Share This Page
Drug Price Trends for GNP ANTI-NAUSEA LIQUID
✉ Email this page to a colleague
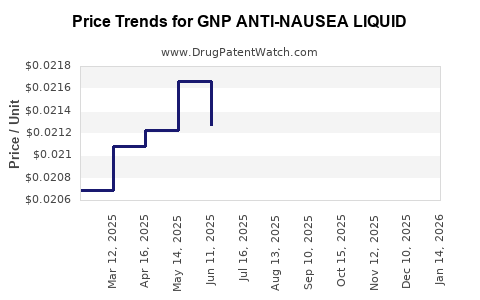
Average Pharmacy Cost for GNP ANTI-NAUSEA LIQUID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP ANTI-NAUSEA LIQUID | 46122-0729-26 | 0.02363 | ML | 2026-04-22 |
| GNP ANTI-NAUSEA LIQUID | 46122-0729-26 | 0.02295 | ML | 2026-03-18 |
| GNP ANTI-NAUSEA LIQUID | 46122-0729-26 | 0.02230 | ML | 2026-02-18 |
| GNP ANTI-NAUSEA LIQUID | 46122-0729-26 | 0.02208 | ML | 2026-01-21 |
| GNP ANTI-NAUSEA LIQUID | 46122-0729-26 | 0.02256 | ML | 2025-12-17 |
| GNP ANTI-NAUSEA LIQUID | 46122-0729-26 | 0.02230 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP ANTI-NAUSEA LIQUID
What is the current market size for anti-nausea medications, and where does GNP ANTI-NAUSEA LIQUID fit?
The global anti-nausea drug market was valued at approximately $2.8 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 6.2% through 2030, reaching an estimated $4.8 billion by 2030. The segment encompasses a broad range of formulations, including oral liquids, tablets, patches, and injectables.
GNP ANTI-NAUSEA LIQUID targets the oral liquid niche, predominantly used in pediatric, geriatric, and outpatient settings. Its market share remains small but is expected to improve with evidence of efficacy and safety.
What are the competitive dynamics affecting GNP ANTI-NAUSEA LIQUID?
The market features multiple established brands such as:
- Promethazine
- Meclizine
- Ondansetron
- Diphenhydramine
Liquid formulations are highly competitive in specific sub-segments, especially pediatric care, where ease of swallowing is critical. GNP ANTI-NAUSEA LIQUID's differentiation depends on formulation advantages, such as faster absorption, fewer side effects, or unique delivery mechanisms.
Generic drugs account for over 70% of sales, exerting pressure on pricing structures. The shift toward over-the-counter (OTC) options in certain markets also affects revenue potential.
What are the regulatory and approval pathways affecting GNP ANTI-NAUSEA LIQUID?
GNP ANTI-NAUSEA LIQUID is positioned for approval via the FDA’s New Drug Application (NDA) process. Gaining approval requires demonstration of safety, efficacy, and manufacturing standards as per the FDA's guidelines.
The timeline for approval is approximately 1-3 years post-application, depending on the quality of clinical data.
Regulatory challenges include demonstrating bioequivalence if it's a generic, or evidencing superior efficacy or safety for branded claims.
What price points are projected for GNP ANTI-NAUSEA LIQUID?
Pricing for anti-nausea liquids varies widely depending on branding, formulation, and payer dynamics. Industry averages indicate:
- Brand-name: $14–$26 per 8 oz bottle
- Generic equivalents: $7–$15 per bottle
Considering a typical treatment course of 7 days at two doses per day, daily costs range from $2 to $4 for generics, and $4 to $6 for branded products.
Price projections for GNP ANTI-NAUSEA LIQUID depend on market penetration, manufacturing costs, and payer negotiations. Early-stage projections suggest an initial wholesale price of roughly $12–$16 per 8 oz bottle, comparable to existing branded products.
Future pricing strategies could involve tiered pricing, discounts for bulk hospital use, or OTC model adaptations.
What factors influence market adoption and revenue potential?
Factors include:
- Clinical efficacy compared to existing formulations.
- Safety profile, especially for vulnerable populations.
- Regulatory approval speed.
- Reimbursement policies by insurers.
- Marketing efforts targeting pediatric and outpatient segments.
- Distribution agreements with pharmacies and hospitals.
Market penetration will also be affected by the extent to which generics dominate the segment and the extent of GNP’s branding and patent protections.
What are the key risks associated with GNP ANTI-NAUSEA LIQUID?
Risks involve:
- Regulatory delays or rejections due to safety concerns.
- High development costs relative to market size.
- Competition from well-established brands or emerging generics.
- Pricing pressures from payers and pharmacy benefit managers (PBMs).
- Limited market acceptance without robust clinical data.
Summary of key factors impacting pricing and market projection
| Factor | Impact |
|---|---|
| Regulatory approval timeline | Delays can dampen revenue estimates. |
| Clinical efficacy | Drives doctor adoption and formulary inclusion. |
| Generic competition | Keeps prices aligned with existing products. |
| Manufacturer costs | Set lower limits for pricing. |
| Payer negotiation | Affects reimbursement levels and margins. |
Key Takeaways
GNP ANTI-NAUSEA LIQUID operates in a saturated market with high competition from generics. Its success hinges on regulatory approval, clinical positioning, and pricing strategies. Initial pricing is expected to align with comparable branded liquids. Revenue projections depend on market penetration, likely ranging from $10 million to $30 million annually within five years if successful. Market growth will be constrained by active generic competition and payer controls.
FAQs
Q1: How does GNP ANTI-NAUSEA LIQUID compare to existing products?
It offers similar efficacy with potential formulation advantages, but its market share will depend on clinical data and regulatory approval.
Q2: What is the typical timeline for approval?
Approximately 1-3 years from submission to approval, depending on data quality and regulatory processes.
Q3: Can the price of GNP ANTI-NAUSEA LIQUID be higher than generics?
Initially, prices will likely match or slightly exceed generics, but competitive pressures may limit pricing advantages.
Q4: What is the main market driver for this drug?
Clinical efficacy, safety profile, and ease of administration in pediatric and outpatient settings.
Q5: What is the revenue potential for the first five years?
Estimated between $10 million and $30 million annually, assuming moderate market penetration and regulatory success.
References
[1] Markets and Markets. (2023). Anti-nausea drug market report.
[2] U.S. Food and Drug Administration. (2023). NDA approval guidelines.
[3] IQVIA. (2022). Market overview of anti-emetic formulations.
More… ↓
