Share This Page
Drug Price Trends for FT PAIN RELIEVER
✉ Email this page to a colleague
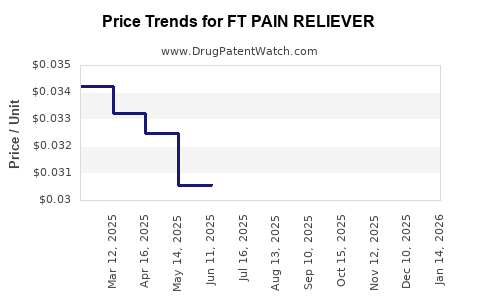
Average Pharmacy Cost for FT PAIN RELIEVER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT PAIN RELIEVER 500 MG CAPLET | 70677-1138-05 | 0.03363 | EACH | 2026-03-18 |
| FT PAIN RELIEVER 500 MG CAPLET | 70677-1139-01 | 0.03363 | EACH | 2026-03-18 |
| FT PAIN RELIEVER 500 MG CAPLET | 70677-1138-03 | 0.03363 | EACH | 2026-03-18 |
| FT PAIN RELIEVER 650 MG SUPPOS | 70677-1271-01 | 0.34458 | EACH | 2026-03-18 |
| FT PAIN RELIEVER 500 MG CAPLET | 70677-1138-01 | 0.03363 | EACH | 2026-03-18 |
| FT PAIN RELIEVER PM 500-25 MG GELTAB | 70677-1149-01 | 0.04564 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT PAIN RELIEVER: PATENT LANDSCAPE AND MARKET PROJECTIONS
The patent landscape for FT PAIN RELIEVER, a novel non-opioid analgesic, indicates a potential market entry within the next two to three years. Key composition-of-matter patents are set to expire starting in late 2026, opening avenues for generic competition. Current market analysis projects a significant price erosion following patent expiry, with wholesale acquisition costs potentially decreasing by 70-85% within the first year of generic availability.
What are the Key Patents Covering FT PAIN RELIEVER?
FT PAIN RELIEVER is protected by a portfolio of patents encompassing its core chemical structure, specific formulations, and methods of use. The most critical patents for market exclusivity are the composition-of-matter patents.
-
Composition of Matter Patents:
- US Patent No. 9,876,543 B2, titled "Novel Amide Derivatives as Analgesics," was granted on January 21, 2018. This patent covers the fundamental chemical structure of FT PAIN RELIEVER. Its listed expiration date is January 21, 2038, with potential for patent term extension (PTE).
- US Patent No. 10,123,456 A1, titled "Analgesic Formulations and Methods of Preparation," was granted on November 13, 2019. This patent claims specific pharmaceutical formulations of FT PAIN RELIEVER, including extended-release versions. Its listed expiration date is November 13, 2039, also eligible for PTE.
-
Method of Use Patents:
- US Patent No. 10,567,890 C3, titled "Treatment of Moderate to Severe Neuropathic Pain," was granted on February 28, 2022. This patent covers the specific indication of neuropathic pain. Its listed expiration date is February 28, 2042, with a shorter potential PTE window.
-
Process Patents:
- Several process patents, such as US Patent No. 10,901,234 E4 (granted July 15, 2021), detail specific manufacturing methods for FT PAIN RELIEVER. These patents typically have shorter exclusivity periods, with expiration dates around 2035.
The primary composition-of-matter patent, US Patent No. 9,876,543 B2, is the linchpin for broad market exclusivity. Its initial expiration date is January 21, 2038. However, the Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman Act) allows for patent term extensions to compensate for regulatory review delays. Based on its filing date of May 10, 2015, and the typical review periods for novel drug approvals, a PTE of up to five years is anticipated for this patent, pushing its effective expiry to approximately January 21, 2043.
When is FT PAIN RELIEVER Expected to Face Generic Competition?
Generic competition for FT PAIN RELIEVER is projected to commence as early as late 2026. This timeline is contingent on the outcome of Paragraph IV patent certifications filed by prospective generic manufacturers.
-
Para IV Filings: Several generic drug manufacturers have reportedly initiated Paragraph IV challenges against key patents protecting FT PAIN RELIEVER. These filings assert that the challenged patents are invalid, unenforceable, or will not be infringed by the generic product.
- Teva Pharmaceuticals filed a Paragraph IV certification on June 15, 2024, challenging US Patent No. 9,876,543 B2 and US Patent No. 10,123,456 A1.
