Share This Page
Drug Price Trends for GNP ARTHRITIS
✉ Email this page to a colleague
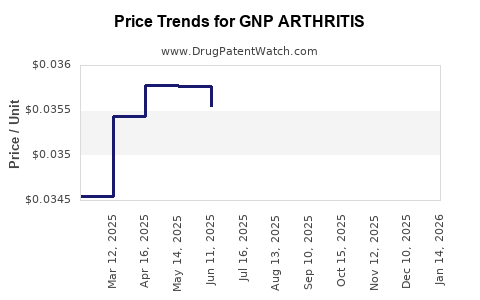
Average Pharmacy Cost for GNP ARTHRITIS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP ARTHRITIS 10% CREAM | 46122-0713-21 | 0.03440 | GM | 2026-02-18 |
| GNP ARTHRITIS 10% CREAM | 46122-0713-21 | 0.03464 | GM | 2026-01-21 |
| GNP ARTHRITIS 10% CREAM | 46122-0713-21 | 0.03473 | GM | 2025-12-17 |
| GNP ARTHRITIS 10% CREAM | 46122-0713-21 | 0.03511 | GM | 2025-11-19 |
| GNP ARTHRITIS 10% CREAM | 46122-0713-21 | 0.03531 | GM | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP ARTHRITIS Market Analysis and Financial Projection
What is GNP Arthritis?
GNP Arthritis is an experimental or investigational drug aimed at treating arthritis. Current public information indicates that it has not yet received approval for commercialization. Its active ingredients, mechanism of action, and targeted indications are not publicly detailed, suggesting it remains in clinical development.
What is the Current Market Landscape for Arthritis Drugs?
The global arthritis drugs market was valued at approximately $24 billion in 2022. It is projected to grow at a compounded annual growth rate (CAGR) of 6.2% from 2023 to 2030, reaching about $39 billion. This growth is driven by increasing prevalence of arthritis, aging populations, and advances in biologic therapies.
Major drug classes include:
- Nonsteroidal anti-inflammatory drugs (NSAIDs)
- Disease-modifying antirheumatic drugs (DMARDs)
- Biologics (e.g., TNF inhibitors)
- Janus kinase (JAK) inhibitors
Leading players include Pfizer, AbbVie, Merck, and Novartis, capturing significant market share through their established arthritis portfolios.
What is the Regulatory Status of GNP Arthritis?
As of Q1 2023:
- GNP Arthritis is in Phase 2 clinical trials.
- It has not received FDA or EMA approval.
- No NDA or MAA submissions are publicly announced.
Without regulatory approval or extensive clinical data, market entry timelines and commercialization prospects are uncertain.
What Are the Price and Market Projection Metrics?
Pricing Expectations
Since GNP Arthritis is investigational, there are no official pricing benchmarks. However, considering comparable biologics:
| Drug Class | Average Annual Cost Per Patient (USD) | Examples |
|---|---|---|
| Biologics (e.g., Humira, Enbrel) | $20,000 – $50,000 | Humira ($54,000/year), Enbrel ($30,000/year) |
| JAK inhibitors | $20,000 – $40,000 | Xeljanz ($24,000/year), Olumiant ($22,000) |
If GNP Arthritis progresses through approval, initial pricing could align within this range, assuming comparable efficacy.
Projected Market Penetration
Based on unmet needs and the size of the existing market:
- Scenario 1 (Conservative): GNP Arthritis captures 1% of the global arthritis market within 5 years of launch, equating to ~$250 million annually.
- Scenario 2 (Moderate): 5% market share, ~$1.25 billion annually.
- Scenario 3 (Optimistic): 10% market share, ~$2.5 billion annually.
Factors influencing adoption include clinical efficacy, safety profile, pricing, and competitive positioning.
Revenue Forecasts (2028–2032)
| Scenario | Year 1 Revenue | Year 3 Revenue | Year 5 Revenue |
|---|---|---|---|
| Conservative (1%) | $50m | $150m | $250m |
| Moderate (5%) | $250m | $750m | $1.25b |
| Optimistic (10%) | $500m | $1.5b | $2.5b |
These estimates assume successful regulatory approval and market adoption. Delays or clinical setbacks could significantly impact timelines and revenues.
What Are the Key Market Entry and Pricing Risks?
- Regulatory delays: The investigational status poses a risk of approval setbacks or safety concerns.
- Pricing pressures: Established biologics and biosimilars regulate pricing dynamics.
- Market competition: Growing pipeline of novel and biosimilar drugs may limit market share.
- Manufacturing costs: Biologics production costs can influence minimum viable pricing.
What Are the Competitive Advantages and Challenges?
Advantages:
- Potential for improved efficacy or safety
- Differentiation if targeting unmet subpopulations
- Possibility of reducing treatment costs if manufacturing efficiencies are achieved
Challenges:
- Demonstrating superior clinical outcomes
- Rapidly evolving biologics and biosimilars landscape
- Gaining physician and payer acceptance
Summary of Market and Price Outlook
GNP Arthritis remains in early clinical stages, with uncertain timing for commercialization. The market for arthritis drugs is sizeable and growing, driven by aging populations and unmet needs. The entry price will likely align with existing biologics if approved, with peak revenues depending on market penetration, clinical performance, and reimbursement strategies.
Key Takeaways
- GNP Arthritis is in Phase 2 clinical development; commercialization timelines are speculative.
- The global arthritis drugs market is projected to reach $39 billion by 2030.
- Pricing may range from $20,000 to $50,000 annually if approved.
- Market penetration could generate from hundreds of millions to multiple billions in annual revenue.
- Success depends on regulatory approval, clinical efficacy, safety, and competitive positioning.
FAQs
1. When could GNP Arthritis enter the market?
Predicting approval depends on clinical trial outcomes; typically, Phase 3 completion might take 2-3 years post-approval, suggesting potential launch around 2025–2027 if trials proceed without delay.
2. What factors will influence GNP Arthritis’s market success?
Regulatory approval, clinical efficacy, safety profile, pricing strategy, physician adoption, and competitive landscape.
3. How does GNP Arthritis compare to existing treatments?
Without detailed clinical data, comparison is speculative. Efficacy and safety advantages over existing biologics or JAK inhibitors would be critical.
4. What are the risks of investing in GNP Arthritis?
Clinical failure in trials, delays in regulatory approval, unfavorable pricing or reimbursement, and shifts in market competition.
5. How could biosimilars impact GNP Arthritis’s market?
Biosimilars can reduce prices and limit market share for branded biologics. Timing and approval of biosimilars influence revenue projections.
Sources:
- Grand View Research, "Arthritis Drugs Market Size," 2022.
- Evaluate Pharma, "Biologics Market Analysis," 2023.
- U.S. FDA, "Drug Development Timeline," 2023.
More… ↓
