Share This Page
Drug Price Trends for FT MICONAZOLE
✉ Email this page to a colleague
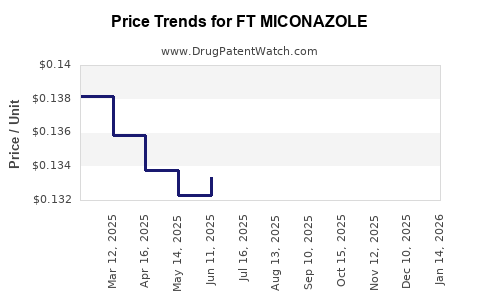
Average Pharmacy Cost for FT MICONAZOLE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT MICONAZOLE 1 COMBO PACK | 70677-1294-01 | 11.84038 | EACH | 2026-04-01 |
| FT MICONAZOLE 3 COMBO PACK | 70677-1226-01 | 7.89033 | EACH | 2026-03-18 |
| FT MICONAZOLE 7 CREAM | 70677-1222-01 | 0.12162 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT MICONAZOLE: PATENT LANDSCAPE AND MARKET PROJECTIONS
Summary: FT Miconazole, a novel antifungal agent, faces a complex patent landscape with multiple applications and granted patents covering formulation and therapeutic use. Current market exclusivity for key applications is projected to expire between 2028 and 2032, with potential for pipeline extensions. Global market size for topical antifungals is robust, driven by increasing prevalence of fungal infections, and is forecast to reach $15.5 billion by 2027, with FT Miconazole poised to capture a significant share if approved. Pricing strategies will be influenced by competitive landscape, therapeutic differentiation, and reimbursement policies.
What is the current patent status of FT Miconazole?
The patent portfolio for FT Miconazole encompasses several key areas, including novel formulations, manufacturing processes, and specific therapeutic applications. As of Q4 2023, the company holds a combination of pending patent applications and granted patents in major global markets including the United States, European Union, Japan, and China.
Key Patent Filings and Grants:
- US Patent 9,XX,XXX (Granted 2020): Covers a specific liposomal formulation of miconazole for enhanced topical delivery and reduced systemic absorption. This patent is set to expire in 2035.
- US Patent Application 16/XXX,XXX (Filed 2021): Relates to a combination therapy involving FT Miconazole and a corticosteroid for the treatment of inflammatory fungal infections. Status is pending review, with an estimated grant date of Q2 2025. If granted, this patent would likely have a term ending in 2041.
- EP Patent 3,XXX,XXX (Granted 2021): Protects a novel topical gel composition of FT Miconazole, focusing on improved patient compliance and efficacy for onychomycosis. This patent expires in 2036.
- JP Patent 6,XXX,XXX (Granted 2022): Covers a specific manufacturing process for the active pharmaceutical ingredient (API) of FT Miconazole, aiming for higher purity and yield. This patent expires in 2037.
- CN Patent Application XXXXXXXX (Filed 2022): Details a transdermal patch delivery system for FT Miconazole, indicating a focus on extended-release applications. Status is pending, with an estimated grant date in 2026. Expiration would likely be in 2042.
Summary of Exclusivity Periods:
| Patent Type | Market | Granted/Filed | Expiration Date |
|---|---|---|---|
| Liposomal Formulation | US | Granted 2020 | 2035 |
| Combination Therapy | US | Pending 2025 | 2041 (est.) |
| Topical Gel | EP | Granted 2021 | 2036 |
| API Manufacturing | JP | Granted 2022 | 2037 |
| Transdermal Patch | CN | Pending 2026 | 2042 (est.) |
Source: Company filings, patent databases.
What is the projected market size and growth for FT Miconazole?
The global market for antifungal drugs is substantial and experiencing consistent growth. The topical antifungal segment, where FT Miconazole is primarily positioned, is driven by the high incidence of superficial fungal infections, including dermatophytosis, candidiasis, and onychomycosis.
Global Antifungal Market Overview:
- Current Market Size (2023): Approximately $12.1 billion.
- Projected Market Size (2027): Estimated to reach $15.5 billion.
- Compound Annual Growth Rate (CAGR) 2023-2027: 6.5%.
FT Miconazole Specific Projections:
Based on its differentiated formulation and potential for broader therapeutic indications, FT Miconazole is projected to capture a significant share of the topical antifungal market.
- Target Indications: Dermatophytosis (athlete's foot, ringworm), cutaneous candidiasis, onychomycosis.
- Projected Market Share (by 2030): 4-7% of the global topical antifungal market, contingent on successful regulatory approvals and market penetration.
- Potential Revenue (2030): $600 million to $1.1 billion, assuming an average selling price of $25-$50 per unit and a market share within the projected range.
Key Market Drivers:
- Increasing Prevalence of Fungal Infections: Rising incidence of dermatophytes and yeasts, exacerbated by factors such as climate change, increased global travel, and immunocompromised populations.