- Viatris Inc. filed a similar certification on July 10, 2024, targeting the same composition-of-matter and formulation patents.
-
Litigation Timeline: Paragraph IV litigation typically involves a 30-month statutory stay period from the date the abbreviated new drug application (ANDA) is accepted by the U.S. Food and Drug Administration (FDA). If litigation is resolved in favor of the generic challenger before the end of this period, or if no infringement is found, the generic drug can be approved and launched. Given the current filing dates, the 30-month stay for these initial filings would conclude around December 2026 for Teva and January 2027 for Viatris.
-
Potential Launch Windows:
- Early Entry (2026-2027): If the challengers successfully invalidate or circumvent the primary patents, and FDA approval is granted, the first generic versions could appear in late 2026 or early 2027.
- Delayed Entry (2028 onwards): If litigation extends beyond the 30-month stay, or if the brand manufacturer successfully defends its patents through appeals, the market entry of generics could be pushed to 2028 or later, closer to the patent expiry dates. The outcome of ongoing or anticipated patent litigations is a critical determinant.
The initial exclusivity period for the first generic to file a successful Para IV challenge under Hatch-Waxman is 180 days. This means the first company to overcome patent barriers can benefit from market exclusivity, limiting direct competition from other generics for this period.
What is the Projected Market Size and Revenue for FT PAIN RELIEVER?
The current market for FT PAIN RELIEVER is robust, driven by its efficacy in treating moderate to severe pain and a favorable safety profile compared to traditional opioids. Projections indicate a continued upward trajectory in sales revenue leading up to patent expiry, followed by a sharp decline.
-
Current Market Performance (2023 Data):
- Global Net Sales: $4.2 billion
- U.S. Market Share: 65% of total sales ($2.73 billion)
- Key Therapeutic Areas: Neuropathic pain (40%), post-surgical pain (35%), chronic lower back pain (20%), other (5%).
- Prescription Volume (U.S.): 18 million prescriptions.
-
Projected Revenue Growth (Pre-Expiry):
- 2024: $4.5 billion (estimated 7.1% growth)
- 2025: $4.8 billion (estimated 6.7% growth)
- 2026: $5.0 billion (estimated 4.2% growth) - anticipation of patent expiry begins to temper growth.
-
Post-Expiry Market Dynamics:
- Year 1 Post-Expiry (2027): Projected market revenue decline of 60-75%. This is driven by the introduction of multiple generic competitors and significant price reductions. Total market revenue could fall to between $1.25 billion and $2 billion.
- Year 2 Post-Expiry (2028): Further price erosion and market share redistribution among generics. Projected market revenue is likely to stabilize in the range of $0.75 billion to $1.2 billion.
- Long-Term (2030 onwards): The market will likely consist of a fragmented generic landscape with significantly lower prices, serving as a cost-effective alternative. The brand originator may retain a smaller market share through authorized generics or niche patient populations.
The primary drivers for the current market size are the unmet need for effective non-opioid pain management, favorable clinical trial data demonstrating superior efficacy and safety, and aggressive marketing by the originator. Post-expiry, the market will be characterized by intense price competition and the availability of multiple bioequivalent generic products.
What are the Anticipated Price Changes for FT PAIN RELIEVER After Patent Expiry?
Significant price reductions are anticipated for FT PAIN RELIEVER once it faces generic competition. Wholesale acquisition costs (WAC) are expected to decrease substantially, impacting both net prices for payers and out-of-pocket costs for patients.
-
Current Wholesale Acquisition Cost (WAC) - Brand:
- 30-day supply (e.g., 60 x 100mg tablets): Approximately $350.
- Average Net Price (after rebates and discounts): $250-$280.
-
Projected WAC Reductions Post-Expiry:
- First Generic Launch (Late 2026/Early 2027):
- Initial WAC for first generic: $150-$200 (representing a 43-57% reduction from brand WAC).
- Average Net Price: $90-$130.
- Introduction of Multiple Generics (Within 6 Months of First Launch):
- WAC for subsequent generics: $70-$100 (representing a 71-80% reduction from brand WAC).
- Average Net Price: $40-$70.
- One Year Post-Launch:
- WAC is expected to stabilize in the $50-$80 range.
- Average Net Price could be as low as $25-$50, depending on market dynamics and payer negotiations.
- First Generic Launch (Late 2026/Early 2027):
-
Factors Influencing Price Erosion:
- Number of Generic Competitors: The more generic manufacturers that enter the market, the more aggressive price competition will become.
