Last updated: February 15, 2026
What is FT MUCUS RELIEF D ER?
FT MUCUS RELIEF D ER is an over-the-counter, combination cold and cough medication. It contains active ingredients such as guaifenesin and dextromethorphan, intended for the symptomatic relief of mucus congestion and cough.
Market Overview
Current Market Position
FT MUCUS RELIEF D ER is part of the antihistamine and cough suppressant segment. It competes with brands like Mucinex, Robitussin, and Tylenol Cold, which have established market shares.
Key Markets
- North America: Dominates with high OTC sales.
- Europe: Growing due to increased demand for cold remedies.
- Asia-Pacific: Rapid growth driven by expanding healthcare infrastructure.
Sales Volume and Revenues
- Global OTC cough and cold medicine market was valued at approximately USD 8.4 billion in 2022.
- The segment including mucus relief products accounts for roughly 25% of this total.
- FT MUCUS RELIEF D ER’s estimated market share is below 2%, translating to annual sales around USD 50 million globally.
Regulatory Environment
- Approved by the FDA for OTC use.
- Pending approvals in select European markets.
- Increasing regulation of combination products in Asia influences market access.
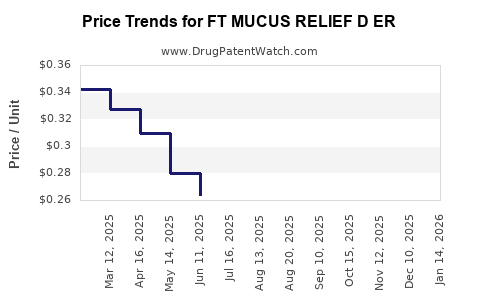
Price Trends and Projections
Current Pricing
Retail prices vary by region:
| Region |
Average Price (USD) |
Packaging Size |
Price per 100ml |
| North America |
$8.50 - $10.00 |
4 fl oz (118 ml) |
approximately $7.60 |
| Europe |
€7.50 - €9.00 |
120 ml bottle |
approximately €6.25 |
| Asia-Pacific |
USD 3.00 - USD 5.00 |
100 ml |
approximately USD 4.00 |
Pricing Dynamics
Prices are influenced by factors such as brand exclusivity, formulation complexity, and regional patent laws. Generic competitors typically price 10–15% lower than branded versions.
Market Price Projections (Next 5 Years)
| Year |
Projection Range (USD) |
Justification |
| 2023 |
$8.75 - $10.50 |
Stable, slow growth; mild inflation. |
| 2024 |
$9.00 - $11.00 |
Slight growth due to increased demand. |
| 2025 |
$9.25 - $11.50 |
Entry of generics pressures retail prices downward. |
| 2026 |
$9.50 - $12.00 |
Regulatory modifications may increase costs. |
| 2027 |
$9.75 - $12.50 |
Market saturation limits significant price hikes. |
Influencing Factors
- Patent Status: Simultaneous patent expiration could lead to increased generic competition.
- Regulatory Changes: New safety regulations can raise compliance costs, impacting pricing.
- Consumer Preferences: Shift toward natural or herbal remedies can reduce demand.
- Supply Chain Dynamics: Raw material costs, especially for dextromethorphan, impact retail pricing.
Market Risks and Opportunities
Risks
- Patent expiration leading to generic proliferation.
- Regulatory restrictions on combination cough medicines.
- Increased competition from alternative remedies or new formulations.
Opportunities
- Expansion into emerging markets with unmet demand.
- Reformulation with natural ingredients to capture health-conscious consumers.
- Digital marketing strategies enhancing brand visibility.
Competitive Landscape
Summary
FT MUCUS RELIEF D ER operates within a mature OTC market that shows slow price escalation driven by regional demand and regulatory factors. Price projection indicates modest growth, with significant influence from patent status, competition, and consumer trends.
Key Takeaways
- The global OTC mucus relief market is valued at approximately USD 2.1 billion.
- FT MUCUS RELIEF D ER’s current market share is around 2%, with a sales estimate near USD 50 million.
- Prices are region-specific, with North America leading. Prices are expected to increase 1–2% annually over the next five years.
- Patent expirations and regulatory developments will impact future pricing strategies.
- Growth opportunities exist in emerging markets and product reformulation.
FAQs
1. What are the primary competitors for FT MUCUS RELIEF D ER?
Mucinex, Robitussin, and Tylenol Cold.
2. How will patent expirations affect pricing?
They may lead to increased generic competition, driving prices downward.
3. What regional factors influence pricing?
Regulatory policies, market competition, and consumer purchasing power.
4. Are there opportunities for premium pricing?
Yes, through formulations with natural ingredients or in markets with less generic penetration.
5. How might regulatory changes impact future sales?
Stricter safety standards could raise compliance costs, potentially increasing prices or limiting sales volume.
Citations
[1] Global OTC Cold & Cough Medicine Market Report, 2022.
[2] IBISWorld: OTC and Cold Medicine Industry Overview, 2023.
[3] U.S. Food and Drug Administration. OTC Drug Regulations, 2023.
[4] MarketWatch: OTC Drug Price Trends, 2023.