplets using a novel granulation process. United States Patent and Trademark Office.
More… ↓