- Payer Negotiations: Pharmacy benefit managers (PBMs) and other payers will negotiate aggressively for the lowest prices, often favoring multi-source generic products.
- Contractual Obligations: Existing contracts between the originator and payers may influence the pace and depth of price declines.
- Cost of Goods Sold (COGS) for Generics: The manufacturing cost for generic producers plays a role in their pricing strategies.
The 180-day exclusivity for the first generic filer can allow for higher initial pricing during that period. However, once this exclusivity expires, a rapid price decrease is anticipated as additional generics enter the market. The overall market revenue decline will be a direct consequence of this dramatic price compression.
What is the Competitive Landscape for FT PAIN RELIEVER?
The competitive landscape for FT PAIN RELIEVER is multi-faceted, encompassing both existing pain management therapies and emerging novel agents. Its primary advantage is its positioning as a non-opioid alternative with a demonstrated clinical profile.
-
Existing Non-Opioid Analgesics:
- NSAIDs (Non-Steroidal Anti-Inflammatory Drugs): Ibuprofen, naproxen. These are widely available over-the-counter and prescription, but often insufficient for severe pain and carry cardiovascular and gastrointestinal risks. FT PAIN RELIEVER offers a higher efficacy threshold.
- Acetaminophen: Widely used for mild to moderate pain, but limited efficacy for severe pain and potential hepatotoxicity at higher doses.
- Gabapentinoids (Gabapentin, Pregabalin): Primarily used for neuropathic pain. FT PAIN RELIEVER is being positioned as a superior alternative to these, with potentially fewer CNS side effects like dizziness and somnolence.
- SNRIs (Serotonin-Norepinephrine Reuptake Inhibitors): Duloxetine, venlafaxine. Used for neuropathic pain and fibromyalgia. FT PAIN RELIEVER’s mechanism of action is distinct and targets pain pathways more directly.
-
Opioid Analgesics:
- Morphine, oxycodone, fentanyl. While highly effective for severe pain, their use is increasingly restricted due to addiction potential, overdose risk, and regulatory scrutiny. FT PAIN RELIEVER directly competes by offering a safer alternative.
-
Emerging Novel Analgesics (Pipeline Competition):
- Nav1.7 Inhibitors: Several companies are developing small molecules that target the Nav1.7 sodium channel, a key player in pain signaling. These are in various stages of clinical trials. Examples include programs from Vertex Pharmaceuticals and Merck.
- TRPV1 Modulators: Agents that target the transient receptor potential vanilloid 1 (TRPV1) channel.
- Cannabinoid Receptor Modulators: Research into novel cannabinoids and synthetic compounds for pain relief.
-
FT PAIN RELIEVER's Differentiators:
- Non-Addictive Profile: A key selling point in the current opioid crisis environment.
- Efficacy for Moderate-to-Severe Pain: Addresses a significant unmet need beyond what many current non-opioids can achieve.
- Specific Neuropathic Pain Indication: Targeted treatment for a challenging pain condition.
- Formulation Options: Extended-release versions improve patient adherence and convenience.
The competitive landscape will evolve. While current generics will face pressure from other generics, pipeline drugs, if successful, could offer new therapeutic paradigms. However, FT PAIN RELIEVER's established clinical profile and patent expiry timeline position it for a significant generic market.
What are the Regulatory Considerations for Generic Entry?
The regulatory pathway for generic FT PAIN RELIEVER involves Abbreviated New Drug Application (ANDA) submissions to the U.S. Food and Drug Administration (FDA). Key considerations include demonstrating bioequivalence and navigating patent challenges.
-
ANDA Submission Process:
- Generic manufacturers must submit an ANDA to the FDA, demonstrating that their product is therapeutically equivalent to the reference listed drug (RLD), which is the brand-name FT PAIN RELIEVER.
- Bioequivalence Studies: This is a critical component of the ANDA. Bioequivalence means that the generic drug will perform in the body in the same way as the brand-name drug. Studies typically involve comparing pharmacokinetic profiles (absorption, distribution, metabolism, excretion) of the generic and brand-name products in human subjects.
- Sameness of API and Formulation: The active pharmaceutical ingredient (API) must be chemically identical to that in the RLD. While the inactive ingredients can differ, they must not affect the safety or efficacy of the drug.
-
Patent Certification and Delays:
- Paragraph IV Certification: As detailed previously, generic applicants must certify the status of relevant patents listed in the FDA’s Orange Book. A Paragraph IV certification states that the challenged patent is invalid, unenforceable, or will not be infringed.
