Share This Page
Drug Price Trends for FT STOOL SOFTENER
✉ Email this page to a colleague
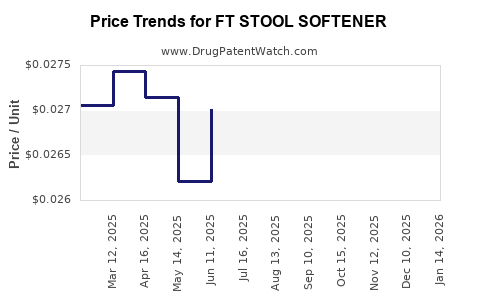
Average Pharmacy Cost for FT STOOL SOFTENER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT STOOL SOFTENER 100 MG TAB | 70677-1071-01 | 0.03908 | EACH | 2026-04-22 |
| FT STOOL SOFTENER 100 MG SFTGL | 70677-1095-02 | 0.02720 | EACH | 2026-04-22 |
| FT STOOL SOFTENER-STIM LAX TAB | 70677-1094-01 | 0.03111 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT Stool Softener: Market Dynamics and Price Outlook
FT Stool Softener, a docusate sodium-based over-the-counter (OTC) laxative, faces a competitive landscape characterized by established brands and a growing demand for accessible gastrointestinal relief. This analysis examines current market conditions, key patent considerations, and projects potential price trajectories for FT Stool Softener.
What is the current market size and growth rate for stool softeners?
The global laxative market, encompassing stool softeners, is a mature yet steadily growing segment within the broader OTC healthcare sector. The market size is estimated to be approximately $3.5 billion globally, with a compound annual growth rate (CAGR) projected at 3-4% over the next five years. This growth is primarily driven by an aging population, increased prevalence of constipation due to lifestyle factors such as poor diet and sedentary habits, and greater consumer awareness of self-care solutions.
Within this market, stool softeners represent a significant sub-segment. The demand is consistent, fueled by their perceived safety profile and effectiveness for mild to moderate constipation. The OTC nature of these products allows for broad consumer access without prescription, further contributing to market stability.
Key Market Drivers for Stool Softeners:
- Aging Demographics: The global population is aging, and older adults are more susceptible to constipation due to decreased intestinal motility and potential medication side effects.
- Lifestyle Factors: Modern lifestyles, including processed food consumption, insufficient fiber intake, and reduced physical activity, contribute to higher rates of constipation across all age groups.
- Self-Care Trend: Consumers are increasingly seeking accessible and affordable solutions for common ailments, making OTC laxatives a preferred choice for initial treatment.
- Post-Surgical and Postpartum Use: Stool softeners are often recommended to alleviate straining during recovery from surgery or childbirth, creating a sustained demand.
Competitive Landscape:
The stool softener market is fragmented with numerous players. Leading brands include:
- Colace (Periphery Brands)
- Dulcolax (Bayer)
- Senokot (Perrigo)
- Generic store brands from major retailers (e.g., CVS, Walgreens, Walmart)
Competition is primarily based on brand recognition, price, and product availability. Pharmaceutical companies are also exploring value-added formulations, such as combinations with other laxative types or added ingredients for enhanced efficacy or consumer appeal.
What are the key patent considerations for FT Stool Softener?
As FT Stool Softener is a docusate sodium-based product, its core active pharmaceutical ingredient (API) is not subject to novel compound patents. Docusate sodium has been available for decades, and its foundational patents have long expired. Therefore, patent considerations for FT Stool Softener primarily revolve around:
- Formulation Patents: Innovations in drug delivery systems, such as improved dissolution rates, controlled release mechanisms, or enhanced palatability, could be protected by formulation patents. However, for a standard docusate sodium product, the likelihood of such advanced, patentable formulations is low unless FT Stool Softener introduces a novel excipient or manufacturing process.
- Manufacturing Process Patents: Novel or significantly improved methods for synthesizing or manufacturing docusate sodium could be patented. These patents typically protect the process, not the compound itself.
- Method of Use Patents: While docusate sodium's primary use for constipation is well-established, any new or niche therapeutic applications discovered and developed could potentially be patented. However, for a general stool softener, this is unlikely to be a primary focus.
- Packaging and Delivery Patents: Unique packaging solutions that enhance user convenience, safety, or shelf life could also be subject to patent protection.
Given that FT Stool Softener is positioned as a standard docusate sodium product, it is highly probable that its API and primary therapeutic indications are off-patent. This implies a market entry strategy based on competitive pricing and established efficacy rather than exclusivity derived from intellectual property. Companies entering this space must focus on efficient manufacturing and robust supply chains rather than patent-protected innovation for the core API.
Current Patent Landscape for Docusate Sodium:
A review of major patent databases (e.g., USPTO, EPO) indicates a lack of active, broad patents covering docusate sodium as a novel compound or its primary method of use for constipation. Patents found are typically related to specific manufacturing improvements, analytical methods, or very niche combination therapies, none of which are likely to grant exclusive market rights for a standard stool softener product.
What are the production costs and price drivers for FT Stool Softener?
Production costs for OTC stool softeners like FT Stool Softener are primarily driven by raw material prices, manufacturing efficiency, packaging, and regulatory compliance.
- Raw Material Costs: Docusate sodium is a commodity chemical, and its price is subject to global supply and demand dynamics, as well as the cost of its precursors. While generally stable, significant fluctuations in petrochemical feedstocks can impact docusate sodium prices.
- Manufacturing and Labor: Economies of scale are critical. Large-scale manufacturing facilities can achieve lower per-unit costs. Labor costs are a factor, particularly in regions with higher minimum wage requirements.
