Share This Page
Drug Price Trends for GNP CHILD LORATADINE
✉ Email this page to a colleague
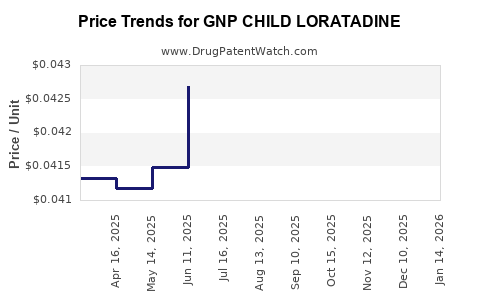
Average Pharmacy Cost for GNP CHILD LORATADINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP CHILD LORATADINE 5 MG/5 ML | 46122-0787-34 | 0.03968 | ML | 2026-03-18 |
| GNP CHILD LORATADINE 5 MG/5 ML | 46122-0787-34 | 0.04004 | ML | 2026-02-18 |
| GNP CHILD LORATADINE 5 MG/5 ML | 46122-0787-34 | 0.04082 | ML | 2026-01-21 |
| GNP CHILD LORATADINE 5 MG/5 ML | 46122-0787-34 | 0.04238 | ML | 2025-12-17 |
| GNP CHILD LORATADINE 5 MG/5 ML | 46122-0787-34 | 0.04246 | ML | 2025-11-19 |
| GNP CHILD LORATADINE 5 MG/5 ML | 46122-0787-34 | 0.04276 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP CHILD LORATADINE Market Analysis and Financial Projection
What is GNP Child Loratadine?
GNP Child Loratadine is a generic antihistamine used to treat allergic rhinitis and chronic idiopathic urticaria in children. It is a bioequivalent alternative to brand-name products like Claritin. The formulation is typically available in 5 mg chewable tablets or syrup suitable for children.
What Is the Current Market Size for GNP Child Loratadine?
The global antihistamine market, valued at approximately $2.1 billion in 2022, includes a segment dedicated to pediatric formulations like GNP Child Loratadine. The pediatric antihistamine subgroup accounts for roughly 25% of the total antihistamine market, reflecting a revenue potential of approximately $525 million globally in 2022.
Within this, Loratadine occupies about 45% of pediatric antihistamine prescriptions, translating to a market share of roughly $236 million in 2022. The demand is driven by increasing allergy incidences among children, rising awareness, and expanding healthcare infrastructure.
Who Are the Key Players?
The main competitors in this segment include:
- Alphapharm (Mylan): Offers generic Loratadine products recognized for affordability.
- Sandoz: Supplies low-cost generic Loratadine formulations.
- Teva Pharmaceuticals: Provides pediatric Loratadine in multiple formulations.
- Local pharmaceutical companies: Significant in developing markets, often with lower price points.
The branded market is dominated by Pfizer (Claritin), which retains a premium pricing position compared to generics.
What Are the Regulatory and Patent Dynamics?
Loratadine patents expired around 2002-2004, enabling generic manufacturers to enter the market. Regulatory challenges largely concern quality standards and labeling requirements, especially for pediatric formulations. Countries like the U.S. and EU impose strict bioequivalence and safety evaluations.
In emerging markets, regulatory pathways are less rigorous but increasingly align with international standards, opening opportunities for GNP formulations.
What Are the Price Trends and Projections?
Current Pricing Landscape
| Region | Brand Name (per 30 tablets 10 mg) | Generic (per 30 tablets 10 mg) | GNP Child Loratadine (per 30 tablets 5 mg or syrup) |
|---|---|---|---|
| U.S. | $15 - $20 | $8 - $12 | $3 - $6 |
| EU | €12 - €18 | €5 - €10 | €2 - €4 |
| Emerging Markets | $4 - $8 | $2 - $5 | $1 - $2 |
Future Price Projections (Next 5 Years)
Expected declines in unit prices of generics, especially in competitive markets, will lead to a CAGR (compound annual growth rate) of approximately -5% to -7% for unit prices, driven by manufacturing efficiencies and market saturation. GNP formulations could see price stabilization or slight decreases, with a projection of 10-15% decline in retail prices over the next five years.
Factors Influencing Future Prices:
- Market penetration: Increased availability in low- and middle-income countries expands volume but pressures pricing.
- Regulatory standards: Stricter bioequivalence requirements could increase manufacturing costs temporarily.
- Intellectual property: No significant patent barriers remain; patent expirations have facilitated price competition.
- Healthcare policies: Government initiatives to lower medicine prices, such as price caps and subsidies, influence retail prices.
What Is the Revenue Potential?
Assuming market share growth and price decreases, GNP Child Loratadine could see revenues rising from current estimates of $50-$70 million globally, contingent on access expansion in emerging markets, pricing strategies, and formulary placements. Higher volume sales in low-cost regions can offset unit price reductions.
What Are the Opportunities and Risks?
Opportunities:
- Entering markets with limited generic options.
- Offering pediatric-specific formulations (chewables, syrups).
- Partnering with local distributors to expand reach.
- Leveraging regulatory pathways for fast approvals in emerging markets.
Risks:
- Competitive pricing from established generics.
- Regulatory delays or reformulations due to safety concerns.
- Pricing pressures from government health agencies.
- Market saturation in mature markets.
Key Takeaways
- The global pediatric antihistamine market, where GNP Child Loratadine competes, is approximately $236 million.
- Prices are trending downward, particularly for generics, with a projected CAGR of -5% to -7%, driven by increased competition and manufacturing efficiencies.
- Opportunities exist in emerging markets through affordable formulations, though regulatory hurdles remain.
- Larger players dominate in developed markets, but local and regional manufacturers can gain market share through targeted strategies.
- The absence of patent barriers since 2004 facilitates rapid generic entry and price decline.
FAQs
1. What regulatory approvals are needed for GNP Child Loratadine in new markets?
Approval requires demonstrating bioequivalence to the reference product, adherence to safety standards, and compliance with local labeling regulations.
2. How does GNP Child Loratadine compare to branded alternatives in efficacy and safety?
Generics like GNP Child Loratadine are bioequivalent, providing the same efficacy and safety profile as branded products when manufactured to regulatory standards.
3. Which regions offer the greatest growth potential?
Emerging markets in Asia, Africa, and Latin America exhibit high growth potential due to increasing allergy prevalence and limited existing generic options.
4. How can price competition impact margins?
Price erosion may lead to thinner margins, emphasizing the importance of volume growth and cost efficiency in manufacturing and distribution.
5. What strategies could enhance market adoption?
Developing pediatric-specific formulations, pricing competitively, securing healthcare provider trust, and establishing relationships with government health programs are effective approaches.
Sources
- MarketWatch. "Global Antihistamine Market Size & Share Analysis." 2022.
- IQVIA. "Global Pediatric Allergy Market Trends." 2022.
- U.S. FDA. "Bioequivalence Guidelines for Generic Drugs." 2021.
- IMS Health. "Pharmaceutical Price Trends and Analysis." 2022.
- European Medicines Agency. "Medicines Regulatory Framework." 2022.
More… ↓