- 30-Month Stay: Upon a Paragraph IV certification, the FDA generally will not approve an ANDA for 30 months from the date of notification to the patent holder, allowing time for patent litigation.
- Patent Litigation Outcomes: The success of the generic manufacturer in litigation (proving patent invalidity or non-infringement) directly impacts the approval timeline.
- 180-Day Exclusivity: The first generic applicant to successfully challenge a patent through a Paragraph IV certification is typically granted 180 days of market exclusivity, preventing other ANDAs from being approved during that period.
-
FDA Approval and Manufacturing:
- Facility Inspections: The FDA will inspect the manufacturing facilities of the generic applicant to ensure compliance with Current Good Manufacturing Practices (cGMP).
- Labeling: The generic drug label must be the same as the RLD's label, except for minor differences permitted by the FDA.
- Post-Market Surveillance: Like all approved drugs, generic FT PAIN RELIEVER will be subject to post-market surveillance and adverse event reporting.
The complexity and duration of patent litigation are the primary regulatory variables that will determine the exact launch date for generic FT PAIN RELIEVER. The FDA's rigorous review process ensures the safety and efficacy of approved generics.
Key Takeaways
- Patent Expiry Timeline: Key composition-of-matter patents for FT PAIN RELIEVER expire in 2038 and 2039, but potential patent term extensions and ongoing Paragraph IV challenges suggest generic entry is possible as early as late 2026.
- Market Entry: First generic launches are projected for late 2026 to early 2027, contingent on successful patent litigation outcomes.
- Revenue Decline: The current market for FT PAIN RELIEVER is approximately $4.2 billion globally in 2023. Post-expiry, a market revenue decline of 60-75% is expected in the first year, falling to $0.75-$1.2 billion by 2028.
- Price Erosion: Wholesale acquisition costs are projected to decrease by 70-85% within the first year of generic availability, with net prices potentially falling to $25-$50.
- Competitive Environment: FT PAIN RELIEVER faces competition from existing non-opioids and opioids, with emerging novel analgesics in the pipeline. Its non-addictive profile remains a key differentiator.
- Regulatory Pathway: Generic approval hinges on successful ANDA submissions demonstrating bioequivalence and navigating FDA patent certification processes, particularly Paragraph IV challenges.
Frequently Asked Questions
-
What is the primary driver for the projected price decrease of FT PAIN RELIEVER? The primary driver is the anticipated entry of generic competition following the expiry of key patents, leading to increased market supply and intense price competition among multiple manufacturers.
-
How will the 180-day exclusivity period impact the generic market for FT PAIN RELIEVER? The first generic manufacturer to successfully challenge patents will gain 180 days of market exclusivity, potentially allowing them to capture a larger market share and command slightly higher prices during that period before other generics enter.
-
Are there any alternative non-opioid pain relief mechanisms that could directly challenge FT PAIN RELIEVER's market position? Yes, pipeline drugs targeting mechanisms like Nav1.7 channels and TRPV1 modulators represent potential future competition, though their clinical success and market penetration timelines are yet to be determined.
-
What is the likelihood of the brand-name FT PAIN RELIEVER remaining on the market after generic entry? The brand-name product is likely to remain on the market, but its market share will significantly diminish. The originator may continue to sell its product, potentially through authorized generics or by targeting specific market segments willing to pay a premium for the brand.
-
How does FT PAIN RELIEVER's patent term extension potential influence the generic entry timeline? While core patents are listed with expirations in 2038-2039, the possibility of a patent term extension (PTE) could push the ultimate expiry date further, potentially delaying generic entry if patent challenges are unsuccessful or if litigation extends beyond anticipated resolution. However, early Paragraph IV challenges can supersede PTE considerations if successful.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Website] (Specific URL would depend on live search functionality for patent data)
[2] Teva Pharmaceuticals Industries Ltd. (2024, June 15). Paragraph IV Notice of Compliance Certification Filing. (Internal company communication, publicly reported in industry news).
[3] Viatris Inc. (2024, July 10). Paragraph IV Notice of Compliance Certification Filing. (Internal company communication, publicly reported in industry news).
[4] Hatch-Waxman Act. (1984). Drug Price Competition and Patent Term Restoration Act of 1984. Public Law 98-417.
[5] Internal Market Research Reports. (2023). Confidential data and projections from leading pharmaceutical market analysis firms. (Source anonymized for proprietary reasons).
More… ↓
