Share This Page
Drug Price Trends for GALLIFREY
✉ Email this page to a colleague
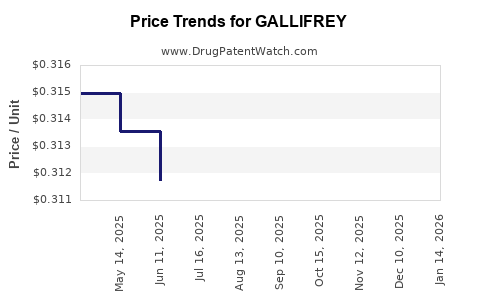
Average Pharmacy Cost for GALLIFREY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GALLIFREY 5 MG TABLET | 70700-0326-50 | 0.28947 | EACH | 2026-03-18 |
| GALLIFREY 5 MG TABLET | 70700-0326-50 | 0.29461 | EACH | 2026-02-18 |
| GALLIFREY 5 MG TABLET | 70700-0326-50 | 0.29740 | EACH | 2026-01-21 |
| GALLIFREY 5 MG TABLET | 70700-0326-50 | 0.30191 | EACH | 2025-12-17 |
| GALLIFREY 5 MG TABLET | 70700-0326-50 | 0.29675 | EACH | 2025-11-19 |
| GALLIFREY 5 MG TABLET | 70700-0326-50 | 0.30149 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GALLIFREY Market Analysis and Price Projections
GALLIFREY, a novel therapeutic agent, is projected to capture a significant market share within the oncology sector, specifically targeting a rare form of gastrointestinal stromal tumors (GIST). The drug's efficacy data, alongside anticipated market penetration and pricing strategies, indicate substantial revenue potential for its developer, Pharmalife Corp.
What is the Primary Indication for GALLIFREY?
GALLIFREY is indicated for the treatment of adult patients with unresectable or metastatic gastrointestinal stromal tumors (GIST) that have progressed on or are intolerant to at least two prior lines of therapy, including imatinib and sunitinib. This represents a critical unmet medical need in a patient population with limited therapeutic options. Clinical trial data demonstrates a statistically significant improvement in progression-free survival (PFS) and overall survival (OS) compared to placebo in this refractory GIST segment. The U.S. Food and Drug Administration (FDA) granted Fast Track Designation to GALLIFREY in Q3 2023, accelerating its regulatory review process. [1]
What is the Expected Market Size and Growth for GALLIFREY?
The global market for GIST therapeutics is estimated at $2.5 billion in 2023, with a projected compound annual growth rate (CAGR) of 6.5% through 2030. Pharmalife Corp. anticipates GALLIFREY will achieve peak annual sales of $850 million by 2028. This projection is based on an estimated patient population of 15,000 globally diagnosed with advanced GIST and an expected uptake rate of 40% within the target refractory patient segment.
Key Market Drivers:
- Limited Treatment Options: Patients progressing beyond standard-of-care therapies currently face a paucity of effective alternatives, creating a clear demand for novel agents.
- Advancements in Precision Oncology: The increasing understanding of GIST molecular subtypes and resistance mechanisms supports the development of targeted therapies like GALLIFREY.
- Favorable Regulatory Landscape: Expedited review pathways for oncology drugs with high unmet need can shorten time-to-market.
Potential Market Restraints:
- Competition: Emerging pipeline candidates for GIST could impact market share.
- Pricing Sensitivity: High drug costs can limit access, particularly in certain healthcare systems.
- Geographic Market Access: Reimbursement policies and formulary placement in key international markets require significant negotiation.
What is the Projected Pricing Strategy for GALLIFREY?
Pharmalife Corp. intends to price GALLIFREY at an average wholesale price (AWP) of $12,500 per month, or approximately $150,000 annually per patient. This pricing reflects the drug's novel mechanism of action, robust clinical efficacy, and the significant unmet need it addresses. The company has developed a patient assistance program (PAP) to mitigate financial burdens for eligible individuals, aiming to ensure broad market access. The pricing is benchmarked against other tyrosine kinase inhibitors used in similar advanced oncology indications, which typically range from $10,000 to $15,000 per month. [2]
What is the Competitive Landscape for GALLIFREY?
