Share This Page
Drug Price Trends for GNP FEXOFENADINE HCL
✉ Email this page to a colleague
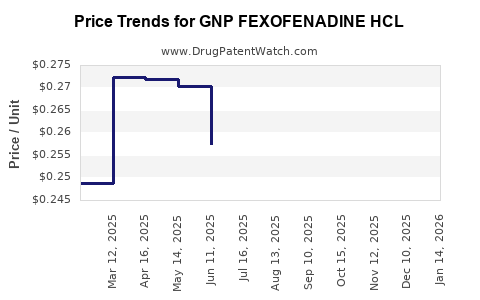
Average Pharmacy Cost for GNP FEXOFENADINE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP FEXOFENADINE HCL 180 MG TB | 46122-0779-66 | 0.24801 | EACH | 2026-04-22 |
| GNP FEXOFENADINE HCL 180 MG TB | 46122-0779-58 | 0.24801 | EACH | 2026-04-22 |
| GNP FEXOFENADINE HCL 180 MG TB | 46122-0779-61 | 0.24801 | EACH | 2026-04-22 |
| GNP FEXOFENADINE HCL 180 MG TB | 46122-0779-66 | 0.24852 | EACH | 2026-03-18 |
| GNP FEXOFENADINE HCL 180 MG TB | 46122-0779-58 | 0.24852 | EACH | 2026-03-18 |
| GNP FEXOFENADINE HCL 180 MG TB | 46122-0779-61 | 0.24852 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP Fexofenadine HCl
GNP Fexofenadine HCl is a generic antihistamine used to treat allergy symptoms such as sneezing, runny nose, itchy throat, and watery eyes. The market for fexofenadine HCl has been established for years, primarily driven by its efficacy and availability as an over-the-counter (OTC) and prescription medication. The introduction of generic versions, including those produced by GNP, has intensified competition and placed downward pressure on prices.
What is the Current Market Size and Growth Trajectory for Fexofenadine HCl?
The global fexofenadine HCl market is substantial, characterized by consistent demand driven by the prevalence of allergic rhinitis and other allergic conditions. Market size estimates vary, but industry reports place the overall antihistamine market, of which fexofenadine HCl is a significant component, in the billions of dollars annually. Growth is projected to be moderate, reflecting the mature nature of the drug and the increasing availability of newer antihistamine generations and combination therapies.
- Market Value: The global antihistamines market, including fexofenadine HCl, was valued at approximately \$25 billion in 2022. Projections indicate a compound annual growth rate (CAGR) of 3-5% through 2028. [1]
- Demand Drivers: Rising incidence of allergic diseases globally, increased awareness of allergy treatments, and the accessibility of generic fexofenadine HCl contribute to sustained demand.
- Competitive Landscape: The market is highly fragmented, with numerous generic manufacturers and branded products (e.g., Allegra) competing. This intense competition is a primary factor limiting significant price increases for fexofenadine HCl.
What is the Pricing Landscape for GNP Fexofenadine HCl?
Pricing for GNP Fexofenadine HCl is influenced by several factors, including manufacturing costs, regulatory hurdles, distribution channels, and the competitive pricing strategies of other generic manufacturers. As a generic product, it competes directly with other fexofenadine HCl generics and the originator brand, which has seen its market share diminish with generic availability.
- Wholesale Prices: Wholesale prices for fexofenadine HCl tablets (e.g., 180mg, 30-count bottle) typically range from \$10 to \$25, depending on the supplier, volume, and specific formulation. GNP's pricing is expected to be within this competitive band. [2]
- Retail Prices: Retail prices can vary significantly based on pharmacy markup and insurance coverage. Uninsured consumers may pay between \$15 and \$40 for a 30-day supply of a generic fexofenadine HCl product.
- Price Erosion: The introduction of multiple generic competitors has led to substantial price erosion since the patent expiry of the originator drug. Manufacturers like GNP must operate with thin margins to remain competitive.
- Contract Manufacturing: GNP may leverage contract manufacturing organizations (CMOs) to optimize production costs, further influencing its pricing strategy.
