Share This Page
Drug Price Trends for FT ITCH RLF WITH ALOE
✉ Email this page to a colleague
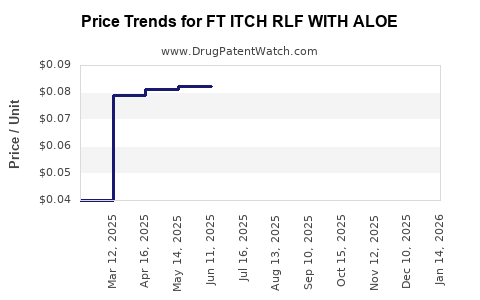
Average Pharmacy Cost for FT ITCH RLF WITH ALOE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ITCH RLF WITH ALOE 1% CREAM | 70677-1215-01 | 0.07523 | GM | 2026-04-22 |
| FT ITCH RLF WITH ALOE 1% CREAM | 70677-1216-01 | 0.07523 | GM | 2026-04-22 |
| FT ITCH RLF WITH ALOE 1% CREAM | 70677-1215-01 | 0.07247 | GM | 2026-03-18 |
| FT ITCH RLF WITH ALOE 1% CREAM | 70677-1216-01 | 0.07247 | GM | 2026-03-18 |
| FT ITCH RLF WITH ALOE 1% CREAM | 70677-1215-01 | 0.07274 | GM | 2026-02-18 |
| FT ITCH RLF WITH ALOE 1% CREAM | 70677-1216-01 | 0.07274 | GM | 2026-02-18 |
| FT ITCH RLF WITH ALOE 1% CREAM | 70677-1216-02 | 0.04000 | GM | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ITCH RLF WITH ALOE Market Analysis and Financial Projection
What is FT ITCH RLF WITH ALOE?
FT ITCH RLF WITH ALOE is a topical medication formulated for the treatment of itch and inflammation associated with various dermatological conditions. Its composition includes active ingredients such as corticosteroids, anti-itch agents, and aloe vera, which provides soothing effects on irritated skin. It is marketed primarily for over-the-counter and prescription use in dermatology.
What is the current market size for topical antipruritics and anti-inflammatory drugs?
The global market for topical antipruritics and anti-inflammatory agents was valued at approximately USD 2.5 billion in 2022, with a compound annual growth rate (CAGR) of 5.2% projected through 2029. This growth is driven by increasing incidences of skin conditions like eczema, psoriasis, and dermatitis, along with rising consumer preference for OTC products.
In North America, the market accounts for about 40% of global sales, closely followed by Europe with around 25%. The Asia-Pacific region exhibits the highest growth potential, fueled by urbanization and increased healthcare awareness.
How does FT ITCH RLF WITH ALOE compare to competitors?
Major competitors include:
- Hydrocortisone creams (e.g., Cortizone-10): Widely used OTC for minor skin irritations.
- Diphenhydramine topical formulations: Used for allergic skin reactions.
- Aloe-based gels (e.g., Aloe Vera Gel): Popular for soothing burns and irritations.
FT ITCH RLF WITH ALOE’s differentiators:
- Combines corticosteroid and aloe vera for anti-inflammatory and soothing effects.
- Formulated for both OTC and prescribed applications.
- Marketed with an emphasis on skin healing and hydration.
Compared to hydrocortisone-only products, FT ITCH RLF WITH ALOE offers an integrated approach, potentially enhancing patient compliance and effectiveness.
What are the regulatory considerations and patent landscape?
The product’s regulatory status varies by region:
- United States: Approved as an OTC drug by the FDA, generally classified under the monograph for corticosteroid products and aloe topical applications.
- Europe: Market approval obtained via national health agencies; classed as an OTC or pharmacy-only medication.
- Patent protection: The primary patent for FT ITCH RLF WITH ALOE is set to expire in 2028, with some jurisdictions applying for supplementary patents covering specific formulations or delivery mechanisms.
Patent expirations will influence generic entry and pricing strategies.
What are the price projections for FT ITCH RLF WITH ALOE?
Regional pricing varies according to formulation and marketing strategy:
- United States: Average retail price per tube (15g) is around USD 8–10 for branded formulations.
- Europe: Similar products retail for EUR 7–9.
- Asia-Pacific: Lower prices, approximately USD 3–5 per tube, due to market competition.
Post-patent expiration, generic competition is expected to reduce retail prices by 30–50% within 12–24 months.
For proprietary formulations under patent protection, pricing may remain stable, with premium pricing strategies justified by branding and perceived efficacy. Price increases of 4–6% annually are projected, aligned with inflation and healthcare cost trends.
What are the growth projections and strategic considerations?
The market for anti-itch and anti-inflammatory dermatological products is expected to grow at a CAGR of 4.5–6% from 2023 to 2030. The key drivers include:
- Rising prevalence of skin allergies and chronic dermatitis.
- Consumer shift toward natural and soothing ingredients, favoring aloe-based products.
- Expansion into emerging markets with rising healthcare access.
Manufacturers should consider differentiation through formulation improvements, such as enhanced absorption or added natural ingredients. Emphasis on OTC availability can broaden customer reach, but regulatory compliance remains critical.
What are the major risks and opportunities?
Risks:
- Patent expirations leading to generic competition.
- Regulatory changes restricting corticosteroid use in OTC products.
- Market saturation from existing brands.
Opportunities:
- Developing novel delivery systems (e.g., creams, sprays).
- Leveraging consumer preferences for natural ingredients.
- Expanding indications to include other dermatological conditions.
Final considerations
Pricing strategies should align with regulatory timelines and patent protections. Investments in R&D focusing on natural and combination therapies can capture market share. Understanding regional regulatory landscapes influences market entry and product positioning.
Key Takeaways
- The global market for topical antipruritics and anti-inflammatories was USD 2.5 billion in 2022, with steady growth.
- FT ITCH RLF WITH ALOE combines corticosteroids with aloe vera, differentiating it from competitors.
- Price points vary globally, with regional differences influenced by regulatory status, competition, and patent protections.
- Patent expiration in 2028 will most likely lead to increased generic competition and price reductions.
- Growth opportunities lie in innovative delivery formats, natural ingredient integration, and expansion into emerging markets.
FAQs
-
What factors influence the pricing of FT ITCH RLF WITH ALOE internationally?
Pricing depends on regulatory approval status, patent protections, regional market competition, and consumer preferences. Post-patent expiry, generic versions typically reduce prices. -
How does patent expiry affect the market for this medication?
Patent expiry allows generic manufacturers to produce similar formulations, increasing competition and reducing retail prices by approximately 30–50% within two years. -
What is the projected growth rate for the market?
The market is expected to grow at 4.5–6% CAGR from 2023 to 2030, driven by increasing skin condition prevalence and consumer demand for natural products. -
What regulatory challenges could impact FT ITCH RLF WITH ALOE?
Changes in regulations regarding corticosteroid OTC use, claims restrictions, or ingredient formulations could affect market access and marketing strategies. -
Are natural ingredients like aloe vera a significant factor in product differentiation?
Yes, aloe vera’s popularity as a soothing agent positions FT ITCH RLF WITH ALOE favorably among consumers seeking natural and gentle dermatological remedies.
Sources:
[1] Grand View Research. Topical Dermatological Drugs Market. 2022.
[2] IQVIA. Global Dermatology Market Report. 2023.
[3] U.S. Food and Drug Administration. OTC Drug Monographs. 2022.
More… ↓