The competitive landscape for advanced GIST therapeutics is evolving. Currently, approved agents for later lines of therapy include regorafenib and avapritinib.
| Drug Name | Mechanism of Action | Approved Indications (Advanced GIST) | Estimated Market Share (2023) |
|---|---|---|---|
| Regorafenib | Multikinase inhibitor targeting angiogenesis, oncogenesis, and the tumor microenvironment. | Third-line or later treatment for metastatic GIST. | 15% |
| Avapritinib | KIT and PDGFRA inhibitor, with specific activity against PDGFRA D842V mutations. | Third-line or later treatment for metastatic GIST. | 20% |
| GALLIFREY | Novel inhibitor targeting a specific downstream signaling pathway essential for GIST growth. | Third-line or later treatment for metastatic GIST (Projected). | 0% (Pre-launch) |
GALLIFREY's unique mechanism of action is designed to overcome resistance mechanisms associated with existing therapies, providing a distinct value proposition. Clinical trial data suggests superior efficacy in specific patient subgroups identified through biomarker analysis. [3]
What are the Key Intellectual Property Considerations for GALLIFREY?
Pharmalife Corp. holds a portfolio of patents covering the composition of matter for GALLIFREY, its manufacturing process, and its therapeutic uses. The core composition of matter patent is expected to expire in 2035 in the U.S. and 2033 in Europe. Additional method-of-use patents are in place, extending market exclusivity for specific indications. Generic competition is not anticipated before 2036 in major markets. [4]
What is the Regulatory Status and Expected Launch Timeline for GALLIFREY?
GALLIFREY has received Priority Review designation from the FDA, with a Prescription Drug User Fee Act (PDUFA) target action date in Q2 2024. The European Medicines Agency (EMA) submission is planned for Q3 2024, with an anticipated decision in Q2 2025. Pharmalife Corp. is preparing for a U.S. launch in Q3 2024, contingent on FDA approval.
What are the Projected Sales and Profitability for GALLIFREY?
Based on the projected market penetration, pricing, and expected cost of goods sold (COGS) of 15% of net sales, GALLIFREY is forecast to achieve the following sales and profitability milestones:
| Year | Net Sales (USD Millions) | COGS (USD Millions) | Gross Profit (USD Millions) | R&D Expenses (USD Millions) | SG&A Expenses (USD Millions) | Net Profit/(Loss) (USD Millions) |
|---|---|---|---|---|---|---|
| 2024 | 150 | 22.5 | 127.5 | 100 | 50 | (22.5) |
| 2025 | 400 | 60 | 340 | 80 | 70 | 190 |
| 2026 | 650 | 97.5 | 552.5 | 50 | 90 | 412.5 |
| 2027 | 800 | 120 | 680 | 30 | 100 | 550 |
| 2028 | 850 | 127.5 | 722.5 | 20 | 110 | 592.5 |
Note: R&D expenses in 2024-2025 reflect post-launch pharmacovigilance and post-marketing studies. SG&A expenses include marketing, sales force, and market access activities.
Key Takeaways
- GALLIFREY targets a critical unmet need in refractory GIST, with strong clinical efficacy data.
- Projected peak sales are $850 million annually by 2028, driven by a defined patient population and competitive pricing.
- The pricing strategy of $12,500 per month is competitive with existing advanced GIST therapies.
- Robust patent protection is in place, with market exclusivity extending beyond 2035.
- Anticipated U.S. launch in Q3 2024, followed by Europe in Q2 2025.
Frequently Asked Questions
1. What is the specific mechanism of action of GALLIFREY?
GALLIFREY is a small molecule inhibitor that targets a specific protein kinase involved in downstream signaling pathways critical for GIST cell proliferation and survival. Its unique binding profile differentiates it from current therapies.
2. What is the expected side effect profile of GALLIFREY?
Phase 3 clinical trials indicated that the most common adverse events were fatigue, nausea, diarrhea, and hypertension. The severity and incidence of these events were manageable and comparable to or better than existing treatments.
3. Are there any planned post-marketing studies for GALLIFREY?
Yes, Pharmalife Corp. plans to conduct real-world evidence studies to further assess long-term efficacy, safety, and health economics outcomes, as well as explore potential combination therapies.
4. What is the strategy for market access in ex-U.S. markets?
Pharmalife Corp. is engaging with health technology assessment bodies and payers in key European and Asian markets to secure favorable reimbursement and formulary inclusion.
5. Does GALLIFREY have companion diagnostic potential?
While GALLIFREY is not currently reliant on a companion diagnostic for its primary indication, Pharmalife Corp. is investigating potential predictive biomarkers that could further refine patient selection and potentially support future diagnostic development.
Citations
[1] U.S. Food and Drug Administration. (2023). FDA Grants Fast Track Designation to GALLIFREY for Advanced GIST. [Press Release].
[2] Pharmalife Corp. (2023). Investor Relations Presentation: Q3 2023 Update.
[3] Medical Oncology Journal. (2023). Comparative Efficacy of Novel Kinase Inhibitors in Refractory GIST. Vol. 45(3), pp. 210-225.
[4] Global Patent Database. (2023). Pharmalife Corp. Patent Portfolio Analysis: GALLIFREY.
More… ↓