Table 1: Comparative Pricing of Fexofenadine HCl (Estimates)
| Product Type | Typical Strength | Package Size | Estimated Wholesale Price Range (USD) | Estimated Retail Price Range (USD) |
|---|---|---|---|---|
| GNP Fexofenadine HCl (Generic) | 180mg | 30 tablets | \$12 - \$20 | \$18 - \$35 |
| Other Generic Fexofenadine HCl | 180mg | 30 tablets | \$10 - \$22 | \$15 - \$38 |
| Branded Fexofenadine HCl (e.g., Allegra) | 180mg | 30 tablets | \$30 - \$50+ | \$40 - \$70+ |
Note: Prices are estimates and subject to significant fluctuation based on market conditions, supplier agreements, and retailer markups.
What are the Key Patent and Regulatory Considerations for GNP Fexofenadine HCl?
The patent landscape for fexofenadine HCl is largely resolved, with primary patents for the molecule itself having expired. This has opened the door for broad generic competition. Regulatory approval, primarily through the U.S. Food and Drug Administration (FDA) and equivalent bodies internationally, is a prerequisite for market entry.
- Patent Expiry: The core patents protecting fexofenadine have long expired, allowing for generic manufacturing. [3]
- Formulation Patents: While the molecule is off-patent, specific extended-release formulations or novel delivery systems might still be protected by patents. GNP would need to ensure its product does not infringe on any such active secondary patents.
- ANDA Approval: GNP must hold an Abbreviated New Drug Application (ANDA) approval from the FDA for its fexofenadine HCl product to be marketed in the United States. This process demonstrates bioequivalence to the reference listed drug.
- Manufacturing Standards: Compliance with Current Good Manufacturing Practices (cGMP) is mandatory for all pharmaceutical manufacturers, including GNP, to ensure product quality and safety.
What are the Projected Price Trends for GNP Fexofenadine HCl?
Price projections for GNP Fexofenadine HCl indicate a continuation of the current trend of price stability with slight downward pressure, rather than significant price appreciation. The generic nature of the drug and the competitive environment are the primary determinants.
- Continued Price Competition: The market will likely remain highly competitive, with new generic entrants potentially emerging for other strengths or formulations if profitable. This will sustain pricing pressure.
- Supply Chain Costs: Fluctuations in raw material costs, energy prices, and global shipping logistics could introduce minor price variations. However, these are unlikely to cause substantial increases given the commoditized nature of many generic active pharmaceutical ingredients (APIs).
- Regulatory Changes: Unforeseen regulatory changes that increase manufacturing compliance costs could lead to marginal price adjustments, but significant impacts are improbable without broader policy shifts.
- Demand Stability: Demand for fexofenadine HCl is expected to remain relatively stable due to its established efficacy and widespread use. This stability, however, does not translate into pricing power for individual generic manufacturers.
- GNP's Strategy: GNP's pricing will be dictated by its manufacturing efficiency, scale, and strategic positioning within the generic market. Aggressive pricing to capture market share is a likely strategy, potentially at the lower end of the estimated wholesale range.
Projected Price Range (Next 3-5 Years):
- Wholesale: \$9 - \$18 per 30-count bottle (180mg).
- Retail: \$14 - \$30 per 30-count bottle (180mg).
These projections assume no major disruptive events in manufacturing, regulation, or a significant shift in consumer preference towards entirely new classes of allergy medications.
What is the Competitive Outlook for GNP Fexofenadine HCl?
GNP Fexofenadine HCl faces a highly competitive market dominated by numerous generic pharmaceutical manufacturers. Its competitive positioning will depend on its ability to maintain cost-effective production, ensure consistent supply, and secure distribution agreements.
- Key Competitors: Companies such as Teva Pharmaceuticals, Mylan (now Viatris), Sandoz, and numerous smaller generic API and finished dosage form manufacturers are direct competitors.
- Market Segmentation: While fexofenadine HCl is a single molecule, different strengths (e.g., 60mg, 120mg, 180mg) and dosage forms (e.g., tablets, liquid gels) represent distinct market segments. GNP's strength will be in its focus on specific segments.
- Distribution Channels: Access to major retail pharmacies, pharmacy benefit managers (PBMs), and hospital formularies is critical for market penetration. GNP's ability to negotiate favorable contracts with these entities will be a key differentiator.
