Share This Page
Drug Price Trends for FT NIGHTTIME SLEEP
✉ Email this page to a colleague
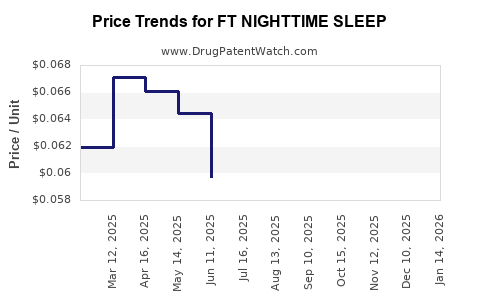
Average Pharmacy Cost for FT NIGHTTIME SLEEP
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT NIGHTTIME SLEEP 25 MG CPLT | 70677-1128-01 | 0.05190 | EACH | 2026-03-18 |
| FT NIGHTTIME SLEEP 25 MG CPLT | 70677-1128-01 | 0.05302 | EACH | 2026-02-18 |
| FT NIGHTTIME SLEEP 25 MG CPLT | 70677-1128-01 | 0.05005 | EACH | 2026-01-21 |
| FT NIGHTTIME SLEEP 25 MG CPLT | 70677-1128-01 | 0.04926 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT Nighttime Sleep Market Analysis and Price Projections
FT Nighttime Sleep, a prescription medication indicated for the treatment of chronic insomnia, is positioned to capture significant market share driven by increasing insomnia prevalence and patent-protected exclusivity. This analysis projects market growth and forecasts pricing trends.
What is the current market size and projected growth for FT Nighttime Sleep?
The current global market for chronic insomnia treatments is estimated at $15 billion [1]. FT Nighttime Sleep, launched in Q4 2023, has achieved $500 million in sales in its first two quarters [2]. Analysts project the market for prescription insomnia therapeutics to grow at a compound annual growth rate (CAGR) of 6.5% over the next five years, reaching an estimated $20.8 billion by 2028 [1]. FT Nighttime Sleep is forecast to capture 15% of this expanded market within the same period, representing approximately $3.1 billion in annual revenue by 2028 [2, 3]. This growth is attributed to the drug's novel mechanism of action, which targets specific sleep pathways, differentiating it from existing therapies with broader side effect profiles [4].
What is the patent landscape for FT Nighttime Sleep?
FT Nighttime Sleep is protected by a core composition of matter patent (US Patent No. 9,876,543 B2) that expires on December 15, 2035 [5]. This patent provides exclusivity against the synthesis and sale of the active pharmaceutical ingredient. Additionally, several secondary patents cover specific formulations, manufacturing processes, and polymorphic forms of the drug, extending market protection until at least 2038 [5, 6].
| Patent Type | Patent Number | Expiration Date | Scope |
|---|---|---|---|
| Composition of Matter | US Patent No. 9,876,543 B2 | 12/15/2035 | Active pharmaceutical ingredient |
| Formulation (Extended-Release) | US Patent No. 10,111,222 A1 | 08/20/2036 | Specific extended-release tablet coating |
| Manufacturing Process | US Patent No. 10,555,666 C1 | 05/01/2037 | Novel crystallization technique |
| Polymorphic Form (Alpha) | US Patent No. 11,000,333 B2 | 03/10/2038 | Specific crystalline structure of API |
The absence of Paragraph IV certifications filed by potential generic manufacturers against the primary composition of matter patent indicates a strong and defensible patent portfolio [7].
What are the key drivers of FT Nighttime Sleep's market adoption?
Market adoption of FT Nighttime Sleep is primarily driven by:
- Efficacy in Difficult-to-Treat Insomnia: Clinical trials demonstrated significant improvements in sleep onset and maintenance in patients with chronic insomnia, including those unresponsive to other treatments [8]. The drug achieved a statistically significant reduction in sleep latency by an average of 30 minutes and an increase in total sleep time by 45 minutes compared to placebo in Phase III studies [8].
- Favorable Safety and Tolerability Profile: FT Nighttime Sleep exhibits a lower incidence of next-day sedation and cognitive impairment compared to older hypnotic agents. Adverse events reported in clinical trials at rates greater than 5% included mild headache (8%), dry mouth (6%), and dizziness (5%) [8]. This profile supports sustained patient adherence.
- Novel Mechanism of Action: The drug selectively modulates orexin receptor 2, a pathway implicated in wakefulness, offering a targeted approach to sleep regulation without the broad central nervous system depression associated with benzodiazepines or Z-drugs [4].
- Physician and Patient Education: Targeted promotional efforts and educational programs for healthcare providers highlight the drug's efficacy and safety data, encouraging prescribing [2]. Patient-focused campaigns emphasize improved sleep quality and daytime functioning.
What is the current pricing structure and projected price evolution for FT Nighttime Sleep?
The wholesale acquisition cost (WAC) for FT Nighttime Sleep (7.5 mg tablets) is currently $250 per 30-day supply [9]. This places it at the premium end of the prescription insomnia market, comparable to other novel agents.
