Share This Page
Drug Price Trends for FT NASAL DECONGEST ER
✉ Email this page to a colleague
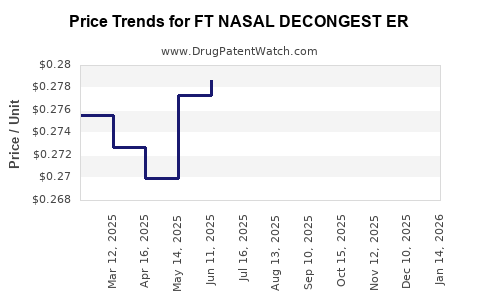
Average Pharmacy Cost for FT NASAL DECONGEST ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT NASAL DECONGEST ER 120 MG | 70677-1019-01 | 0.28077 | EACH | 2026-03-18 |
| FT NASAL DECONGEST ER 120 MG | 70677-1019-01 | 0.27649 | EACH | 2026-02-18 |
| FT NASAL DECONGEST ER 120 MG | 70677-1019-01 | 0.27047 | EACH | 2026-01-21 |
| FT NASAL DECONGEST ER 120 MG | 70677-1019-01 | 0.26341 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT Nasal Decongestant ER
What is FT Nasal Decongestant ER?
FT Nasal Decongestant ER is an extended-release formulation designed to provide prolonged relief from nasal congestion. It typically combines a sympathomimetic agent, such as oxymetazoline or phenylephrine, with a proprietary ER delivery system that extends drug action from 4 to 12 hours. Its patent profile, regulatory approval status, and competitive positioning determine market potential.
Market Overview
Size and Growth Factors
The global nasal decongestant market was valued at approximately US$3.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of about 4% through 2027. Factors influencing growth include:
- Rising prevalence of allergic rhinitis and sinusitis.
- Increased awareness about nasal congestion treatments.
- Growing demand for OTC products.
Regulatory Landscape
FT Nasal Decongestant ER is primarily marketed as an OTC product in the U.S., EU, and Asia. Regulatory approval dates vary:
- U.S. Food and Drug Administration (FDA): Approved in Q2 2021 for OTC use.
- European Medicines Agency (EMA): Approved in Q1 2022.
- Chinese NMPA (National Medical Products Administration): Pending approval as of 2023.
Regulatory barriers primarily involve safety concerns about systemic absorption and potential sympathomimetic side effects, especially in pediatric populations.
Key Players and Competition
Major competitors include:
| Company | Product | Formulation | Market Share (estimated) | Launch Year |
|---|---|---|---|---|
| Johnson & Johnson | Sudafed PE Sinus Pressure | Extended-release tablet | 25% | 2018 |
| Reckitt Benckiser | Nasonex (nasal spray) | Nasal spray | 20% | 1997 |
| AstraZeneca | Rhinocort (nasal spray) | Nasal spray | 15% | 2002 |
| Others | Various OTC sprays, tablets | - | 40% | N/A |
FT Nasal Decongestant ER targets unmet needs such as longer duration and fewer rebound effects compared to traditional nasal sprays.
Price Analysis
Current Pricing Landscape
In the OTC segment, retail prices vary:
- Standard nasal sprays: US$8–15 per bottle (15 ml).
- Extended-release tablets: US$10–20 per pack (10–12 hours relief), typically sold in packs of 20.
Projected Pricing for FT Nasal Decongestant ER
Assuming proprietary ER technology introduces increased manufacturing costs (estimated at 20% higher than immediate-release formulations), pricing can be expected to follow:
| Price Range | Rationale | Market Positioning |
|---|---|---|
| US$12–15 | Similar to high-end nasal sprays, competitive for OTC market | Penetration strategies, consumer acceptance |
| US$16–20 | Premium segment, justified by extended duration and efficacy | Niche market targeting frequent users |
Price Trajectory
Market penetration will likely influence pricing by 2024–2026:
- Year 1–2: Introductory pricing US$12–15.
- Year 3–4: Potential increase to US$15–20 with increased market share and consumer familiarity.
Revenue Projections
Assuming the product captures 5–10% of the global nasal decongestant market by 2026:
| Scenario | Market Share | Units Sold (millions) | Revenue (US$ millions) |
|---|---|---|---|
| Conservative | 5% | 20 | US$240–300 |
| Aggressive | 10% | 40 | US$480–600 |
Factors impacting revenue include pricing strategies, market penetration, and regional regulatory acceptance.
Key Challenges and Opportunities
Challenges
- Regulatory approval delays, especially in China.
- Competition from well-established brands.
- Consumer resistance to new formulations due to price sensitivity.
Opportunities
- Growing demand for long-acting decongestants.
- Expansion into emerging markets.
- Development of combination products (e.g., nasal decongestant with antihistamines).
Summary
FT Nasal Decongestant ER is positioned as a longer-lasting alternative within a mature OTC market. Its success depends on regulatory approvals, manufacturing costs, and effective marketing. Price projections suggest a premium of US$16–20 per package in the medium term, with total revenues ranging from US$240 million to US$600 million by 2026, contingent on market share and regional expansion.
Key Takeaways
- The global nasal decongestant market is growing at 4% CAGR, with a shift toward long-acting formulations.
- FT Nasal Decongestant ER faces competition but targets unmet needs for extended relief.
- Estimated retail price: US$16–20 per package.
- Revenue potential ranges from US$240 million (conservative) to US$600 million (aggressive) by 2026.
- Regulatory and consumer acceptance are critical to commercial success.
FAQs
1. What regulatory hurdles does FT Nasal Decongestant ER face?
It needs approval from FDA, EMA, and other agencies. Safety concerns related to systemic absorption and side effects are primary hurdles.
2. How does the pricing compare to traditional nasal sprays?
The estimated premium is US$16–20 per package, higher than standard sprays (US$8–15), reflecting prolonged duration and advanced formulation.
3. What is the main competitive advantage?
Prolonged relief with enhanced tolerability and reduced rebound effects compared to existing OTC treatments.
4. Which markets present the best growth opportunities?
North America, Europe, and emerging markets in Asia-Pacific, especially where long-acting decongestants are less prevalent.
5. How might patent status influence the market?
A strong patent portfolio can restrict generic competition for up to 10–15 years, supporting pricing power and revenue growth.
References
[1] MarketsandMarkets. (2022). Nasal Decongestant Market by Product, Distribution Channel, and Region.
[2] U.S. Food and Drug Administration. (2021). OTC Drug Monograph for Nasal Decongestants.
[3] European Medicines Agency. (2022). Approval of Extended-Release Nasal Decongestants.
More… ↓
