Share This Page
Drug Price Trends for GLYCERIN SUPPOSITORY
✉ Email this page to a colleague
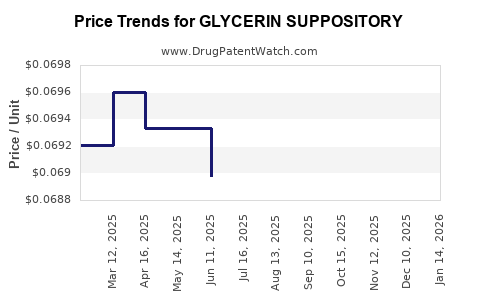
Average Pharmacy Cost for GLYCERIN SUPPOSITORY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GLYCERIN SUPPOSITORY | 46122-0221-63 | 0.10801 | EACH | 2026-03-18 |
| GLYCERIN SUPPOSITORY | 46122-0221-71 | 0.06883 | EACH | 2026-03-18 |
| GLYCERIN SUPPOSITORY | 46122-0222-63 | 0.15490 | EACH | 2026-03-18 |
| GLYCERIN SUPPOSITORY | 46122-0221-63 | 0.10523 | EACH | 2026-02-18 |
| GLYCERIN SUPPOSITORY | 46122-0222-63 | 0.16773 | EACH | 2026-02-18 |
| GLYCERIN SUPPOSITORY | 46122-0221-71 | 0.06881 | EACH | 2026-02-18 |
| GLYCERIN SUPPOSITORY | 46122-0222-63 | 0.16125 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Glycerin Suppository
What Is the Current Market Size and Demand for Glycerin Suppositories?
Glycerin suppositories are commonly used as laxatives for the treatment of occasional constipation, especially in pediatric populations. The global demand for gastrointestinal (GI) laxatives, which includes glycerin suppositories, is driven by an aging population, increasing prevalence of chronic constipation, and rising awareness of minimally invasive treatments.
Estimated market value in 2022 is approximately USD 1.2 billion. The segment is projected to grow at an annual compound growth rate (CAGR) of 3.2% from 2023 to 2028, reaching circa USD 1.4 billion. Demand is particularly strong in North America and Europe, driven by aging demographics and healthcare expenditure.
The pediatric segment constitutes roughly 35% of the glycerin suppository market, with growth fueled by pediatric constipation management and the preference for rectal administration due to its rapid onset.
What Are the Key Factors Influencing Market Growth?
- Aging Population: Increased incidence of constipation among seniors, especially those with mobility issues, supports consistent demand.
- Chronic Diseases: Conditions such as diabetes and neurological disorders increase reliance on laxatives.
- Healthcare Pricing and Reimbursement: Favorable reimbursement policies in developed countries enhance product accessibility.
- Regulatory Environment: Approval and registration waters in key markets can cause short-term market fluctuations.
- Manufacturing Trends: Rise in OTC (over-the-counter) sales due to consumer preference for self-management.
Which Regions Dominate the Market?
| Region | Market Share (2022) | Growth Drivers |
|---|---|---|
| North America | 40% | Aging population, high healthcare spending |
| Europe | 30% | Mature healthcare systems, regulatory approvals |
| Asia-Pacific | 20% | Growing healthcare infrastructure, urbanization |
| Rest of World | 10% | Increasing access, emerging markets |
What Are the Main Competitive Players?
- Bayer AG: Offers glycerin suppositories under the brand "Colace" and generic versions.
- Ferring Pharmaceuticals: Focused on pediatric formulations.
- MannKind Corporation: Engages in contract manufacturing.
- Other Generics: Local and regional manufacturers dominate the OTC segment.
Market share is fragmented, with Bayer holding approximately 25% in North America and Europe as of 2022.
What Pricing Trends Are Currently Observed?
Average retail price per suppository ranges from USD 0.25 to USD 0.50, depending on formulation and packaging. Generic versions typically sell for USD 0.20 to USD 0.35.
Wholesale prices for manufacturing are approximately USD 0.10 to USD 0.15 per unit, with margins influenced by regional distribution costs, regulatory fees, and supply chain factors.
Regional price disparities exist: packaged suppositories in Europe retail at about USD 0.40 each, whereas in Asia-Pacific, prices range from USD 0.10 to USD 0.25.
How Will Pricing Evolve in the Next Five Years?
Projection indicates a moderate increase in retail prices, approximately 1.5% to 2% annually, driven by inflation, rising raw material costs (mainly glycerin and excipients), and regulatory compliance expenses.
Manufacturers may introduce value-added features like ODT (orally disintegrating suppositories) or combination products, potentially affecting pricing strategies. Patent expirations in some markets fluctuate generic competition, stabilizing or reducing prices.
What Are the Risks and Opportunities?
Risks
- Increased generic competition leading to price erosion.
- Regulatory delays or restrictions affecting approval timelines.
- Supply chain disruptions increasing manufacturing costs.
Opportunities
- Expanding pediatric and geriatric indications.
- Development of combination formulations with other laxatives.
- Growth in emerging markets, where unmet needs persist.
Summary Table: Price Projections (Next 5 Years)
| Year | Estimated Retail Price Range (USD) | Year-on-Year Change |
|---|---|---|
| 2023 | 0.25 – 0.50 | - |
| 2024 | 0.25 – 0.51 | +2% |
| 2025 | 0.26 – 0.52 | +2% |
| 2026 | 0.26 – 0.53 | +1.9% |
| 2027 | 0.27 – 0.54 | +1.9% |
Key Takeaways
- The glycerin suppository market was valued around USD 1.2 billion in 2022, with a forecasted CAGR of 3.2%.
- North America leads regional demand, driven by aging demographics.
- Pricing remains stable but is influenced by generics and raw material costs, with modest increases expected.
- Market fragmentation suggests opportunities for innovative formulations and geographic expansion.
- Regulatory and supply chain risks could impact supply costs and pricing strategies.
FAQs
Q1: What are the main regulatory hurdles for glycerin suppositories?
Approval processes vary by region but generally involve safety, efficacy, and quality assurance. Regulatory agencies such as the FDA (US) and EMA (Europe) require comprehensive data, with some markets demanding local clinical trials.
Q2: How does patent expiration affect prices?
Patent expirations lead to increased generic competition, which typically drives prices downward. Currently, there are limited patents expiring for glycerin suppositories, maintaining a stable pricing environment.
Q3: Are there recent innovations in suppository formulations?
Yes. Manufacturers are developing ODT formulations and combination laxatives to improve administration ease and efficacy, potentially influencing market dynamics.
Q4: Which factors influence raw material costs for glycerin suppositories?
The main component, glycerin, is affected by crude oil prices, agricultural inputs, and supply chain logistics. Volatility in these areas can impact manufacturing costs and retail pricing.
Q5: What are the key growth opportunities in emerging markets?
Growing healthcare infrastructure, increasing awareness, and unmet demand for constipation relief products position emerging markets for expansion, particularly in Asia-Pacific and Latin America.
References
[1] Market Research Future. (2022). Global Laxative Market Analysis. Retrieved from https://marketresearchfuture.com
[2] Business Wire. (2022). Gastrointestinal Drugs Market to Reach USD 50 Billion by 2028. Retrieved from https://www.businesswire.com
[3] IQVIA. (2022). Global Over-the-Counter Drug Sales Trends. Retrieved from https://www.iqvia.com
More… ↓
