Last updated: February 14, 2026
Market Overview
GENTLE LAXATIVE is positioned in the over-the-counter (OTC) laxative segment, which has shown consistent growth globally. The increasing prevalence of constipation, driven by aging populations, sedentary lifestyles, and dietary habits, sustains demand.
Current Market Size and Growth
- The global laxative market was valued at approximately $3.6 billion in 2022.
- Projected Compound Annual Growth Rate (CAGR): 4.5% (2023-2028).
- Major markets: North America (45% share), Europe (25%), Asia-Pacific (20%), Rest of World (10%).
Competitive Landscape
Leading players include Johnson & Johnson, Procter & Gamble, and Sanofi. The market features a mix of traditional and novel formulations, including fiber-based, osmotic, saline, stimulant, and stool softeners.
Regulatory Environment
GENTLE LAXATIVE, as an OTC product, must comply with FDA regulations in the U.S., EMA standards in Europe, and equivalent authorities elsewhere. Regulatory approval standards influence formulation, labeling, and marketing.
Pricing Dynamics
- Average retail price for OTC laxatives: $5–$12 per package.
- Pricing varies based on formulation, dosage, pack size, and brand positioning.
- Private labels generally price 10–20% lower than brand-name equivalents.
Market Entry Considerations
- Brand differentiation: Emphasizing "gentle" effects and natural ingredients can influence pricing and market penetration.
- Distribution channels: Pharmacies, supermarkets, and online platforms are primary channels.
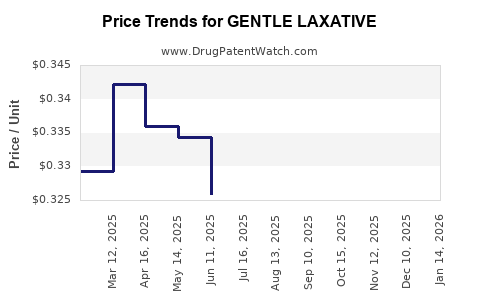
Price Projections (2023–2028)
| Year |
Average Price per Package |
Key Influencing Factors |
| 2023 |
$8.00 |
Current pricing trends, market competition, inflation |
| 2024 |
$8.30 |
Slight inflation, new product launches, ingredient costs |
| 2025 |
$8.65 |
Increased marketing, formulation innovation, packaging costs |
| 2026 |
$9.00 |
Higher penetration in emerging markets, regulatory costs |
| 2027 |
$9.40 |
Brand expansion, inflation, supply chain stabilization |
| 2028 |
$9.80 |
Market maturity, premium product positioning |
Factors Affecting Price Trends
- Ingredient Costs: Fluctuations due to supply chain constraints or raw material prices.
- Regulatory Changes: Stricter standards may incur additional costs.
- Competition: Entry of generic or new formulations can drive prices down.
- Consumer Preferences: Preference for natural or organic ingredients can command premium pricing.
Potential Market Risks
- Increased regulation could elevate compliance costs.
- Market saturation may limit price increases.
- Price wars among brands could compress margins.
Key Competitors' Pricing Strategies
- Johnson & Johnson's Dulcolax: Approx. $9–$12 per box.
- P&G’s Metamucil: Similar price range, targeting health-conscious consumers.
- Sanofi’s Fleet: Slightly lower pricing to penetrate budget segments.
Conclusions
GENTLE LAXATIVE's price is expected to rise modestly over the next five years, aligned with inflation and product differentiation strategies. To optimize profitability, branding efforts emphasizing "gentleness" and natural ingredients are advisable.
Key Takeaways
- The global OTC laxative market is valued at $3.6 billion, with a 4.5% CAGR forecast to 2028.
- Average retail prices are projected to increase from $8.00 in 2023 to nearly $9.80 in 2028.
- Price trends are influenced by ingredient costs, regulatory standards, competition, and consumer preferences.
- Differentiation through product attributes can support premium pricing.
- Market risks include regulation shifts and competitive pressures.
FAQs
1. What are the main factors influencing the pricing of GENTLE LAXATIVE?
Ingredient costs, regulatory compliance, competition, marketing strategies, and consumer demand for natural ingredients.
2. How does the OTC market impact the pricing of laxatives?
OTC market competition and brand positioning directly influence retail prices, with private labels typically priced lower than brand names.
3. Are emerging markets expected to have different price trends?
Yes, emerging markets may see lower initial prices due to lower purchasing power but could experience faster growth and potential price increases as market penetration improves.
4. How does formulation affect pricing strategies?
Natural, organic, or specialized formulations often command higher prices, while basic stimulant laxatives tend to be priced lower to attract cost-sensitive consumers.
5. What regulatory considerations could impact future prices?
Stricter safety and efficacy standards may increase development and compliance costs, which could be reflected in higher retail prices.
Cited Sources
[1] MarketsandMarkets, "Laxatives Market," 2022.
[2] IBISWorld, "Over-the-Counter (OTC) Medication in the US," 2022.
[3] Statista, "Global Digestive Disorder OTC Market," 2023.