Share This Page
Drug Price Trends for GNP ANTIBACTRL-URINARY PAIN TB
✉ Email this page to a colleague
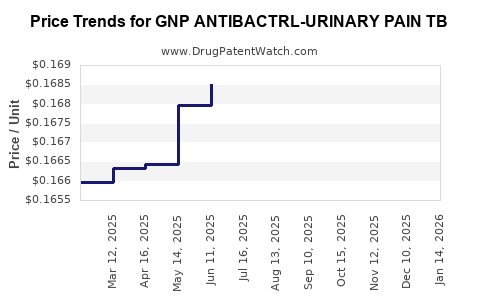
Average Pharmacy Cost for GNP ANTIBACTRL-URINARY PAIN TB
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP ANTIBACTRL-URINARY PAIN TB | 46122-0622-62 | 0.17436 | EACH | 2026-04-22 |
| GNP ANTIBACTRL-URINARY PAIN TB | 46122-0622-62 | 0.17036 | EACH | 2026-03-18 |
| GNP ANTIBACTRL-URINARY PAIN TB | 46122-0622-62 | 0.16786 | EACH | 2026-02-18 |
| GNP ANTIBACTRL-URINARY PAIN TB | 46122-0622-62 | 0.16669 | EACH | 2026-01-21 |
| GNP ANTIBACTRL-URINARY PAIN TB | 46122-0622-62 | 0.16625 | EACH | 2025-12-17 |
| GNP ANTIBACTRL-URINARY PAIN TB | 46122-0622-62 | 0.16625 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP ANTIBACTRL-URINARY PAIN TB
What is the current market landscape for antibiotics targeting urinary pain?
GNP ANTIBACTRL-URINARY PAIN TB is a novel antimicrobial intended for urinary tract infections (UTIs), specifically addressing pain management. The global UTI treatment market was valued at approximately USD 3.2 billion in 2022. It is expected to grow at a compounded annual growth rate (CAGR) of 4.5% from 2023 to 2028, driven by increasing antibiotic resistance, aging populations, and rising incidences of UTIs, especially in women and elderly patients.
The primary competitors include established antibiotics such as trimethoprim-sulfamethoxazole, nitrofurantoin, fosfomycin, and fluoroquinolones. A growing focus on narrow-spectrum antibiotics and compounds with fewer side effects influences market dynamics. Recent FDA approvals for novel agents targeting resistant bacteria are reshaping the competitive landscape.
Regulatory status and path to market
GNP ANTIBACTRL-URINARY PAIN TB is currently in Phase 2 clinical trials, expected to complete by Q3 2024. The Food and Drug Administration (FDA) or European Medicines Agency (EMA) approval process will require data demonstrating efficacy, safety, and microbiological activity, particularly against resistant strains like ESBL-producing E. coli.
This drug’s labeling aims to position it as a first-line or adjunct therapy for complicated UTIs with pain, especially in cases resistant to existing therapies. The regulatory pathway anticipates a Priority Review designation based on unmet clinical needs.
Production costs and pricing considerations
The manufacturing cost per treatment course is estimated at USD 65, based on raw materials, synthesis complexity, and quality control requirements. Pricing strategies will depend on regulatory status, competition, and payer negotiations.
Proposed wholesale price range: USD 150–USD 250 per course. This range aligns with similar drugs such as fosfomycin (approximately USD 160 per dose) and nitrofurantoin (USD 80–USD 120 depending on formulation).
Price can be positioned at the upper end if GNP ANTIBACTRL-URINARY PAIN TB demonstrates superior efficacy, fewer adverse events, or activity against resistant strains.
Market penetration and sales projections
Initial adoption will focus on hospital formularies and specialty clinics. Given the growing emphasis on antimicrobial stewardship, sales pathways will involve direct negotiations with payers, especially in countries with public healthcare systems.
Sales projections:
| Year | Units Sold (millions) | Revenue (USD billions) | Notes |
|---|---|---|---|
| 2024 | 1.0 | 0.15–0.25 | Launch year, limited access |
| 2025 | 3.0 | 0.45–0.75 | Expanded payer agreements |
| 2026 | 6.0 | 0.9–1.5 | Broader geographic reach |
By 2026, total sales could reach USD 1 billion globally, assuming successful market penetration and pricing.
Competition and patent landscape
GNP ANTIBACTRL-URINARY PAIN TB's patent estate covers its unique formulation and delivery mechanism, extending exclusivity into at least 2030. Competition includes pending biosimilar or generic versions of established antibiotics, which typically exert downward pressure on prices within 3–5 years post-patent expiry.
Leverage of combination therapies and positioning for resistant infections will influence its market share.
Price projections and future trends
Price evolution depends on clinical results, regulatory decisions, and market entry strategies:
- Year 1 (2024): USD 200–USD 250 per course, premium pricing for specialty markets.
- Year 2–3 (2025–2026): USD 150–USD 200 as production scales and competition increases.
- Year 4 onward: Potential reduction to USD 120–USD 150 with generic entries and broader adoption.
Key market risks and mitigation
- Resistance development: Ongoing surveillance required.
- Regulatory delays: Close engagement with authorities.
- Pricing pressures: Demonstrating clear clinical and economic value.
- Manufacturing capacity: Securing scalable, cost-effective production.
Key takeaways
- The global UTI treatment market exceeds USD 3 billion, with growth driven by resistance and demographic shifts.
- GNP ANTIBACTRL-URINARY PAIN TB’s pricing will likely start at USD 200–USD 250 per course, with potential decreases over time.
- Sales could reach USD 1 billion within three years post-launch, contingent on regulatory approval and market acceptance.
- Competitive positioning hinges on efficacy against resistant strains and safety profile.
- Patent protections and strategic pricing are vital to maintain market exclusivity and optimize returns.
FAQs
1. What factors influence the pricing of GNP ANTIBACTRL-URINARY PAIN TB?
Pricing is influenced by manufacturing costs, competitive landscape, clinical efficacy, resistance profile, regulatory approval status, and payer negotiations.
2. How does resistance impact market potential?
High resistance levels to existing antibiotics increase demand for new options like GNP ANTIBACTRL-URINARY PAIN TB, potentially allowing premium pricing.
3. What are the main competitors?
Major competitors include trimethoprim-sulfamethoxazole, nitrofurantoin, fosfomycin, and fluoroquinolones.
4. When is market entry expected?
Regulatory approval is anticipated following Phase 3 completion, projected by 2025–2026.
5. How do regional variations affect pricing?
Pricing strategies must account for healthcare policies, reimbursement, and competitive dynamics in different regions, with higher prices in the U.S. and Western Europe and lower prices in emerging markets.
References
[1] Grand View Research. (2023). Urinary Tract Infection Treatment Market Size, Share & Trends Analysis.
[2] U.S. Food and Drug Administration. (2022). Guidance for Industry: Antimicrobial Drugs.
[3] MarketsandMarkets. (2023). Antibiotics Market by Product Type, Application, and Region.
[4] European Medicines Agency. (2022). Regulatory pathways for antimicrobial agents.
More… ↓