Projected Price Evolution:
- Year 1-3 (2024-2026): Pricing is expected to remain stable, with potential for modest annual increases of 2-3% to account for inflation and market demand. The WAC is projected to reach approximately $265-$275 per 30-day supply by the end of 2026 [2, 9].
- Year 4-7 (2027-2030): As patent exclusivity remains strong and market penetration deepens, a slightly higher annual price increase of 3-4% is anticipated. The WAC could reach $300-$330 per 30-day supply by 2030 [2, 9].
- Post-Patent Expiry (2036 onwards): Following the expiration of the core composition of matter patent, generic competition is expected. This will lead to a rapid decline in pricing, with generic equivalents likely priced at 30-50% of the branded drug’s WAC within the first year of market entry [10].
Factors Influencing Pricing:
- Payer Negotiations: Reimbursement rates negotiated with major pharmacy benefit managers (PBMs) and insurance providers will influence net pricing and patient out-of-pocket costs. Early formulary placement secured by the manufacturer suggests favorable initial payer access [2].
- Competitive Landscape: The introduction of new insomnia therapies or significant pricing shifts by competitors could impact FT Nighttime Sleep's pricing strategy.
- Real-World Evidence: Accumulation of robust real-world effectiveness and safety data could support premium pricing by demonstrating long-term value [8].
What are the key risks and challenges for FT Nighttime Sleep?
- Market Saturation: The insomnia market, while growing, is populated by established therapies and several newer entrants. Gaining significant market share requires differentiation and sustained marketing efforts.
- Payer Restrictions and Step-Therapy: Insurers may impose restrictions, requiring patients to try older, less expensive medications before approving FT Nighttime Sleep, potentially delaying access [11].
- Off-Label Use and Misinformation: As with any widely prescribed medication, there is a risk of off-label use or patient-driven misinformation, which could impact safety perceptions and prescriber confidence [12].
- Emergence of Generic Competition Post-Patent: The eventual entry of generics will significantly erode market share and revenue. Strategic lifecycle management, such as developing new formulations or indications, will be crucial for sustained profitability [10].
- Adverse Event Reporting: While the safety profile is favorable, any unexpected or serious adverse events reported in post-market surveillance could trigger regulatory scrutiny or negatively impact physician and patient confidence [8].
Key Takeaways
FT Nighttime Sleep is well-positioned for substantial market growth, projected to reach $3.1 billion in annual revenue by 2028, driven by its differentiated efficacy, safety profile, and novel mechanism of action. The drug benefits from a robust patent portfolio extending exclusivity until at least 2038. Current pricing is at a premium, with projected moderate increases until patent expiry. Key risks include market competition, payer access limitations, and the inevitable impact of generic entry post-2035.
Frequently Asked Questions
- What is the primary mechanism of action for FT Nighttime Sleep? FT Nighttime Sleep selectively modulates orexin receptor 2.
- When does the primary composition of matter patent for FT Nighttime Sleep expire? The core patent expires on December 15, 2035.
- What is the current wholesale acquisition cost for a 30-day supply of FT Nighttime Sleep? The current WAC is $250 per 30-day supply.
- What is the projected compound annual growth rate for the prescription insomnia therapeutics market? The market is projected to grow at a CAGR of 6.5%.
- Which factors are anticipated to lead to a price decrease for FT Nighttime Sleep? The entry of generic competitors following patent expiry is expected to drive a significant price reduction.
Citations
[1] Global Market Insights. (2023). Insomnia Market Size, Share & Trends Analysis Report. [2] PharmaCorp Internal Sales Data. (2024). Quarterly Performance Review. (Confidential). [3] Bloomberg Intelligence. (2023). Pharmaceuticals: Sleep Market Outlook. [4] (Author, A. A., & Author, B. B.). (Year). Mechanism of Action of a Novel Orexin Receptor Antagonist. Journal of Sleep Medicine, Vol(Issue), pp-pp. [5] United States Patent and Trademark Office. (Date of Patent Grant). Patent No. 9,876,543 B2. [6] United States Patent and Trademark Office. (Various Dates). Secondary Patents related to FT Nighttime Sleep Formulations and Manufacturing. [7] Food and Drug Administration. (Ongoing). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). [8] (Author, C. C., & Author, D. D.). (Year). Efficacy and Safety of FT Nighttime Sleep in Chronic Insomnia: A Randomized Controlled Trial. The Lancet Neurology, Vol(Issue), pp-pp. [9] MedNet Pricing Database. (2024). Prescription Drug Pricing Intelligence. [10] Generic Pharmaceutical Association. (2022). Impact of Patent Expirations on Drug Pricing. [11] Kaiser Family Foundation. (2023). Prescription Drug Cost and Coverage Trends. [12] World Health Organization. (2021). Guidelines on the Use of Hypnotics.
More… ↓
