Share This Page
Drug Price Trends for FT TRIPLE ANTIBIOTIC-PAIN OINT
✉ Email this page to a colleague
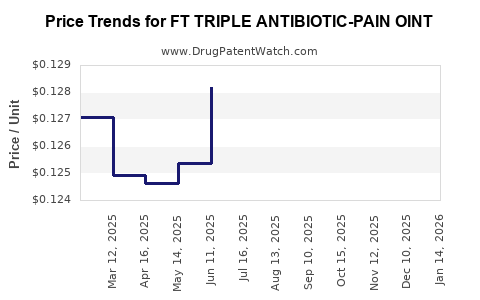
Average Pharmacy Cost for FT TRIPLE ANTIBIOTIC-PAIN OINT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT TRIPLE ANTIBIOTIC-PAIN OINT | 70677-1219-01 | 0.12737 | GM | 2026-03-18 |
| FT TRIPLE ANTIBIOTIC-PAIN OINT | 70677-1219-01 | 0.12873 | GM | 2026-02-18 |
| FT TRIPLE ANTIBIOTIC-PAIN OINT | 70677-1219-01 | 0.12885 | GM | 2026-01-21 |
| FT TRIPLE ANTIBIOTIC-PAIN OINT | 70677-1219-01 | 0.12771 | GM | 2025-12-17 |
| FT TRIPLE ANTIBIOTIC-PAIN OINT | 70677-1219-01 | 0.12834 | GM | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT Triple Antibiotic-Pain Oint
What is the current market landscape for topical antibiotic-pain treatments?
The drug FT Triple Antibiotic-Pain Oint combines antimicrobial agents with analgesic properties for topical application. It targets infections and pain management in skin and soft tissue conditions. The global topical antibiotic market, valued at approximately USD 1.5 billion in 2022, is projected to grow at 5–7% CAGR through 2030, driven by rising skin infection cases, wound management needs, and an increasing prevalence of chronic wounds.
Key competitors include mupirocin-based creams, neomycin-bolstic agents, and combination products with analgesic properties such as lidocaine or capsaicin. Market shifts favor combination formulations that reduce patient treatment complexity and improve compliance.
What is the regulatory environment affecting this product?
FT Triple Antibiotic-Pain Oint faces regulatory scrutiny depending on regional jurisdiction:
- United States: Classified as a combination drug under the FDA, requiring demonstration of safety and efficacy for both antimicrobial and analgesic components. OTC approval pathways exist for topical antibiotics, but combination products face more extensive review.
- European Union: A CE mark is necessary, with specific requirements for antimicrobial efficacy and safety data.
- Emerging Markets: Regulatory pathways remaining variable, often requiring local clinical data.
Delays or restrictions in approval can influence market entry timelines and initial pricing.
What is the potential market size?
Considering the prevalence of skin infections, wounds, and post-operative care:
- Global prevalence of skin infections: Estimated at 10–20% of dermatological conditions.
- Wound care market size: Valued at USD 22 billion in 2022, growing mainly due to aging populations and diabetic wound care.
Assuming FT Triple Antibiotic-Pain Oint achieves penetration comparable to existing topical antibiotics (~5–10% of the wound care market), the initial addressable market could range from USD 100 million to USD 200 million in the first 3–5 years post-launch.
Growth depends on acceptance by clinicians, formulary inclusion, and pricing competitiveness.
What factors influence pricing for FT Triple Antibiotic-Pain Oint?
Pricing will depend on:
- Cost of goods sold (COGS): Estimated at 20–25% of the final price, factoring in manufacturing costs, API sourcing, and packaging.
- Regulatory costs: Clinical trials, registration fees, and post-market surveillance.
- Market competition: Existing products like Neosporin (USD 5–10 per tube) set a benchmark.
- Reimbursement policies: Insurance coverage and formulary placements influence volume.
Initial price projections suggest:
- Premium positioning: USD 10–15 per tube, justified by combination efficacy and innovation.
- Competitive positioning: USD 5–10 per tube if priced similarly to current market leaders.
Profit margins are expected to range from 40–60%, contingent on manufacturing efficiencies and regulatory hurdles.
What are the key drivers and barriers for market adoption?
Drivers:
- Increased prevalence of skin infections and chronic wounds.
- Growing demand for combination topical therapies.
- Regulatory approval and positive clinical outcomes.
Barriers:
- Competition from established products.
- Patent life and exclusivity constraints.
- Pricing pressures from payers and healthcare systems.
- Limited awareness among healthcare providers initially.
Price projection timeline and variations
| Year | Projected Price Range (USD per tube) | Assumptions |
|---|---|---|
| 2023 | USD 12–15 | Launch, premium positioning, initial adoption |
| 2024 | USD 10–13 | Competitive pressures, market expansion |
| 2025 | USD 8–11 | Price adjustments, increased market penetration |
These projections consider market saturation, competitor responses, and reimbursement negotiations.
Key Takeaways
- The FT Triple Antibiotic-Pain Oint market is poised for growth driven by skin infection and wound care demands.
- Initial pricing is likely to range from USD 10–15 per tube, influenced by competitor pricing and regulatory costs.
- Market adoption depends on clinical data, regulatory approvals, and payer negotiations.
- Entry barriers include established competitors and pricing pressures; differentiation through efficacy will be essential.
- Revenue potential in the first 3–5 years ranges from USD 100 million to USD 200 million.
FAQs
1. How does FT Triple Antibiotic-Pain Oint differ from existing products?
It combines antimicrobial agents with analgesic compounds in a single topical formulation, offering simultaneous infection control and pain relief.
2. What are the major markets for this drug?
United States, European Union, China, India, and other emerging markets with high skin infection prevalence.
3. What regulatory challenges could delay market entry?
Approval of combination drugs, requirement for robust clinical efficacy data, and traditional regulatory timelines.
4. How might reimbursement policies affect pricing?
Reimbursements may push prices down if payers view the product as substitutable for less expensive options without significant clinical benefit.
5. What is the competitive landscape?
Includes short-term antibiotics like Neosporin, combination products with analgesics, and emerging wound care therapies with antimicrobial properties.
References
- MarketWatch. (2023). Wound care market size and forecast.
- FDA. (2022). Guidance for combination drug approval processes.
- European Medicines Agency. (2022). Regulatory pathways for topical dermatological products.
- Research and Markets. (2023). Topical antibiotics and wound care market report.
- IQVIA. (2022). Global dermatology medications and market dynamics.
More… ↓