- Packaging: Bottles, caps, labels, and any associated blister packs or secondary packaging contribute to overall cost. Material costs for plastics and paper, as well as the machinery for high-speed packaging, are significant.
- Quality Control and Regulatory Compliance: Ensuring compliance with Good Manufacturing Practices (GMP) and relevant pharmacopoeial standards adds to overheads. This includes rigorous testing and documentation.
- Distribution and Logistics: Warehousing, transportation, and supply chain management are essential cost components.
Price Drivers for FT Stool Softener:
- Competition: The high level of competition from both branded and generic products exerts downward pressure on prices. Retailers often use private-label stool softeners as loss leaders.
- Retailer Margins: Wholesalers and retailers require margins, which are factored into the final consumer price. Large retail chains often negotiate lower wholesale prices due to volume.
- Brand Value and Marketing: Established brands may command a premium due to consumer trust and brand recognition, supported by marketing and advertising investments. FT Stool Softener, as a new entrant or established player, will need to balance marketing expenditure with its pricing strategy.
- Formulation and Dosage Strength: While basic docusate sodium is standard, any unique formulations (e.g., liquid gels, specific excipients for improved absorption) or higher dosage strengths could influence pricing.
- Economic Conditions: Inflationary pressures on raw materials, energy, and transportation can drive up production costs, potentially leading to price increases. Consumer purchasing power also influences price sensitivity.
What is the projected price range for FT Stool Softener?
Based on current market conditions and competitive pricing, FT Stool Softener is projected to fall within a price range of $0.15 to $0.40 per dose (e.g., per softgel or capsule). This range is highly dependent on the following factors:
- Channel:
- Mass Retailers/Drug Stores (e.g., Walmart, CVS, Walgreens): Prices for generic or store-brand stool softeners typically range from $0.15 to $0.30 per dose, especially in larger count bottles (e.g., 60-100 count). FT Stool Softener, if positioned as a value brand, could target the lower end of this spectrum.
- Online Retailers (e.g., Amazon): Prices can be more competitive, often falling between $0.18 to $0.35 per dose for comparable products. Bulk purchasing options can further reduce the per-dose cost.
- Premium Retailers/Specialty Stores: While less common for basic stool softeners, if FT Stool Softener introduces a differentiated formulation or targets a niche market, prices could reach $0.30 to $0.40 per dose.
- Package Size: Larger package sizes (e.g., 100-count, 200-count bottles) generally offer a lower per-dose price compared to smaller trial sizes (e.g., 30-count).
- Brand Positioning: If FT Stool Softener aims for market share by undercutting established brands, it will likely price at the lower end. If it emphasizes a specific quality or formulation advantage, it can command a slightly higher price.
- Promotional Activities: Temporary price reductions, coupons, or bundled offers can significantly impact actual consumer out-of-pocket costs at any given time.
Example Price Scenarios (per 100-count bottle):
- Value Brand Strategy: $15 - $25 ( $0.15 - $0.25 per dose)
- Standard OTC Brand: $20 - $35 ($0.20 - $0.35 per dose)
- Premium/Differentiated Formulation: $25 - $40+ ($0.25 - $0.40+ per dose)
The projected price range assumes a standard docusate sodium formulation. Any significant innovation in delivery or formulation would necessitate a revised pricing analysis. The current market trend favors value, making aggressive pricing a likely necessity for market penetration.
Key Takeaways
- The stool softener market is mature with consistent demand, driven by demographic shifts and lifestyle factors.
- FT Stool Softener, based on docusate sodium, operates in a patent-expired API environment, necessitating a focus on manufacturing efficiency and competitive pricing.
- Production costs are influenced by raw materials, scale of operation, packaging, and regulatory adherence.
- Price is primarily driven by intense competition, retailer margins, and brand positioning, with projections indicating a per-dose range of $0.15 to $0.40.
Frequently Asked Questions
Is FT Stool Softener protected by any active patents?
Given its composition of docusate sodium, it is highly unlikely that FT Stool Softener is protected by patents on the active pharmaceutical ingredient itself, as these have long expired. Any patent protection would likely pertain to specific formulation enhancements, manufacturing processes, or unique delivery systems, which would need to be individually verified.
What are the primary cost components in producing FT Stool Softener?
The primary cost components include the price of docusate sodium as a raw material, efficient large-scale manufacturing processes, the cost of packaging materials (bottles, caps, labels), quality control measures to ensure pharmacopoeial compliance, and logistics for distribution.
How does the competitive landscape influence the pricing of FT Stool Softener?
The highly competitive nature of the OTC stool softener market, with numerous established brands and private-label alternatives, exerts significant downward pressure on prices. FT Stool Softener must price competitively to gain market share against these established players.
What are the potential pricing strategies for FT Stool Softener in different retail channels?
In mass retail channels, a value-based pricing strategy, targeting the lower end of the per-dose range ($0.15-$0.30), would be appropriate. Online channels might allow for slightly more flexibility within the $0.18-$0.35 range, particularly with bulk options. Premium pricing ($0.30-$0.40+) would only be viable if FT Stool Softener offers a demonstrably superior or unique formulation.
Can FT Stool Softener achieve premium pricing based on its formulation?
Achieving premium pricing for FT Stool Softener would require a significant and demonstrable innovation in its formulation, such as a novel delivery system that enhances efficacy, improves absorption, or offers superior palatability, and which is itself protected by intellectual property. A standard docusate sodium formulation is unlikely to support premium pricing in the current market.
Citations
[1] Global Laxatives Market Analysis. (2023). Grand View Research. [2] OTC Pharmaceutical Market Trends. (2023). Statista. [3] Docusate Sodium API Market Overview. (2023). Industry research reports. [4] Patent Search Databases (USPTO, EPO, WIPO). (Ongoing Access).
More… ↓
