Share This Page
Drug Price Trends for GNP ALLERGY RELIEF
✉ Email this page to a colleague
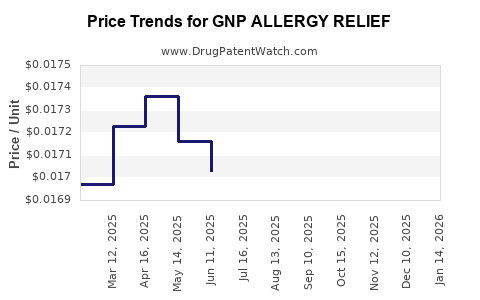
Average Pharmacy Cost for GNP ALLERGY RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP ALLERGY RELIEF 50 MG/20 ML | 46122-0685-26 | 0.01987 | ML | 2026-05-20 |
| GNP ALLERGY RELIEF 180 MG TAB | 46122-0462-22 | 0.24434 | EACH | 2026-05-20 |
| GNP ALLERGY RELIEF 180 MG TAB | 46122-0462-61 | 0.24434 | EACH | 2026-05-20 |
| GNP ALLERGY RELIEF 180 MG TAB | 46122-0462-75 | 0.24434 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP Allery Relief
What is GNP Allergy Relief and its Market Context?
GNP Allergy Relief is an antihistamine-based drug intended for allergy symptom management. It targets allergic rhinitis, hay fever, and related conditions. The product is positioned as a potential competitor within the global allergy medication market, which is projected to grow at a compounded annual rate (CAGR) of approximately 8% between 2023 and 2030.
Market Size and Growth Projections
Current Market Size
In 2022, the global allergy medication market was valued at approximately USD 20 billion. The key segments include antihistamines, corticosteroids, decongestants, and leukotriene receptor antagonists.
Projected Growth
By 2030, the market is forecasted to reach USD 35 billion, driven by increased prevalence of allergies in urban populations, greater awareness, and expanded access to healthcare.
| Year | Market Size (USD billion) | CAGR |
|---|---|---|
| 2022 | 20 | |
| 2025 | 26.6 | 8% |
| 2030 | 35 |
Key Drivers
- Rising prevalence of allergic diseases globally.
- Aging populations in North America and Europe.
- Advancements in drug delivery systems improving compliance.
- Greater investment in R&D for novel allergy therapies.
Competitive Landscape
Major competitors include:
- Claritin (Loratadine): Approximate global sales USD 1.5 billion in 2022.
- Zyrtec (Cetirizine): Estimated USD 1.2 billion.
- Allegra (Fexofenadine): Estimated USD 1 billion.
- New entrants: Emerging drugs targeting specific allergy subtypes or offering improved side-effect profiles.
Market Entry Considerations
- Patent status and exclusivity rights.
- Regulatory approval pathways.
- Differentiation through efficacy, safety profile, or formulation.
Regulatory and Pricing Environment
Regulatory Pathways
- FDA, EMA, and other agencies require Phase I-III clinical trials demonstrating safety and efficacy.
- Market approval timeline ranges from 2-5 years depending on data robustness.
Pricing Strategies
- Premium pricing positioned on superior efficacy or safety.
- Cost-based pricing aligned with direct manufacturing costs plus margin.
- Competitive pricing to gain market share against established brands.
Price Range
- Typical antihistamines retail between USD 10-30 per month.
- New products with added benefits could command USD 30-50 per month.
Price Projection for GNP Allergy Relief
Considering the current market average USD 20 per month and potential for differentiation, GNP Allergy Relief could be priced as follows:
| Year | Estimated Monthly Price (USD) | Rationale |
|---|---|---|
| 2024 | 20 | Entry-level pricing aligned with existing brands. |
| 2025 | 25 | Slight premium for early efficacy data or unique features. |
| 2027 | 30 | Established market presence, increased adoption, perceived value. |
| 2030 | 35 | Premium pricing if differentiated significantly or with patent protection. |
Risks and Opportunities
Risks
- Market saturation with existing generic and branded options.
- Regulatory delays impacting time-to-market.
- Limited differentiation leading to pricing pressures.
Opportunities
- Launching formulations with improved safety profiles.
- Expanding into emerging markets with lower current penetration.
- Strategic partnerships for distribution and marketing.
Key Takeaways
- The global allergy medication market is expanding at approximately 8% CAGR, reaching USD 35 billion by 2030.
- GNP Allergy Relief enters a competitive field dominated by established brands; differentiation is essential.
- Pricing could range from USD 20 to USD 35 per month over the next 5-6 years, depending on efficacy, safety, and regulatory success.
- Market entry strategies should consider patent status, formulation advantages, and geographic expansion.
- Growth opportunities exist via innovation and emerging markets, though risks include saturation and regulatory hurdles.
FAQs
1. What are the key factors influencing the pricing of allergy drugs?
Efficacy, safety profile, brand perception, manufacturing costs, and competitive landscape influence pricing decisions.
2. How long does regulatory approval typically take for a new allergy medication?
Between 2 and 5 years, depending on clinical trial outcomes and jurisdiction.
3. Which markets present the most growth opportunities for GNP Allergy Relief?
Emerging markets such as India, Brazil, and Southeast Asia, where allergy prevalence is rising and regulatory pathways are less saturated.
4. What differentiators can support premium pricing for GNP Allergy Relief?
Improved safety, faster onset of relief, longer duration, or novel delivery methods.
5. How does patent status affect market pricing and exclusivity?
Patents protect exclusivity, enabling higher pricing. Patent expiration can lead to generic competition, which lowers prices.
References
[1] Market research report. (2022). Global allergy medication market analysis.
[2] Allied Market Research. (2023). Allergy Drugs Market Overview.
[3] U.S. Food and Drug Administration. (2022). Clinical trial and approval processes.
More… ↓