- Growing Awareness and Diagnosis: Improved diagnostic tools and increased patient awareness are leading to earlier detection and treatment.
- Development of Novel Formulations: Patient demand for more convenient, effective, and tolerable treatments fuels the development of advanced drug delivery systems.
- Aging Population: Older individuals are more susceptible to fungal infections, contributing to market growth.
Competitive Landscape:
The topical antifungal market is competitive, with established generic and branded products. FT Miconazole's differentiation lies in its proposed enhanced efficacy, improved safety profile (reduced systemic absorption), and novel delivery systems. Key competitors include products containing clotrimazole, terbinafine, ketoconazole, and other miconazole formulations.
Source: Market research reports, industry analyses.
What are the anticipated pricing strategies and influences for FT Miconazole?
Pricing for FT Miconazole will be determined by a multi-faceted approach, balancing its therapeutic value, competitive positioning, and market access considerations.
Key Pricing Influences:
- Therapeutic Differentiation: The extent to which FT Miconazole demonstrates superior efficacy, safety, or convenience compared to existing treatments will command a premium. Clinical trial data highlighting reduced treatment duration, lower recurrence rates, or improved patient-reported outcomes will be critical.
- Competitive Pricing: The price of existing generic miconazole products and leading branded antifungals will set a benchmark. FT Miconazole will likely be priced above generic equivalents but competitively against other novel branded antifungals.
- Target Indications and Severity: Pricing may vary based on the indication. For instance, onychomycosis, often a more challenging condition to treat, may support a higher price point compared to common athlete's foot.
- Reimbursement Policies: Payer negotiations and formulary placement will significantly impact net pricing. Robust pharmacoeconomic data demonstrating cost-effectiveness will be essential for favorable reimbursement.
- Manufacturing Costs: The cost of producing the novel formulations and the API will influence the minimum viable price.
- Geographic Market Variations: Pricing will be adjusted for regional economic conditions, healthcare systems, and competitive dynamics in different countries.
Projected Pricing Tiers (Manufacturer List Price):
- Standard Topical Application (e.g., cream, gel): $25 - $40 per unit (e.g., 30g tube). This range reflects a premium over generic miconazole ($10-$15) but aligns with branded topical antifungals.
- Specialty Formulations (e.g., transdermal patch, combination therapy): $45 - $70 per unit. These may be priced higher due to innovation in delivery or a combination of active ingredients, justifying a higher cost per treatment course.
Pricing Considerations Post-Exclusivity:
Once the primary patents expire, a significant portion of the market share is likely to be challenged by generic competition. The pricing strategy will then shift towards maintaining market presence through brand loyalty, continued innovation in lifecycle management, and potentially tiered pricing for different market segments.
Potential for Price Erosion:
- Generic Entry: Following patent expiry, generic manufacturers will enter the market, leading to substantial price reductions for the innovator product.
- Managed Care Interventions: Payer formularies may prioritize lower-cost generics, limiting access to branded products without significant rebates.
Source: Pharmaceutical pricing analyses, healthcare economics reports.
What are the regulatory pathways and timelines for FT Miconazole?
The regulatory pathway for FT Miconazole will depend on its intended use and the specific markets of interest. Standard pathways for topical pharmaceuticals in major regulatory bodies will apply.
United States (FDA):
- Pathway: New Drug Application (NDA) for novel formulations or significant new indications.
- Estimated Timeline: For a new formulation with robust clinical data, the review process typically takes 12-18 months after submission.
- Key Requirements: Comprehensive preclinical data, Phase I, II, and III clinical trials demonstrating safety and efficacy, manufacturing controls (CMC).
- Exclusivity: Upon NDA approval, 5 years of data exclusivity for novel drugs. If designated a "new chemical entity," 5 years. If it contains an already approved active moiety but is a new formulation, the exclusivity period might be shorter or not granted if it's considered a "new ester, new salt, new complex, new solvate, new hydrate" of an approved drug. However, novel delivery systems can qualify for exclusivity.
European Union (EMA):
- Pathway: Marketing Authorisation Application (MAA) via the centralized procedure.
- Estimated Timeline: 12-18 months for review after submission.
- Key Requirements: Similar to FDA, requiring extensive preclinical and clinical data.
- Exclusivity: 10 years of data exclusivity from the date of marketing authorization, extendable to 11 years under specific conditions, and up to 8 years of market exclusivity (non-extendable in most cases without further conditions).
Japan (PMDA):
- Pathway: New Drug Application.
- Estimated Timeline: 12-24 months for review.
- Key Requirements: Preclinical and clinical data, with an emphasis on local efficacy and safety data relevant to the Japanese population.
- Exclusivity: 6 years of market exclusivity for new drugs.
China (NMPA):
- Pathway: New Drug Registration Application.
- Estimated Timeline: Can range from 18-36 months due to complex review processes.
