Share This Page
Drug Price Trends for GENOTROPIN MINIQUICK
✉ Email this page to a colleague
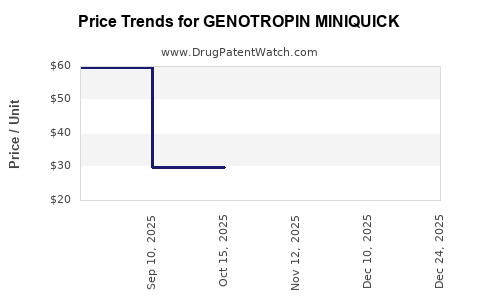
Average Pharmacy Cost for GENOTROPIN MINIQUICK
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GENOTROPIN MINIQUICK 0.4 MG | 00013-2650-02 | 61.63980 | EACH | 2026-01-01 |
| GENOTROPIN MINIQUICK 0.6 MG | 00013-2651-02 | 93.05003 | EACH | 2026-01-01 |
| GENOTROPIN MINIQUICK 0.2 MG | 00013-2649-02 | 30.89868 | EACH | 2026-01-01 |
| GENOTROPIN MINIQUICK 0.4 MG | 00013-2650-02 | 59.84447 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GENOTROPIN MINIQUICK
What is GENOTROPIN MINIQUICK?
GENOTROPIN MINIQUICK is a human growth hormone (hGH) formulation produced by Pfizer. Approved by the U.S. Food and Drug Administration (FDA) in 2004, it is designed for subcutaneous injection via a pre-filled, single-use device. The product is used to treat growth failure in children and adults with growth hormone deficiency, as well as other off-label indications such as Turner syndrome and chronic kidney disease.
Market Size and Demand Drivers
Global Human Growth Hormone Market
The global growth hormone market was valued at approximately USD 4.1 billion in 2022 and is projected to reach USD 6.2 billion by 2028, registering a CAGR of around 7.6% (Fortune Business Insights, 2022). The expansion is driven by increasing diagnoses of growth hormone deficiency, rising pediatric growth disorders, and broader acceptance of hormone therapy.
Key Market Segments
- Pediatric growth deficiency: Accounts for approximately 60% of demand.
- Adult growth hormone deficiency: Constitutes about 30% of total market.
- Off-label uses: Comprise the remaining 10%, including growth hormone-releasing hormone testing and anti-aging markets.
Regional Demand Patterns
- North America: Largest share, approximately 50%, driven by established healthcare infrastructure and high awareness.
- Europe: Holds around 25%, with steady growth.
- Asia-Pacific: Fastest growth rate, estimated CAGR of 9%, due to expanding healthcare access and awareness.
Competitive Landscape
Major competitors include Novo Nordisk, Eli Lilly, Sandoz, and Teva Pharmaceuticals. Pfizer’s GENOTROPIN MINIQUICK holds about 15-20% market share, primarily in North America. Patents expire in key markets between 2024 and 2028, opening potential for biosimilar entry.
Pricing Benchmarks
Pricing varies significantly by region, formulation, and payment framework. As of 2023, average wholesale prices are:
| Region | Price per Dose (USD) | Notes |
|---|---|---|
| U.S. | $300 - $370 | Single-use, pre-filled autoinjector. |
| Europe | €250 - €330 | Similar formulations; price varies by country. |
| Japan | ¥25,000 - ¥35,000 | Prices managed under national health schemes. |
| Emerging markets | $150 - $250 | Lower, due to regulatory and purchasing power differences. |
Price Trends
- Discounts and rebates: Common in the U.S., reducing net price by 20-30% for payers and providers.
- Biosimilar competition: Entry in 2024-2028 expected to pressure prices downward by 15-25% over subsequent years.
- Regulatory impacts: Price controls in Europe and Japan restrict upward movement of list prices.
Price Projections (Next Five Years)
| Year | U.S. Average Price per Dose | European Average Price per Dose | Asian Average Price per Dose | Key Factors |
|---|---|---|---|---|
| 2023 | $330 | €330 | ¥35,000 | Stable, pending biosimilar entry |
| 2024 | $310 | €300 | ¥32,000 | Biosimilar approvals in U.S., Europe |
| 2025 | $290 | €280 | ¥29,000 | Increased biosimilar market share |
| 2026 | $270 | €260 | ¥27,000 | Market saturation, price pressures |
| 2027 | $250 | €250 | ¥25,000 | Continued biosimilar penetration |
Regulatory Impact and Market Risks
- Patent cliff: Pfizer’s patent protections expire in key territories between 2024 and 2028.
- Biosimilar development: Several biosimilar products are in late-stage development; widespread approval expected within the next three to five years.
- Pricing reforms: Governments, especially in Europe and Japan, implement stricter price controls, potentially limiting revenue growth.
- Supply chain constraints: Manufacturing bottlenecks can impact availability and pricing.
Strategic Opportunities
- Biosimilar competition: Companies entering biosimilar markets should consider aggressive pricing and early launches to capture market share.
- Market expansion: Increasing access in emerging markets offers growth potential, especially with tiered pricing models.
- Formulation innovations: Development of longer-acting formulations and alternative delivery methods could sustain demand.
Key Takeaways
- GENOTROPIN MINIQUICK remains a significant player in the growth hormone market, with a current share of 15-20%.
- Prices are region-dependent, with U.S. prices averaging around USD 330 per dose in 2023.
- Biosimilar entry, expected from 2024 onward, is likely to exert significant downward pressure on prices.
- Global market growth is driven by increasing diagnoses and expanding healthcare infrastructure, notably in Asia-Pacific.
- Regulatory and patent expiries between 2024 and 2028 will shape pricing strategies and market share.
FAQs
1. How does the price of GENOTROPIN MINIQUICK compare with other growth hormone products?
GENOTROPIN MINIQUICK’s prices are in line with other branded growth hormones, typically ranging between USD 300 to USD 370 per dose in the U.S., with biosimilars expected to price 15-25% lower post-entry.
2. When will biosimilars likely impact the market price?
Biosimilar approvals in the U.S., Europe, and Japan are anticipated between 2024 and 2026, with their market impact materializing over the subsequent one to three years.
3. What regulatory policies influence growth hormone pricing?
Price controls in Europe and Japan restrict increases, while the U.S. largely relies on private negotiations, with Medicaid and Medicare also influencing net prices through rebates.
4. How are emerging markets influencing the overall market?
Emerging markets project a CAGR of approximately 9% due to expanding healthcare access and increased awareness, offering growth opportunities despite lower price points.
5. What strategies can manufacturers adopt to maintain profitability?
Early biosimilar development, geographic expansion, formulation innovation, and negotiation of favorable reimbursement policies strengthen profitability amid price erosion.
References
- Fortune Business Insights. (2022). Human Growth Hormone Market Size, Share & Industry Analysis. Retrieved from https://www.fortunebusinessinsights.com
- Pfizer. (2023). GENOTROPIN MINIQUICK product details and approval information.
- IQVIA. (2023). Global growth hormone market report.
- European Medicines Agency. (2022). Regulatory landscape for biosimilar growth hormones.
- Japan Ministry of Health, Labour and Welfare. (2023). Pricing frameworks for biologic products in Japan.
More… ↓