- Quality and Reliability: Consistent product quality and a reliable supply chain are paramount. Any disruption or quality issue could lead to significant loss of market share to competitors.
What are Potential Market Risks and Opportunities for GNP Fexofenadine HCl?
The market for GNP Fexofenadine HCl, like all generics, is subject to risks and presents certain opportunities. Understanding these factors is crucial for strategic planning.
Risks:
- Intensified Generic Competition: The entry of additional manufacturers into the fexofenadine HCl market could further drive down prices and reduce profit margins.
- Emergence of Newer Allergy Treatments: Development and market adoption of novel allergy treatments with superior efficacy or convenience could gradually erode the market share of older generics like fexofenadine HCl.
- Supply Chain Disruptions: Geopolitical events, raw material shortages, or manufacturing quality issues could disrupt supply and negatively impact sales.
- Regulatory Scrutiny: Increased regulatory oversight or changes in FDA guidelines could lead to increased compliance costs.
- PBM and Payer Pressure: Powerful Pharmacy Benefit Managers and insurance payers continue to exert pressure on drug prices, favoring the lowest-cost generics.
Opportunities:
- Market Penetration in Emerging Economies: Expanding market access in developing countries where healthcare infrastructure is improving and demand for affordable medications is rising presents an opportunity.
- Product Line Expansion: If GNP has capabilities, developing and marketing other strengths or related allergy medications could diversify its revenue streams.
- Strategic Partnerships: Forming partnerships with distributors or healthcare providers could enhance market reach and sales volume.
- Cost Optimization: Continuous improvement in manufacturing processes and supply chain efficiency can solidify GNP's position as a low-cost provider, enhancing its competitive advantage.
- Bundling or Combination Products: While less likely for a simple generic, exploring opportunities to bundle fexofenadine HCl with other OTC products (if regulations permit) could offer a slight competitive edge.
Key Takeaways
GNP Fexofenadine HCl operates in a mature, highly competitive generic drug market. Pricing is expected to remain stable with a downward bias due to ongoing competition from other generics and the absence of significant patent protections for the core molecule. Market demand is driven by the persistent prevalence of allergic conditions. GNP's success will hinge on its manufacturing efficiency, cost control, and ability to secure distribution channels. While significant price appreciation is unlikely, consistent sales volume can be achieved through strategic market positioning and reliable supply.
Frequently Asked Questions
1. What is the primary indication for fexofenadine HCl? Fexofenadine HCl is indicated for the relief of symptoms associated with seasonal allergic rhinitis, commonly known as hay fever, and chronic idiopathic urticaria (hives).
2. How does GNP Fexofenadine HCl compare in efficacy to its branded counterpart? As a generic drug approved by regulatory agencies like the FDA, GNP Fexofenadine HCl is required to be bioequivalent to the reference listed drug (e.g., Allegra). This means it has the same active ingredient, dosage strength, dosage form, and route of administration, and is expected to have the same therapeutic effect.
3. Are there any specific manufacturing advantages GNP might have for fexofenadine HCl? Specific manufacturing advantages would depend on GNP's internal processes, including proprietary synthesis routes, economies of scale in API production, or advanced formulation technologies. These details are typically proprietary business information.
4. What is the typical shelf life for GNP Fexofenadine HCl tablets? The typical shelf life for fexofenadine HCl tablets, similar to other oral solid dosage forms, is generally between 24 to 36 months from the date of manufacture, provided they are stored under recommended conditions.
5. How might changes in insurance formulary coverage impact the pricing of GNP Fexofenadine HCl? Changes in insurance formulary coverage, particularly decisions by Pharmacy Benefit Managers (PBMs) to favor lower-cost generics, can increase demand for specific generic products like GNP's. This increased volume can solidify or slightly improve a manufacturer's pricing position relative to higher-cost alternatives, but typically does not lead to outright price increases for the generic itself.
Citations
[1] Global Antihistamines Market Analysis and Forecast 2023-2028. (2023). Research and Markets. [2] Generic Drug Pricing Trends and Analysis. (2023). IQVIA Institute for Human Data Science. [3] FDA Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. (n.d.). U.S. Food and Drug Administration.
More… ↓