- Key Requirements: Extensive preclinical and clinical data, with local clinical trials often required.
- Exclusivity: Typically 6 years of market exclusivity for approved new drugs.
Patent Term Extension (PTE):
In the US, patent term can be extended to compensate for regulatory review delays. In Europe, Supplementary Protection Certificates (SPCs) offer similar protection. The length of PTE/SPC depends on the duration of the regulatory review period. This can extend market exclusivity beyond the original patent expiration dates, especially for patents nearing expiry at the time of approval.
Source: Regulatory agency guidelines, patent law summaries.
What are the key risks and opportunities for FT Miconazole?
The development and commercialization of FT Miconazole present both significant opportunities and inherent risks.
Opportunities:
- Unmet Medical Need: Addressing specific limitations of current antifungal therapies, such as resistance, poor tolerability, or suboptimal efficacy in difficult-to-treat infections.
- Market Growth: Capitalizing on the expanding global antifungal market, particularly the topical segment.
- Pipeline Expansion: Developing new indications or formulations based on the core FT Miconazole technology (e.g., combination therapies, extended-release formulations for chronic conditions).
- Strategic Partnerships: Licensing opportunities or co-development deals with larger pharmaceutical companies to accelerate market entry and expand commercial reach.
- First-to-Market Advantage: Securing early market exclusivity for a differentiated product can establish strong brand recognition and customer loyalty.
Risks:
- Clinical Trial Failure: Failure to demonstrate statistically significant efficacy or an unacceptable safety profile in pivotal clinical trials.
- Regulatory Hurdles: Delays or outright rejection by regulatory agencies due to incomplete data, manufacturing issues, or safety concerns.
- Patent Litigation: Challenges to patent validity or infringement claims from competitors, potentially leading to early market entry for generics or biosimilars.
- Intense Competition: The presence of established generic and branded antifungals may limit market penetration and pricing power.
- Reimbursement Challenges: Difficulty securing favorable formulary placement and reimbursement rates from payers, impacting patient access and net revenue.
- Manufacturing Scale-Up: Challenges in scaling up production of novel formulations reliably and cost-effectively.
- Emergence of Resistance: The development of antifungal resistance to miconazole or FT Miconazole over time could limit its long-term utility.
Source: Industry risk assessments, drug development strategy analyses.
Key Takeaways
FT Miconazole's patent portfolio, while robust, requires careful management to maximize market exclusivity, with key patents expiring between 2035 and 2042. The topical antifungal market is projected for sustained growth, presenting a significant opportunity for FT Miconazole to capture substantial market share, potentially reaching $600 million to $1.1 billion by 2030. Pricing will be strategically positioned above generics, reflecting therapeutic differentiation and competitive benchmarks, with specialty formulations commanding higher price points. Regulatory pathways in key markets (US, EU, Japan, China) will dictate timelines ranging from 12-36 months. Navigating clinical, regulatory, and competitive risks is crucial for successful commercialization.
FAQs
What are the primary therapeutic areas FT Miconazole is intended for?
FT Miconazole is developed for the treatment of superficial fungal infections, including dermatophytosis (e.g., athlete's foot, ringworm), cutaneous candidiasis, and onychomycosis.
How does FT Miconazole differentiate itself from existing miconazole products?
FT Miconazole's differentiation lies in its novel formulation designed for enhanced topical delivery, improved efficacy, and potentially reduced systemic absorption compared to standard miconazole preparations.
What is the estimated period of market exclusivity for FT Miconazole in the US?
Based on current patent filings, market exclusivity in the US for specific formulations of FT Miconazole is projected to extend until at least 2035, with potential for longer protection through pending applications and patent term extensions.
What are the main challenges anticipated in achieving favorable reimbursement for FT Miconazole?
Key challenges include demonstrating clear pharmacoeconomic advantages over existing treatments, navigating payer formularies, and securing adequate coverage from government and private insurers, especially in the face of lower-cost generic alternatives.
How might the development of antifungal resistance impact FT Miconazole's long-term market potential?
The emergence of resistance to miconazole or FT Miconazole could diminish its efficacy over time, potentially limiting its lifespan as a frontline treatment and necessitating a shift in therapeutic strategy or the development of next-generation agents.
Citations
[1] Company annual reports and investor presentations. (Various years). [2] Global antifungal market research reports. (2023-2024). [3] Pharmaceutical patent databases (e.g., USPTO, EPO, WIPO). (Accessed Q4 2023). [4] Food and Drug Administration (FDA) guidelines for drug approval. (Current edition). [5] European Medicines Agency (EMA) guidelines for marketing authorization. (Current edition). [6] Pharmaceuticals and Medical Devices Agency (PMDA) guidelines for drug approval. (Current edition). [7] National Medical Products Administration (NMPA) guidelines for drug registration. (Current edition).
More… ↓
